Abstract
Purpose
Insulin-like factor 3 (INSL3) is an emerging testicular marker, yet larger studies elucidating the clinical role of INSL3 in patients with hypogonadism are lacking. The aim was to describe serum INSL3 concentrations analyzed by LC–MS/MS methodology in males with hypogonadotropic hypogonadism (HH) and Klinefelter syndrome (KS).
Methods
This was a combined study from two tertiary centers in Denmark and France analyzing INSL3 concentrations by LC–MS/MS. In total, 103 patients with HH and 82 patients with KS were grouped into treated (HH: n = 96; KS: n = 71) or untreated (HH: n = 7; KS: n = 11). Treatment modalities included testosterone and hCG. Serum concentrations and standard deviation (SD) scores of INSL3, total testosterone, and LH according to age and treatment were evaluated.
Results
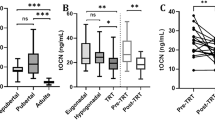
In both HH and KS, INSL3 concentrations were low. In HH, INSL3 was low regardless of treatment, except for some hCG-treated patients with normal concentrations. In untreated HH, testosterone was low, while normal to high in most testosterone- and hCG-treated patients. In untreated KS, INSL3 and testosterone concentrations were low to normal, while in testosterone-treated KS, serum INSL3 was low in most patients. INSL3 SD scores were significantly lower in untreated HH than in untreated KS (p = 0.01).
Conclusions
The dichotomy between lower INSL3 and higher testosterone concentrations, particularly observed in hCG-treated patients with HH, confirms that INSL3 is a different marker of Leydig cell function than testosterone. However, the clinical application of INSL3 in males with hypogonadism remains unclear.




Similar content being viewed by others
Data availability
Restrictions apply to some or all the availability of data generated or analyzed during this study to preserve patient confidentiality or because they were used under license. The corresponding author will on request detail the restrictions and any conditions under which access to some data may be provided.
References
K. Bay, A.S. Cohen, F.S. Jørgensen, C. Jørgensen, A.M. Lind, N.E. Skakkebæk, A.-M. Andersson, Insulin-like factor 3 levels in second-trimester amniotic fluid. J. Clin. Endocrinol. Metab. 93(10), 4048–4051 (2008). https://doi.org/10.1210/jc.2008-0358
K. Kawamura, J. Kumagai, S. Sudo, S.-Y. Chun, M. Pisarska, H. Morita, J. Toppari, P. Fu, J.D. Wade, R.A.D. Bathgate, A.J.W. Hsueh, Paracrine regulation of mammalian oocyte maturation and male germ cell survival. Proc. Natl Acad. Sci. USA. 101(19), 7323–7328 (2004). https://doi.org/10.1073/pnas.0307061101
K. Bay, H.E. Virtanen, S. Hartung, R. Ivell, K.M. Main, N.E. Skakkebaek, A.-M. Andersson, The Nordic Cryptorchidism Study Group, J. Toppari, Insulin-like factor 3 levels in cord blood and serum from children: effects of age, postnatal hypothalamic-pituitary-gonadal axis activation, and cryptorchidism. J. Clin. Endocrinol. Metab. 92(10), 4020–4027 (2007). https://doi.org/10.1210/jc.2007-0974
J. Albrethsen, M.L. Ljubicic, A. Juul, Longitudinal increases in serum insulin-like factor 3 and testosterone determined by LC-MS/MS in pubertal Danish boys. J. Clin. Endocrinol. Metab. 105(10), 3173–3178 (2020). https://doi.org/10.1210/clinem/dgaa496
R. Ivell, J.D. Wade, R. Anand-Ivell, INSL3 as a Biomarker of Leydig Cell Functionality. Biol. Reprod. 88(6), 147,1–8 (2013). https://doi.org/10.1095/biolreprod.113.108969
