Abstract
Purpose
Despite a biological plausibility of a direct link between low vitamin D and androgen deficiency, the association remains inconclusive in epidemiological studies. Therefore, this systematic review and meta-analysis of case-control studies aim to assess whether and in what populations such an association can be demonstrated.
Methods
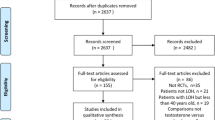
A systematic search was performed in PubMed, EMBASE, Cochrane Library, Web of science, Science Direct, and CINAHL. Standardized mean differences (SMDs) and 95% confidence intervals (CIs) in total testosterone (TT) levels between men with 25-hydroxyvitamin D (25(OH)D) <20 and ≥20 ng/mL were combined using random-effects models. Funnel plot and trim-and-fill analysis were used to assess publication bias. Heterogeneity source was explored by a sub-group analysis according to health-related characteristics of the study populations.
Results
Eighteen included studies collectively gave information on 9892 men with vitamin D deficiency and 10,675 controls. The pooled SMD revealed a slight, albeit just significant, positive association between 25(OH)D and TT (pooled SMD: −0.23, 95% CI: −0.45 to −0.01; P = 0.04) with a large between-study heterogeneity (I2 = 98%, Pfor heterogeneity < 0.00001). At the sub-group analysis, a significant positive association, along with noticeable decrease in heterogeneity, could only be demonstrated in studies of patients with frailty states (pooled SMD: −0.19; 95% CI: −0.27, −0.10, P < 0.0001; I2 = 51%, Pfor heterogeneity = 0.06). A sensitivity analysis revealed a high stability of the result and the trim-and-fill adjustment for publication bias did not affect pooled estimate.
Conclusions
Both hypovitaminosis D and androgen deficiency should be regarded as markers of a poor health status, sharing common underlying aetiologies and risk factors.





Similar content being viewed by others
References
P. Lips, Vitamin D deficiency and secondary hyperparathyroidism in the elderly: consequences for bone loss and fractures and therapeutic implications. Endocr. Rev. 22, 477–501 (2001). https://doi.org/10.1210/edrv.22.4.0437
D.M. Lee, A. Tajar, T.W. O’Neill, D.B. O’Connor, G. Bartfai, S. Boonen, R. Bouillon, F.F. Casanueva, J.D. Finn, G. Forti, A. Giwercman, T.S. Han, I.T. Huhtaniemi, K. Kula, M. Ej Lean, M. Punab, A.J. Silman, D. Vanderschueren, F. Cw Wu, N. Pendleton, Lower vitamin D levels are associated with depression among community-dwelling European men. J. Psychopharmacol. 25, 1320–1328 (2011). https://doi.org/10.1177/0269881110379287
A. Barbonetti, F. Cavallo, S. D’Andrea, M. Muselli, G. Felzani, S. Francavilla, F. Francavilla, Lower Vitamin D levels are associated with depression in people with chronic spinal cord injury. Arch. Phys. Med. Rehabil. 98, 940–946 (2017). https://doi.org/10.1016/j.apmr.2016.11.006
S. Pilz, A. Tomaschitz, E. Ritz, T.R. Pieber, Vitamin D status and arterial hypertension: a systematic review. Nat. Rev. Cardiol. 6, 621–630 (2009). https://doi.org/10.1038/nrcardio.2009.135
A.G. Pittas, J. Lau, F.B. Hu, B. Dawson-Hughes, The role of vitamin D and calcium in type 2 diabetes. A systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 92, 2017–2029 (2007). https://doi.org/10.1210/jc.2007-0298
N.G. Forouhi, J. Luan, A. Cooper, B.J. Boucher, N.J. Wareham, Baseline serum 25-hydroxy vitamin D is predictive of future glycemic status and insulin resistance: the Medical Research Council Ely Prospective Study 1990–2000. Diabetes 57, 2619–2625 (2008). https://doi.org/10.2337/db08-0593
T.J. Wang, M.J. Pencina, S.L. Booth, P.F. Jacques, E. Ingelsson, K. Lanier, E.J. Benjamin, R.B. D’Agostino, M. Wolf, R.S. Vasan, Vitamin D deficiency and risk of cardiovascular disease. Circulation 117, 503–511 (2008). https://doi.org/10.1161/CIRCULATIONAHA.107.706127
R. Illescas-Montes, L. Melguizo-Rodríguez, C. Ruiz, V.J. Costela-Ruiz, Vitamin D and autoimmune diseases. Life Sci. 233, 116744 (2019). https://doi.org/10.1016/j.lfs.2019.116744
I.S. Wicherts, N.M. van Schoor, A.J. Boeke, M. Visser, D.J. Deeg, J. Smit, D.L. Knol, P. Lips, Vitamin D status predicts physical performance and its decline in older persons. J. Clin. Endocrinol. Metab. 92, 2058–2065 (2007). https://doi.org/10.1210/jc.2006-1525
