Abstract
Purpose
Anti-CD20 therapy delays type 1 diabetes mellitus (T1DM) progression in both nonobese diabetic (NOD) mice and new-onset patients. The mechanism is not completely defined. This study aimed to investigate the effects of anti-CD20 therapy on T helper 17 (Th17) cells and regulatory T cells (Tregs) in NOD mice. The role of B cell depletion in T1DM development was also examined.
Methods
NOD mice were randomly divided into two groups. The mice in the experimental group were treated with an anti-CD20 antibody, while the control mice were treated with an isotype-matched control antibody. After treatment, islet morphology and inflammation, Th17 and Treg cell frequencies in the pancreas and spleen, serum cytokine and anti-glutamic acid decarboxylase (GAD) antibody levels, interleukin (IL)-17A levels in the pancreas and spleen, insulin expression in islet cells and islet β cell function were measured.
Results
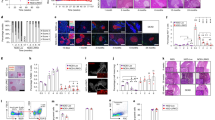
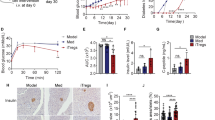
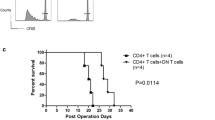
Decreased blood glucose and increased insulin secretion were found in the exprimental group compared with the CON group. A lower islet inflammation score was also found in the experimental group. Decreased Th17 cell and IL-17A levels and augmented Treg cell levels were found in the spleen and pancreas after anti-CD20 treatment. The serum levels of B cell activating factor (BAFF), IL-17A, IL-17F, IL-23 and anti-GAD autoantibodies were decreased in the experimental group, while higher serum levels of IL-10 and transforming growth factor (TGF)-β were found.
Conclusion
Anti-CD20 therapy might have some beneficial effects that improve β cell function by relieving islet inflammation through regulation of Th17/Treg cells and the proinflammatory/anti-inflammatory balance.






Similar content being viewed by others
References
K.C. Herold, D.A. Vignali, A. Cooke, J.A. Bluestone, Type 1 diabetes: translating mechanistic observations into effective clinical outcomes. Nat. Rev. Immunol. 13, 243–256 (2013)
J.M. Norris, R.K. Johnson, L.C. Stene, Type 1 diabetes-early life origins and changing epidemiology. lancet Diabetes Endocrinol. 8, 226–238 (2020)
M.S. Anderson, J.A. Bluestone, The NOD mouse: a model of immune dysregulation. Annu. Rev. Immunol. 23, 447–485 (2005)
M. Pawlak, A.W. Ho, V.K. Kuchroo, Cytokines and transcription factors in the differentiation of CD4(+) T helper cell subsets and induction of tissue inflammation and autoimmunity. Curr. Opin. Immunol. 67, 57–67 (2020)
A. Abdel-Moneim, H.H. Bakery, G. Allam, The potential pathogenic role of IL-17/Th17 cells in both type 1 and type 2 diabetes mellitus. Biomedicine Pharmacother. = Biomedecine pharmacotherapie 101, 287–292 (2018)
S.M. Cabrera, M.R. Rigby, R.G. Mirmira, Targeting regulatory T cells in the treatment of type 1 diabetes mellitus. Curr. Mol. Med. 12, 1261–1272 (2012)
M. Ryba-Stanisławowska, M. Skrzypkowska, J. Myśliwska, M. Myśliwiec, The serum IL-6 profile and Treg/Th17 peripheral cell populations in patients with type 1 diabetes. Mediators Inflamm. 2013, 205284 (2013)
A. Ferraro, C. Socci, A. Stabilini, A. Valle, P. Monti, L. Piemonti, R. Nano, S. Olek, P. Maffi, M. Scavini, A. Secchi, C. Staudacher, E. Bonifacio, M. Battaglia, Expansion of Th17 cells and functional defects in T regulatory cells are key features of the pancreatic lymph nodes in patients with type 1 diabetes. Diabetes 60, 2903–2913 (2011)
S.K. O’Neill, E. Liu, J.C. Cambier, Change you can B(cell)eive in: recent progress confirms a critical role for B cells in type 1 diabetes. Curr. Opin. Endocrinol. Diabetes, Obes. 16, 293–298 (2009)