K. Bay, S. Hartung, R. Ivell, M. Schumacher, D. Jürgensen, N. Jorgensen, M. Holm, N.E. Skakkebaek, A.-M. Andersson, Insulin-like factor 3 serum levels in 135 normal men and 85 men with testicular disorders: relationship to the luteinizing hormone-testosterone axis. J. Clin. Endocrinol. Metab. 90(6), 3410–3418 (2005). https://doi.org/10.1210/jc.2004-2257
J. Toppari, Insulin-like factor 3 emerges from the shadow of testosterone as a Leydig cell biomarker. J. Clin. Endocrinol. Metab. 106(1), e370–e371 (2021). https://doi.org/10.1210/clinem/dgaa603
K. Bay, K.L. Matthiesson, R.I. McLachlan, A.-M. Andersson, The effects of gonadotropin suppression and selective replacement on insulin-like factor 3 secretion in normal adult men. J. Clin. Endocrinol. Metab. 91(3), 1108–1111 (2006). https://doi.org/10.1210/jc.2005-1865
M.Y. Roth, K. Lin, K. Bay, J.K. Amory, B.D. Anawalt, A.M. Matsumoto, B.T. Marck, W.J. Bremner, S.T. Page, Serum insulin-like factor 3 is highly correlated with intratesticular testosterone in normal men with acute, experimental gonadotropin deficiency stimulated with low-dose human chorionic gonadotropin: a randomized, controlled trial. Fertil. Steril. 99(1), 132–139 (2013). https://doi.org/10.1016/j.fertnstert.2012.09.009
C. Foresta, A. Bettella, C. Vinanzi, P. Dabrilli, M.C. Meriggiola, A. Garolla, A. Ferlin, Insulin-like factor 3: a novel circulating hormone of testis origin in humans. J. Clin. Endocrinol. Metab. 89(12), 5952–5958 (2004). https://doi.org/10.1210/jc.2004-0575
D. Santi, R. Ivell, R. Anand-Ivell, L. De Toni, F. Fanelli, M. Mezzullo, C. Pelusi, U. Pagotto, S. Belli, A.R.M. Granata, L. Roli, V. Rochira, T. Trenti, A. Ferlin, M. Simoni, Effects of acute hCG stimulation on serum INSL3 and 25-OH vitamin D in Klinefelter syndrome. Andrology 8(6), 1720–1727 (2020). https://doi.org/10.1111/andr.12851
K. Bay, A.-M. Andersson, Human testicular insulin-like factor 3: in relation to development, reproductive hormones and andrological disorders. Int. J. Androl. 34(2), 97–109 (2011). https://doi.org/10.1111/j.1365-2605.2010.01074.x
J. Albrethsen, H. Frederiksen, A.-M. Andersson, R. Anand-Ivell, L. Nordkap, A.K. Bang, N. Jørgensen, A. Juul, Development and validation of a mass spectrometry-based assay for quantification of insulin-like factor 3 in human serum. Clin. Chem. Lab. Med. 56(11), 1913–1920 (2018). https://doi.org/10.1515/cclm-2018-0171
J. Albrethsen, T.H. Johannsen, N. Jørgensen, H. Frederiksen, H.P. Sennels, H.L. Jørgensen, J. Fahrenkrug, J.H. Petersen, A. Linneberg, L. Nordkap, A.K. Bang, A.-M. Andersson, A. Juul, Evaluation of serum insulin-like factor 3 quantification by LC-MS/MS as a biomarker of Leydig cell function. J. Clin. Endocrinol. Metab. 105(6), 1868–1877 (2020). https://doi.org/10.1210/clinem/dgaa145
L. Aksglaede, N.E. Skakkebaek, A. Juul, Abnormal sex chromosome constitution and longitudinal growth: serum levels of insulin-like growth factor (IGF)-I, IGF binding protein-3, luteinizing hormone, and testosterone in 109 males with 47,XXY, 47,XYY, or sex-determining region of the Y chromosome (SRY)-positive 46,XX karyotypes. J. Clin. Endocrinol. Metab. 93(1), 169–176 (2008). https://doi.org/10.1210/jc.2007-1426
J. Young, C. Xu, G.E. Papadakis, J.S. Acierno, L. Maione, J. Hietamäki, T. Raivio, N. Pitteloud, Clinical management of congenital hypogonadotropic hypogonadism. Endocr. Rev. 40(2), 669–710 (2019). https://doi.org/10.1210/er.2018-00116
T. Raivio, J. Falardeau, A. Dwyer, R. Quinton, F. J. Hayes, V. A. Hughes, L. W. Cole, S. H. Pearce, H. Lee, P. Boepple, W. F. Crowley, Jr. N. Pitteloud, Reversal of idiopathic hypogonadotropic hypogonadism. N. Engl. J. Med. 357(9), 863–873 (2007). https://doi.org/10.1056/NEJMoa066494.
