Abstract
Background
The aldosterone-to-renin activity ratio (ARR) is recommended as a screening test for primary aldosteronism (PA). The use of antihypertensive medication is one of the most important factors to take into account when interpreting the ARR.
Objectives
The goal of this study was to compare the effect of various antihypertensive drug classes on biochemical testing and provide practical recommendations for antihypertensive medication regimens in patients suspected of having PA.
Methods
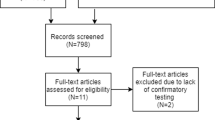
We retrospectively investigated 4218 hypertensive patients who underwent PA detection between January 2020 and February 2023. Finally, 25 patients with essential hypertension (EH) and 39 with PA were involved. The enrolled EH patients were selected from EH patients with at least two screening tests with interfering medication changes for at least 4 weeks.
Results
A total of 2/18 (11.1%) EH patients had opposite screening results after angiotensin II receptor blockers (ARBs), angiotensin-converting enzyme inhibitors (ACEIs), dihydropyridine-calcium channel blockers (DHP-CCBs) and diuretics were withdrawn. A total of 3/3 (100%) of the patients were initially screened as positive but had results that tested negative after β-blockers withdrawal. A total of 3/39 (7.7%) PA patients were misdiagnosed with EH with drug effects. Only plasma renin activity was markedly reduced after antihypertensive medication washout (P = 0.0173).
Conclusions
Not every patient receiving β-blockers, ACEIs, ARBs and DHP-CCBs with negative reports needed withdrawal or switching antihypertensive drugs. We recommend that patients be left on a regimen that includes β-blockers, ACEIs, ARBs, DHP-CCBs and spironolactone when determining the ARR during the initial test.



Similar content being viewed by others
References
J.M. Brown, M. Siddiqui, D.A. Calhoun, R.M. Carey, P.N. Hopkins, G.H. Williams, A. Vaidya, The unrecognized prevalence of primary aldosteronism: a cross-sectional study. Ann. Intern. Med. 173(1), 10–20 (2020)
K. Zekarias, K.M. Tessier, Screening rate for primary aldosteronism among patients with apparent treatment-resistant hypertension: retrospective analysis of current practice. Endocr. Pract. 28(3), 271–275 (2022)
Z. Xu, J. Yang, J. Hu, Y. Song, W. He, T. Luo, Q. Cheng, L. Ma, R. Luo, P.J. Fuller, J. Cai, Q. Li, S. Yang; Chongqing Primary Aldosteronism Study (CONPASS) Group, Primary aldosteronism in patients in China with recently detected hypertension. J. Am. Coll. Cardiol. 75(16), 1913–1922 (2020)
L. Zhang, J. Li, N. Li, N. Sun, L. Xie, Q. Han, Y. Li, X.Z. Lu, P. Sun, Y. Li, Y. Shi, H. Wang, Y. Zhang, H. Chen, Y. Huo, Trends in cause-related comorbidities in hospitalized patients with secondary hypertension in China from 2013 to 2016: a retrospective analysis of hospital quality monitoring system data. J. Hypertens. 39(10), 2015–2021 (2021)
S. Monticone, F. D’Ascenzo, C. Moretti, T.A. Williams, F. Veglio, F. Gaita, P. Mulatero, Cardiovascular events and target organ damage in primary aldosteronism compared with essential hypertension: a systematic review and meta-analysis. Lancet Diabetes Endocrinol. 6(1), 41–50 (2018)