D.V. Serreze, H.D. Chapman, D.S. Varnum, M.S. Hanson, P.C. Reifsnyder, S.D. Richard, S.A. Fleming, E.H. Leiter, L.D. Shultz, B lymphocytes are essential for the initiation of T cell-mediated autoimmune diabetes: analysis of a new “speed congenic” stock of NOD.Ig mu null mice. J. Exp. Med. 184, 2049–2053 (1996)
R.M. Hinman, M.J. Smith, J.C. Cambier, B cells and type 1 diabetes…in mice and men. Immunol. Lett. 160, 128–132 (2014)
G. Zekavat, S.Y. Rostami, A. Badkerhanian, R.F. Parsons, B. Koeberlein, M. Yu, C.D. Ward, T.S. Migone, L. Yu, G.S. Eisenbarth, M.P. Cancro, A. Naji, H. Noorchashm, In vivo BLyS/BAFF neutralization ameliorates islet-directed autoimmunity in nonobese diabetic mice. J. Immunol. (Baltim., Md: 1950) 181, 8133–8144 (2008)
A. Tang, C. Li, Z. Chen, T. Li, Anti-CD20 monoclonal antibody combined with adenovirus vector-mediated IL-10 regulates spleen CD4+/CD8+ T cells and T-bet/GATA-3 expression in NOD mice. Mol. Med. Rep. 16, 3974–3982 (2017)
G. Pavlasova, M. Mraz, The regulation and function of CD20: an “enigma” of B-cell biology and targeted therapy. Haematologica 105, 1494–1506 (2020)
Y. Hamaguchi, Y. Xiu, K. Komura, F. Nimmerjahn, T.F. Tedder, Antibody isotype-specific engagement of Fcgamma receptors regulates B lymphocyte depletion during CD20 immunotherapy. J. Exp. Med. 203, 743–753 (2006)
Y. Zhao, P. Alard, M.M. Kosiewicz, High thymic output of effector CD4(+) cells may lead to a Treg: T effector imbalance in the periphery in NOD mice. J. Immunol. Res. 2019, 8785263 (2019)
T. Hayashi, K. Yoshinaka, K. Hasegawa, K. Maeda, T. Onodera, CpG oligodeoxynucleotides accelerate reovirus type 2-triggered insulitis in DBA/1 suckling mice. Int. J. Exp. Pathol. 83, 217–223 (2002)
M. Basu, K. Pandit, M. Banerjee, S.A. Mondal, P. Mukhopadhyay, S. Ghosh, Profile of auto-antibodies (disease related and other) in children with type 1 diabetes. Indian J. Endocrinol. Metab. 24, 256–259 (2020)
J. Ilonen, J. Lempainen, R. Veijola, The heterogeneous pathogenesis of type 1 diabetes mellitus. Nat. Rev. Endocrinol. 15, 635–650 (2019)
F.S. Wong, L. Wen, M. Tang, M. Ramanathan, I. Visintin, J. Daugherty, L.G. Hannum, C.A. Janeway Jr., M.J. Shlomchik, Investigation of the role of B-cells in type 1 diabetes in the NOD mouse. Diabetes 53, 2581–2587 (2004)
C.R. Smulski, H. Eibel, BAFF and BAFF-receptor in B cell selection and survival. Front. Immunol. 9, 2285 (2018)
S. Nakayamada, Y. Tanaka, BAFF- and APRIL-targeted therapy in systemic autoimmune diseases. Inflamm. regeneration 36, 6 (2016)
G.A. Lied, A. Berstad, Functional and clinical aspects of the B-cell-activating factor (BAFF): a narrative review. Scand. J. Immunol. 73, 1–7 (2011)
Y. Xiu, C.P. Wong, J.D. Bouaziz, Y. Hamaguchi, Y. Wang, S.M. Pop, R.M. Tisch, T.F. Tedder, B lymphocyte depletion by CD20 monoclonal antibody prevents diabetes in nonobese diabetic mice despite isotype-specific differences in Fc gamma R effector functions. J. Immunol. (Baltim., Md: 1950) 180, 2863–2875 (2008)
C.Y. Hu, D. Rodriguez-Pinto, W. Du, A. Ahuja, O. Henegariu, F.S. Wong, M.J. Shlomchik, L. Wen, Treatment with CD20-specific antibody prevents and reverses autoimmune diabetes in mice. J. Clin. Investig. 117, 3857–3867 (2007)
T.S. Olson, G. Bamias, M. Naganuma, J. Rivera-Nieves, T.L. Burcin, W. Ross, M.A. Morris, T.T. Pizarro, P.B. Ernst, F. Cominelli, K. Ley, Expanded B cell population blocks regulatory T cells and exacerbates ileitis in a murine model of Crohn disease. J. Clin. Investig. 114, 389–398 (2004)
J.L. Quinn, R.C. Axtell, Emerging Role of Follicular T Helper cells in multiple sclerosis and experimental autoimmune encephalomyelitis. Int. J. Mol. Sci 19, 3233 (2018).
