Abstract
Purpose
The challenge in the diagnosis and treatment of thyroid carcinoma is to correctly classify neoplasias with overlapping features and to identify the high-risk patients among those with a less aggressive form, in order to personalize the treatment of thyroid carcinoma patients accordingly.
Methods
MiR-203a-3p, miR-204-3p, and miR-222-3p levels were determined in 99 cases of thyroid neoplasias (77 papillary thyroid carcinomas (PTC) of diverse variants, 12 follicular thyroid adenomas (FTA) and 10 nodular goiters (NG)) along with 99 adjacent non-malignant thyroid tissues using quantitative RT-PCR. The results were evaluated in comparison with the clinicopathological features of the patients and available TCGA data.
Results
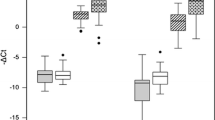
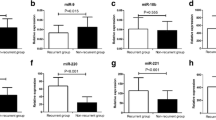
Down-regulated miR-203a-3p indicates the presence of thyroid tumor (PTC or FTA) with high sensitivity (75%) and specificity (73%), while its up-regulation indicates NG. If miR-203a-3p is down-regulated, up-regulated miR-204-3p with high sensitivity (83.3%) and specificity (74.4%) indicates FTA presence, while up-regulated miR-222-3p, with high sensitivity (76.6%) and specificity (75.0%), points to PTC. The expression of miR-204-3p and miR-222-3p depends on the PTC subtype (P < 0.05). While the deregulated expression of tested miRs is associated with a long-range of unfavorable clinicopathological parameters of PTC, only abundant expression of miR-222-3p may be used as an independent predictive factor for the presence of extrathyroid invasion and advanced pTNM stage of PTC (P < 0.05).
Conclusion
Successive evaluation of miR-203a-3p, miR-204-3p, and miR-222-3p expression can help in the differential diagnosis of thyroid neoplasias. A high relative value of miR-222-3p expression is an independent predictive factor for the presence of extrathyroid invasion and advanced pTNM stage of PTC. The panel consisting of miR-203a-3p, miR-204-3p, and miR-222-3p could be used as a diagnostic and prognostic tool for personalizing the treatment of thyroid cancer patients.




Similar content being viewed by others
Data availability
The data supporting the findings of this study are available from the corresponding author, upon reasonable request.
References
J.M. Cameselle-Teijeiro, C. Eloy, M. Sobrinho-Simões, Pitfalls in Challenging Thyroid Tumors: emphasis on Differential Diagnosis and Ancillary Biomarkers. Endocr. Pathol 31(3), 197–217 (2020). https://doi.org/10.1007/s12022-020-09638-x
S. Suster, Thyroid tumors with a follicular growth pattern: problems in differential diagnosis. Arch. Pathol. Lab. Med. 130(7), 984–988 (2006). https://doi.org/10.5858/2006-130-984-TTWAFG
O. Mete, S.L. Asa, Pitfalls in the diagnosis of follicular epithelial proliferations of the thyroid. Adv. Anat. Pathol. 19(6), 363–373 (2012). https://doi.org/10.1097/PAP.0b013e318271a5ac.
L. Davies, H.G. Welch, Current thyroid cancer trends in the United States. JAMA Otolaryngol. Head. Neck Surg. 140(4), 317–322 (2014). https://doi.org/10.1001/jamaoto.2014.1
R.L. Siegel, K.D. Miller, A. Jemal, Cancer Statistics, 2017. CA Cancer J. Clin. 67(1), 7–30 (2017). https://doi.org/10.3322/caac.21387
V.A. LiVolsi, J. Albores-Saavedra, S.L. Asa et al. Papillary carcinoma. In: ed. R. De Lellis, R. Lloyd, P.U. Heitz et al. WHO Classification of Tumors. Pathology and Genetics of Tumours of Endocrine Organs. (IARC Press, Lyon, France, 2004). p. 57–66.
M.R. Pelizzo, I.M. Boschin, A. Toniato et al. Papillary thyroid carcinoma: 35-year outcome and prognostic factors in 1858 patients. Clin. Nucl. Med. 32(6), 440–444 (2007). https://doi.org/10.1097/RLU.0b013e31805375ca
Y. Ito, A. Miyauchi, M. Kihara et al. Overall survival of papillary thyroid carcinoma patients: a single-institution longterm follow-up of 5897 patients. World J. Surg. 42, 615e22 (2018). https://doi.org/10.1007/s00268-018-4479-z
F.D. Gilliland, W.C. Hunt, D.M. Morris et al. Prognostic factors for thyroid carcinoma. A population-based study of 15,698 cases from the Surveillance, Epidemiology and End Results (SEER) program 1973-1991. Cancer 79(3), 564–573 (1997). https://doi.org/10.1002/(sici)1097-0142(19970201)79:3<564::aid-cncr20>3.0.co;2-0
V. Ambros, The functions of animal microRNAs. Nature 431(7006), 350–355 (2004). https://doi.org/10.1038/nature02871.
K. Saliminejad, H.R. Khorram Khorshid, S. Soleymani Fard et al. An overview of microRNAs: biology, functions, therapeutics, and analysis methods. J. Cell Physiol. 234(5), 5451–5465 (2019). https://doi.org/10.1002/jcp.27486.