T.A. Williams, J.W.M. Lenders, P. Mulatero, J. Burrello, M. Rottenkolber, C. Adolf, F. Satoh, L. Amar, M. Quinkler, J. Deinum, F. Beuschlein, K.K. Kitamoto, U. Pham, R. Morimoto, H. Umakoshi, A. Prejbisz, T. Kocjan, M. Naruse, M. Stowasser, T. Nishikawa, W.F. Young Jr, C.E. Gomez-Sanchez, J.W. Funder, M. Reincke; Primary Aldosteronism Surgery Outcome (PASO) investigators, Outcomes after adrenalectomy for unilateral primary aldosteronism: an international consensus on outcome measures and analysis of remission rates in an international cohort. Lancet Diabetes Endocrinol. 5(9), 689–699 (2017)
K. Hiramatsu, T. Yamada, Y. Yukimura, I. Komiya, K. Ichikawa, M. Ishihara, H. Nagata, T. Izumiyama, A screening test to identify aldosterone-producing adenoma by measuring plasma renin activity. Results in hypertensive patients. Arch. Intern. Med. 141(12), 1589–1593 (1981)
T. Unger, C. Borghi, F. Charchar, N.A. Khan, N.R. Poulter, D. Prabhakaran, A. Ramirez, M. Schlaich, G.S. Stergiou, M. Tomaszewski, R.D. Wainford, B. Williams, A.E. Schutte, 2020 International Society of Hypertension Global Hypertension Practice Guidelines. Hypertension 75(6), 1334–1357 (2020)
H. Schunkert, H.W. Hense, U. Bröckel, A. Luchner, M. Muscholl, S.R. Holmer, A.H. Danser, B. Mayer, G.A. Riegger, Differential effects of antihypertensive drugs on neurohormonal activation: insights from a population-based sample. J. Intern. Med. 244(2), 109–119 (1998)
P. Mulatero, S. Monticone, J. Deinum, L. Amar, A. Prejbisz, M.C. Zennaro, F. Beuschlein, G.P. Rossi, T. Nishikawa, A. Morganti, T.M. Seccia, Y.H. Lin, F. Fallo, J. Widimsky, Genetics, prevalence, screening and confirmation of primary aldosteronism: a position statement and consensus of the Working Group on Endocrine Hypertension of The European Society of Hypertension. J. Hypertens. 38(10), 1919–1928 (2020)
G.P. Rossi, V. Bisogni, A.V. Bacca, A. Belfiore, M. Cesari, A. Concistrè et al. The 2020 Italian Society of Arterial Hypertension (SIIA) practical guidelines for the management of primary aldosteronism. Int. J. Cardiol. Hypertens. 15(5), 100029 (2020)
J.W. Funder, R.M. Carey, F. Mantero, M.H. Murad, M. Reincke, H. Shibata, M. Stowasser, W.F. Young Jr, The management of primary aldosteronism: case detection, diagnosis, and treatment: an Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 101(5), 1889–1916 (2016)
C. Seifarth, S. Trenkel, H. Schobel, E.G. Hahn, J. Hensen, Influence of antihypertensive medication on aldosterone and renin concentration in the differential diagnosis of essential hypertension and primary aldosteronism. Clin. Endocrinol. (Oxf.) 57(4), 457–465 (2002)
P. Mulatero, F. Rabbia, A. Milan, C. Paglieri, F. Morello, L. Chiandussi, F. Veglio, Drug effects on aldosterone/plasma renin activity ratio in primary aldosteronism. Hypertension 40(6), 897–902 (2002)
Y. Tezuka, A.F. Turcu, Mineralocorticoid receptor antagonists decrease the rates of positive screening for primary aldosteronism. Endocr. Pract. 26(12), 1416–1424 (2020)
P. Jędrusik, B. Symonides, J. Lewandowski, Z. Gaciong, The effect of antihypertensive medications on testing for primary aldosteronism. Front. Pharm. 13(12), 684111 (2021)
P. Mulatero, C. Bertello, F. Veglio, S. Monticone, Approach to the patient on antihypertensive therapy: screen for primary aldosteronism. J. Clin. Endocrinol. Metab. 107(11), 3175–3181 (2022)
Y. Song, S. Yang, W. He, J. Hu, Q. Cheng, Y. Wang, T. Luo, L. Ma, Q. Zhen, S. Zhang, M. Mei, Z. Wang, H. Qing, D. Bruemmer, B. Peng, Q. Li; Chongqing Primary Aldosteronism Study (CONPASS) Group, Confirmatory tests for the diagnosis of primary aldosteronism: a prospective diagnostic accuracy study. Hypertension 71(1), 118–124 (2018)