S.A. Olalekan, Y. Cao, K.M. Hamel, A. Finnegan, B cells expressing IFN-γ suppress Treg-cell differentiation and promote autoimmune experimental arthritis. Eur. J. Immunol. 45, 988–998 (2015)
A. Bar-Or, L. Fawaz, B. Fan, P.J. Darlington, A. Rieger, C. Ghorayeb, P.A. Calabresi, E. Waubant, S.L. Hauser, J. Zhang, C.H. Smith, Abnormal B-cell cytokine responses a trigger of T-cell-mediated disease in MS? Ann. Neurol. 67, 452–461 (2010)
X. Lin, L. Lu, B cell-mediated autoimmune diseases. Adv. Exp. Med. Biol. 1254, 145–160 (2020)
D. Bhatia, A. Sinha, P. Hari, S. Sopory, S. Saini, M. Puraswani, H. Saini, D.K. Mitra, A. Bagga, Rituximab modulates T- and B-lymphocyte subsets and urinary CD80 excretion in patients with steroid-dependent nephrotic syndrome. Pediatr. Res. 84, 520–526 (2018)
C. Hu, H. Ding, X. Zhang, F.S. Wong, L. Wen, Combination treatment with anti-CD20 and oral anti-CD3 prevents and reverses autoimmune diabetes. Diabetes 62, 2849–2858 (2013)
J. Lu, J. Liu, L. Li, Y. Lan, Y. Liang, Cytokines in type 1 diabetes: mechanisms of action and immunotherapeutic targets. Clin. Transl. Immunol. 9, e1122 (2020)
J.A. Emamaullee, J. Davis, S. Merani, C. Toso, J.F. Elliott, A. Thiesen, A.M. Shapiro, Inhibition of Th17 cells regulates autoimmune diabetes in NOD mice. Diabetes 58, 1302–1311 (2009)
C.L. Langrish, Y. Chen, W.M. Blumenschein, J. Mattson, B. Basham, J.D. Sedgwick, T. McClanahan, R.A. Kastelein, D.J. Cua, IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J. Exp. Med. 201, 233–240 (2005)
E.P. Mensah-Brown, A. Shahin, M. Al-Shamisi, X. Wei, M.L. Lukic, IL-23 leads to diabetes induction after subdiabetogenic treatment with multiple low doses of streptozotocin. Eur. J. Immunol. 36, 216–223 (2006)
F. Abbasi, P. Amiri, F.A. Sayahpour, S. Pirmoradi, M. Abolhalaj, B. Larijani, J.T. Bazzaz, M.M. Amoli, TGF-β and IL-23 gene expression in unstimulated PBMCs of patients with diabetes. Endocrine 41, 430–434 (2012)
C. Li, L. Zhang, Y. Chen, X. Lin, T. Li, Protective role of adenovirus vector-mediated interleukin-10 gene therapy on endogenous islet β-cells in recent-onset type 1 diabetes in NOD mice. Exp. Therapeutic Med. 11, 1625–1632 (2016)
S. Robert, C. Gysemans, T. Takiishi, H. Korf, I. Spagnuolo, G. Sebastiani, K. Van Huynegem, L. Steidler, S. Caluwaerts, P. Demetter, C.H. Wasserfall, M.A. Atkinson, F. Dotta, P. Rottiers, T.L. Van Belle, C. Mathieu, Oral delivery of glutamic acid decarboxylase (GAD)-65 and IL10 by Lactococcus lactis reverses diabetes in recent-onset NOD mice. Diabetes 63, 2876–2887 (2014)
S. Kleffel, A. Vergani, S. Tezza, M. Ben Nasr, M.A. Niewczas, S. Wong, R. Bassi, F. D’Addio, T. Schatton, R. Abdi, M. Atkinson, M.H. Sayegh, L. Wen, C.H. Wasserfall, K.C. O’Connor, P. Fiorina, Interleukin-10+ regulatory B cells arise within antigen-experienced CD40+ B cells to maintain tolerance to islet autoantigens. Diabetes 64, 158–171 (2015)
Peng Y., Laouar Y., Li M.O., Green E.A., Flavell R.A. TGF-beta regulates in vivo expansion of Foxp3-expressing CD4+CD25+ regulatory T cells responsible for protection against diabetes. Proc. Natl. Acad. Sci. USA101,4572-7(2004).
Funding
This work was supported by the Hubei Province Natural Science Fund (2019CFB102) and the Health & Family Planning Commission Fund (WJ2015MB098).
Author contributions
XQC participated in the design of the study. MC and QHZ contributed to completion of the experiments. YHW, QQW and XM analyzed the data. MC and XQC wrote the main manuscript. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
All procedures in the study involving animals were performed in accordance with the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health (NIH Pub. No. 85–23, revised 1996) and approved by the Institutional Animal Care and Use Committee (IACUC) at the Centre for Animal Experiments, Wuhan University.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, M., Zhang, Q., Wei, Y. et al. Anti-CD20 therapy ameliorates β cell function and rebalances Th17/Treg cells in NOD mice. Endocrine 76, 44–52 (2022). https://doi.org/10.1007/s12020-021-02965-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-021-02965-x