J. Tommiska, J. Känsäkoski, P. Christiansen, N. Jørgensen, J.G. Lawaetz, A. Juul, T. Raivio, Genetics of congenital hypogonadotropic hypogonadism in Denmark. Eur. J. Med. Genet. 57(7), 345–348 (2014). https://doi.org/10.1016/j.ejmg.2014.04.002
S. Trabado, L. Maione, H. Bry-Gauillard, H. Affres, S. Salenave, J. Sarfati, C. Bouvattier, B. Delemer, P. Chanson, Y. Le Bouc, S. Brailly-Tabard, J. Young, Insulin-like peptide 3 (INSL3) in men with congenital hypogonadotropic hypogonadism/Kallmann syndrome and effects of different modalities of hormonal treatment: a single-center study of 281 patients. J. Clin. Endocrinol. Metab. 99(2), E268–E275 (2014). https://doi.org/10.1210/jc.2013-2288
F. Giton, S. Trabado, L. Maione, J. Sarfati, Y. Le Bouc, S. Brailly-Tabard, J. Fiet, J. Young, Sex steroids, precursors, and metabolite deficiencies in men with isolated hypogonadotropic hypogonadism and panhypopituitarism: a GCMS-based comparative study. J. Clin. Endocrinol. Metab. 100(2), E292–E296 (2015). https://doi.org/10.1210/jc.2014-2658
M. Aadahl, M. Zacho, A. Linneberg, B.H. Thuesen, T. Jørgensen, Comparison of the Danish step test and the watt-max test for estimation of maximal oxygen uptake: the Health2008 study. Eur. J. Prev. Cardiol. 20(6), 1088–1094 (2013). https://doi.org/10.1177/2047487312462825
A. Damgaard-Olesen, T.H. Johannsen, S.A. Holmboe, T. Søeborg, J.H. Petersen, A.-M. Andersson, M. Aadahl, A. Linneberg, A. Juul, Reference ranges of 17-hydroxyprogesterone, DHEA, DHEAS, androstenedione, total and free testosterone determined by TurboFlow-LC-MS/MS and associations to health markers in 304 men. Clin. Chim. Acta 454, 82–88 (2016). https://doi.org/10.1016/j.cca.2015.12.042
T. Søeborg, H. Frederiksen, T.H. Johannsen, A.-M. Andersson, A. Juul, Isotope-dilution TurboFlow-LC-MS/MS method for simultaneous quantification of ten steroid metabolites in serum. Clin. Chim. Acta 468, 180–186 (2017). https://doi.org/10.1016/j.cca.2017.03.002
M.L. Ljubicic, K. Jespersen, L. Aksglaede, C.P. Hagen, J.H. Petersen, H.R. Andersen, A. Linneberg, K.M. Main, A.-M. Andersson, T.H. Johannsen, A. Juul, The LH/FSH ratio is not a sex-dimorphic marker after infancy: data from 6417 healthy individuals and 125 patients with Differences of Sex Development. Hum. Reprod. 35(10), 2323–2335 (2020). https://doi.org/10.1093/humrep/deaa182
J.H. Petersen, Two bivariate geometrically defined reference regions with applications to male reproductive hormones and human growth. Stat. Med. 22(16), 2603–2618 (2003). https://doi.org/10.1002/sim.1480
S. Trabado, S. Lamothe, L. Maione, C. Bouvattier, J. Sarfati, S. Brailly-Tabard, J. Young, Congenital hypogonadotropic hypogonadism and Kallmann syndrome as models for studying hormonal regulation of human testicular endocrine functions. Ann. Endocrinol. (Paris). 75(2), 79–87 (2014). https://doi.org/10.1016/j.ando.2014.04.011
A.M. Wikström, K. Bay, M. Hero, A.-M. Andersson, L. Dunkel, Serum insulin-like factor 3 levels during puberty in healthy boys and boys with Klinefelter syndrome. J. Clin. Endocrinol. Metab. 91(11), 4705–4708 (2006). https://doi.org/10.1210/jc.2006-0669