W.F. Young Jr, Diagnosis and treatment of primary aldosteronism: practical clinical perspectives. J. Intern. Med. 285(2), 126–148 (2019)
T.A. Williams et al. Primary Aldosteronism Surgery Outcome (PASO) investigators. Outcomes after adrenalectomy for unilateral primary aldosteronism: an international consensus on outcome measures and analysis of remission rates in an international cohort. Lancet Diabetes Endocrinol. 5(9), 689–699 (2017)
H. Kobayashi et al. Development and validation of subtype prediction scores for the workup of primary aldosteronism. J. Hypertens. 36(11), 2269–2276 (2018)
B.J. Gallay, S. Ahmad, L. Xu, B. Toivola, R.C. Davidson, Screening for primary aldosteronism without discontinuing hypertensive medications: plasma aldosterone-renin ratio. Am. J. Kidney Dis. 37(4), 699–705 (2001)
P.M. Jansen, B.J. van den Born, W.J. Frenkel, E.L. de Bruijne, J. Deinum, M.N. Kerstens, Y.M. Smulders, A.J. Woittiez, J.A. Wijbenga, R. Zietse, A.H. Danser, A.H. van den Meiracker, Test characteristics of the aldosterone-to-renin ratio as a screening test for primary aldosteronism. J. Hypertens. 32(1), 115–126 (2014)
J.W. Funder, R.M. Carey, C. Fardella, C.E. Gomez-Sanchez, F. Mantero, M. Stowasser, W.F. Young Jr, V.M. Montori, Endocrine Society. Case detection, diagnosis, and treatment of patients with primary aldosteronism: an endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 93(9), 3266–3281 (2008)
A. Tanabe, M. Naruse, S. Takagi, K. Tsuchiya, T. Imaki, K. Takano, Variability in the renin/aldosterone profile under random and standardized sampling conditions in primary aldosteronism. J. Clin. Endocrinol. Metab. 88(6), 2489–2494 (2003)
N. Yozamp, G.L. Hundemer, M. Moussa, J. Underhill, T. Fudim, B. Sacks, A. Vaidya, Intraindividual variability of aldosterone concentrations in primary aldosteronism: implications for case detection. Hypertension 77(3), 891–899 (2021)
S. Niizuma, H. Nakahama, K. Kamide, K. Fukuchi, Y. Iwanaga, H. Nakata, F. Yoshihara, T. Horio, S. Nakamura, Y. Kawano, The cutoff value of aldosterone-to-renin ratio for the diagnosis of primary aldosteronism in patients taking antihypertensive medicine. Clin. Exp. Hypertens. 30(7), 640–647 (2008)
W.F. Young, Primary aldosteronism: renaissance of a syndrome. Clin. Endocrinol. (Oxf.) 66(5), 607–618 (2007)
Funding
The authors declare financial support was received for the research, authorship. Ningbo Key Clinical Specialty (Endocrinology) (Grant No. 2022-B07).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Q.Z. provided the initial experimental design. Material preparation, data collection were performed by J.L., L.L. and C.Z. Analysis was performed by H.Y, X.L. and Z.Z. The first draft of the manuscript was written by J.L. and Q.Z. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Consent to publish/participate
The authors affirm that human research participants provided informed consent for the publication of Table 1 and Figs.1–3.
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of the First Affiliated Hospital of Ningbo University (June 23, 2023/084RS).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, J., Yan, H., Li, L. et al. Practical recommendations for antihypertensive therapy during the primary aldosteronism screening test. Endocrine 83, 188–195 (2024). https://doi.org/10.1007/s12020-023-03580-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-023-03580-8