W.P. Kloosterman, R.H. Plasterk, The diverse functions of microRNAs in animal development and disease. Dev. Cell. 11(4), 441–450 (2006). https://doi.org/10.1016/j.devcel.2006.09.009.
Z. Ali Syeda, S.S.S. Langden, C. Munkhzul et al. Regulatory Mechanism of MicroRNA Expression in Cancer. Int. J. Mol. Sci. 21(5), 1723 (2020). https://doi.org/10.3390/ijms21051723.
T.A. Farazi, J.I. Hoell, P. Morozov et al. MicroRNAs in human cancer. Adv. Exp. Med. Biol. 774, 1–20 (2013). https://doi.org/10.1007/978-94-007-5590-1_1.
Y.S. Lee, A. Dutta, MicroRNAs in cancer. Annu. Rev. Pathol. 4, 199–227 (2009). https://doi.org/10.1146/annurev.pathol.4.110807.092222.
K. Santiago, Y. Chen Wongworawat, S. Khan. Differential MicroRNA-Signatures in Thyroid Cancer Subtypes. J. Oncol. 2052396 (2020). https://doi.org/10.1155/2020/2052396.
M. Celano, F. Rosignolo, V. Maggisano, et al. MicroRNAs as Biomarkers in Thyroid Carcinoma. Int. J. Genom. 6496570 (2017). https://doi.org/10.1155/2017/6496570.
M.S. Dettmer, A. Perren, H. Moch et al. MicroRNA profile of poorly differentiated thyroid carcinomas: new diagnostic and prognostic insights. J. Mol. Endocrinol. 52(2), 181–189 (2014). https://doi.org/10.1530/JME-13-0266
S. Schwertheim, S.Y. Sheu, K. Worm et al. Analysis of deregulated miRNAs is helpful to distinguish poorly differentiated thyroid carcinoma from papillary thyroid carcinoma. Horm. Metab. Res. 41(6), 475–481 (2009). https://doi.org/10.1055/s-0029-1215593
L. Yang, H. Liang, Y. Wang et al. MiRNA‐203 suppresses tumor cell proliferation, migration and invasion by targeting Slug in gastric cancer. Protein Cell 7(5), 383–387 (2016). https://doi.org/10.1007/s13238-016-0259-4
A. You, L. Fu, Y. Li et al. MicroRNA-203 restrains epithelial-mesenchymal transition, invasion and migration of papillary thyroid cancer by downregulating AKT3. Cell Cycle 19(10), 1105–1121 (2020). https://doi.org/10.1080/15384101.2020.1746490
J. Zhu, X. Zheng, X. Yang. Diagnostic and mechanistic values of microRNA-130a and microRNA-203 in patients with papillary thyroid carcinoma. J. Cell Biochem. Online ahead of print (2019). https://doi.org/10.1002/jcb.29498.
F. Le, P. Luo, Q. Ouyang et al. WT1-AS Downregulates Survivin by Upregulating miR-203 in Papillary Thyroid Carcinoma. Cancer Manag. Res. 12, 443–449 (2020). https://doi.org/10.2147/CMAR.S232294
X. Wu, L. Dai, Z. Zhang et al. Overexpression of microRNA-203 can downregulate survivin and function as a potential therapeutic target in papillary thyroid cancer. Oncol. Lett. 19(1), 61–68 (2020). https://doi.org/10.3892/ol.2019.11082
Z.Y. Wu, S.M. Wang, Z.H. Chen et al. MiR-204 regulates HMGA2 expression and inhibits cell proliferation in human thyroid cancer. Cancer Biomark. 15(5), 535–542 (2015). https://doi.org/10.3233/CBM-150492
L. Liu, J. Wang, X. Li et al. MiR-204-5p suppresses cell proliferation by inhibiting IGFBP5 in papillary thyroid carcinoma. Biochem. Biophys. Res. Commun. 457(4), 621–626 (2015). https://doi.org/10.1016/j.bbrc.2015.01.037
F. Xia, W. Wang, B. Jiang et al. DNA methylation-mediated silencing of miR-204 is a potential prognostic marker for papillary thyroid carcinoma. Cancer Manag. Res. 11, 1249–1262 (2019). https://doi.org/10.2147/CMAR.S184566
M. Ye, S. Dong, H. Hou et al. Oncogenic Role of Long Noncoding RNAMALAT1 in Thyroid Cancer Progression through Regulation of the miR-204/IGF2BP2/m6A-MYC Signaling. Mol. Ther. Nucleic Acids 23, 1–12 (2020). https://doi.org/10.1016/j.omtn.2020.09.023
S.B. Edge, D.R. Byrd, C.C. Compton, editors: AJCC cancer staging manual. 7th edn. (Springer, New York, NY, 2010). p. 87–96
F. Basolo, L. Torregrossa, R. Giannini et al. Correlation between the BRAF V600E mutation and tumor invasiveness in papillary thyroid carcinomas smaller than 20 millimeters: analysis of 1060 cases. J. Clin. Endocrinol. Metab. 95(9), 4197–4205 (2010). https://doi.org/10.1210/jc.2010-0337
Y. Ito, T. Kudo, K. Kobayashi et al. Prognostic factors for recurrence of papillary thyroid carcinoma in the lymph nodes, lung, and bone: analysis of 5,768 patients with average 10-year follow-up. World J. Surg. 36(6), 1274–1278 (2012). https://doi.org/10.1007/s00268-012-1423-5
M.J. Goldman, B. Craft, M. Hastie et al. Visualizing and interpreting cancer genomics data via the Xena platform. Nat. Biotechnol. (2020). https://doi.org/10.1038/s41587-020-0546-8.