S. Overvad, K. Bay, A. Bojesen, C.H. Gravholt, Low INSL3 in Klinefelter syndrome is related to osteocalcin, testosterone treatment and body composition, as well as measures of the hypothalamic-pituitary-gonadal axis. Andrology 2(3), 421–427 (2014). https://doi.org/10.1111/j.2047-2927.2014.00204.x
A. Ferlin, M. Schipilliti, C. Foresta, Bone density and risk of osteoporosis in Klinefelter syndrome. Acta Paediatr. 100(6), 878–884 (2011). https://doi.org/10.1111/j.1651-2227.2010.02138.x
I.A. Olesen, A.-M. Andersson, L. Aksglaede, N.E. Skakkebaek, E. Rajpert-de Meyts, N. Joergensen, A. Juul, Clinical, genetic, biochemical, and testicular biopsy findings among 1,213 men evaluated for infertility. Fertil. Steril. 107(1), 74–82.e7 (2017). https://doi.org/10.1016/j.fertnstert.2016.09.015
A.-M. Andersson, J.H. Petersen, N. Jørgensen, T.K. Jensen, N.E. Skakkebæk, Serum inhibin B and follicle-stimulating hormone levels as tools in the evaluation of infertile men: significance of adequate reference values from proven fertile men. J. Clin. Endocrinol. Metab. 89(6), 2873–2879 (2004). https://doi.org/10.1210/jc.2003-032148
L. Aksglaede, A.-M. Andersson, N. Jørgensen, T.K. Jensen, E. Carlsen, R.I. McLachlan, N.E. Skakkebæk, J.H. Petersen, A. Juul, Primary testicular failure in Klinefelter’s syndrome: the use of bivariate luteinizing hormone-testosterone reference charts. Clin. Endocrinol. 66(2), 276–281 (2007). https://doi.org/10.1111/j.1365-2265.2006.02722.x
Funding
The study was funded by the Absalon Foundation (M.L.L.), Innovation Fund Denmark (14–2013–4), and The ReproUnion collaboration (J.A., A.J.U.), co-financed by The European Union, Interreg V ÖKS (NYPS-ID 20200407).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
Denmark: The study was approved by the ethics committees of the Capital Region of Denmark (H-KA20060011). All patient samples were taken as part of the clinical follow-up. Access to Danish patient data was approved by the Danish Patient Safety Authority (no. 3–3013–1376/1/) and the Danish Data Protection Agency (no. 2015–235, I-Suite no. 04204). In Denmark, the permission for INSL3 measurements was approved as part of a quality assurance project at Rigshospitalet, University Hospital of Copenhagen (approval no. 20012618). France: The study was approved by Assistance Publique-Hôpitaux de Paris and the institutional review boards at Bicêtre teaching hospital (Comité de Protection des Personnes Ile de France, Hôpital Bicêtre, Programme Hospitalier de Recherche Clinique [PHRC-2009, Hypoproteo].
Consent to participate
France: All participants gave written informed consent according to the French Bioethics Law and the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Johannsen, T.H., Ljubicic, M.L., Young, J. et al. Serum insulin-like factor 3 quantification by LC–MS/MS in male patients with hypogonadotropic hypogonadism and Klinefelter syndrome. Endocrine 71, 578–585 (2021). https://doi.org/10.1007/s12020-021-02609-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-021-02609-0