M.N. Nikiforova, G.C. Tseng, D. Steward et al. MicroRNA expression profiling of thyroid tumors: biological significance and diagnostic utility. J. Clin. Endocrinol. Metab. 93(5), 1600–1608 (2008). https://doi.org/10.1210/jc.2007-2696
F. Rosignolo, L. Memeo, F. Monzani et al. MicroRNA-based molecular classification of papillary thyroid carcinoma. Int. J. Oncol. 50(5), 1767–1777 (2017). https://doi.org/10.3892/ijo.2017.3960
Y. Lin, J. Jiang, Long non-coding RNA LINC00704 promotes cell proliferation, migration, and invasion in papillary thyroid carcinoma via miR-204-5p/HMGB1 axis. Open Life Sci. 15(1), 561–571 (2020). https://doi.org/10.1515/biol-2020-0057
M. Dettmer, A. Perren, H. Moch et al. Comprehensive MicroRNA expression profiling identifies novel markers in follicular variant of papillary thyroid carcinoma. Thyroid 23(11), 1383–1389 (2013). https://doi.org/10.1089/thy.2012.0632
Z. Wang, H. Zhang, L. He et al. Association between the expression of four upregulated miRNAs and extrathyroidal invasion in papillary thyroid carcinoma. OncoTargets Ther. 6, 281–287 (2013). https://doi.org/10.2147/OTT.S43014
J.C. Lee, J.T. Zhao, R.J. Clifton-Bligh et al. MicroRNA-222 and microRNA-146b are tissue and circulating biomarkers of recurrent papillary thyroid. Cancer Cancer 119(24), 4358–4365 (2013). https://doi.org/10.1002/cncr.28254
L. Yip, L. Kelly, Y. Shuai et al. MicroRNA signature distinguishes the degree of aggressiveness of papillary thyroid carcinoma. Ann. Surg. Oncol. 18(7), 2035–2041 (2011). https://doi.org/10.1245/s10434-011-1733-0
A. Kondrotienė, A. Daukša, D. Pamedytytė et al. Papillary Thyroid Carcinoma Tissue miR-146b, -21, -221, -222, -181b Expression in Relation with Clinicopathological Features. Diagnostics (Basel) 11(3), 418 (2021). https://doi.org/10.3390/diagnostics11030418
Acknowledgements
The authors wish to express their gratitude to Dubravka Cvejić, PhD for the great help in revising the paper.
Funding
The Ministry of Education, Science and Technological Development of the Republic of Serbia, no. 451-03-68/2022-14/ 200019.
Author information
Authors and Affiliations
Contributions
S.S.—investigation, data curation, writing original draft; Z.D.—methodology, validation; S.Š.—project administration; I.Đ.—writing - review & editing; J.J.M.—software, visualization; V.Ž.—resources; T.I.D.—conceptualization, formal analysis, supervision, writing - review & editing. All authors read and approved the final paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
This study was conducted in accordance with the Helsinki Declaration and was approved by the Ethics Committee at the Center for Endocrine Surgery, Clinical Center of Serbia, Belgrade, Serbia (695/7-A).
Consent to participate
Written informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Stojanović, S., Dobrijević, Z., Šelemetjev, S. et al. MiR-203a-3p, miR-204-3p, miR-222-3p as useful diagnostic and prognostic tool for thyroid neoplasia spectrum. Endocrine 79, 98–112 (2023). https://doi.org/10.1007/s12020-022-03185-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-022-03185-7