A. Barbonetti, A. Sperandio, A. Micillo, S. D’Andrea, F. Pacca, G. Felzani, S. Francavilla, F. Francavilla, Independent association of Vitamin D with physical function in people with chronic spinal cord injury. Arch. Phys. Med. Rehabil. 97, 726–732 (2016). https://doi.org/10.1016/j.apmr.2016.01.002
A. Barbonetti, S. D’Andrea, A. Martorella, G. Felzani, S. Francavilla, F. Francavilla, Low vitamin D levels are independent predictors of 1-year worsening in physical function in people with chronic spinal cord injury: a longitudinal study. Spinal Cord 5, 494–501 (2018). https://doi.org/10.1038/s41393-017-0058-7
T. Haykal, V. Samji, Y. Zayed, I. Gakhal, H. Dhillo, B. Kheir, J. Kerbag, V. Veerapanen, M. Obeid, R. Danish, G. Bachuwa, The role of vitamin D supplementation for primary prevention of cancer: meta-analysis of randomized controlled trials. J. Community Hosp. Intern. Med. Perspect. 9, 480–488 (2019). https://doi.org/10.1080/20009666.2019.1701839
Y. Wang, J. Zhu, H.F. DeLuca, Where is the vitamin D receptor? Arch. Biochem. Biophys. 523, 123–133 (2012). https://doi.org/10.1016/j.abb.2012.04.001
M. Blomberg Jensen, J.E. Nielsen, A. Jørgensen, E. Rajpert-De Meyts, D.M. Kristensen, N. Jørgensen, N.E. Skakkebaek, A. Juul, H. Leffers, Vitamin D receptor and vitamin D metabolizing enzymes are expressed in the human male reproductive tract. Hum. Reprod. 25, 1303–1311 (2010). https://doi.org/10.1093/humrep/deq024
J.B. Cheng, M.A. Levine, N.H. Bell, D.J. Mangelsdorf, D.W. Russell, Genetic evidence that the human CYP2R1 enzyme is a key vitamin D 25-hydroxylase. Proc. Natl Acad. Sci. USA 101, 7711–7715 (2004). https://doi.org/10.1073/pnas.0402490101
C. Foresta, G. Strapazzon, L. De Toni, L. Perilli, A. Di Mambro, B. Muciaccia, L. Sartori, R. Selice, Bone mineral density and testicular failure: evidence for a role of vitamin D 25-hydroxylase in human testis. J. Clin. Endocrinol. Metab. 96, E646–E652 (2011). https://doi.org/10.1210/jc.2010-1628
C. Foresta, R. Selice, A. Di Mambro, G. Strapazzon, Testiculopathy and vitamin D insufficiency. Lancet 376, 1301 (2010). https://doi.org/10.1016/S0140-6736(10)61916-2
R. Bouillon, G. Carmeliet, L. Verlinden, E. van Etten, A. Verstuyf, H.F. Luderer, L. Lieben, C. Mathieu, M. Demay, Vitamin D and human health: lessons from vitamin D receptor null mice. Endocr. Rev. 29, 726–776 (2008). https://doi.org/10.1210/er.2008-0004
D. Hofer, J. Münzker, V. Schwetz, M. Ulbing, K. Hutz, P. Stiegler, R. Zigeuner, T.R. Pieber, H. Müller, B. Obermayer-Pietsch, Testicular synthesis and vitamin D action. J. Clin. Endocrinol. Metab. 99, 3766–3767 (2014). https://doi.org/10.1210/jc.20141690
E. Wehr, S. Pilz, B.O. Boehm, W. März, B. Obermayer-Pietsch, Association of vitamin D status with serum androgen levels in men. Clin. Endocrinol. 73, 243–248 (2010). https://doi.org/10.1111/j.1365-2265.2009.03777.x
D.M. Lee, A. Tajar, S.R. Pye, S. Boonen, D. Vanderschueren, R. Bouillon, T.W. O’Neill, G. Bartfai, F.F. Casanueva, J.D. Finn, G. Forti, A. Giwercman, T.S. Han, I.T. Huhtaniemi, K. Kula, M.E.J. Lean, N. Pendleton, M. Punab, F.C.W. Wu, Association of hypogonadism with vitamin D status: the European Male Ageing Study. Eur. J. Endocrinol. 166, 77–85 (2012). https://doi.org/10.1530/EJE-11-0743
K. Nimptsch, E.A. Platz, W.C. Willett, E. Giovannucci, Association between plasma 25-OH vitamin D and testosterone levels in men. Clin. Endocrinol. 77, 106–112 (2012). https://doi.org/10.1111/j.1365-2265.2012.04332.x
K.Y. Chin, S. Ima-Nirwana, W.Z. Wan Ngah, Vitamin D is significantly associated with total testosterone and sex hormone-binding globulin in Malaysian men. Aging Male 18, 175–179 (2015). https://doi.org/10.3109/13685538.2015.1034686
A. Ferlin, R. Selice, A. Di Mambro, M. Ghezzi, A. Di Nisio, N. Caretta, C. Foresta, Role of vitamin D levels and vitamin D supplementation on bone mineral density in Klinefelter syndrome. Osteoporos. Int. 26, 2193–2202 (2015). https://doi.org/10.1007/s00198015-3136-8
L. Laczmanski, F. Lwow, M. Mossakowska, M. Puzianowska-Kuznicka, M. Szwed, K. Kolackov, B. Krzyzanowska-Swiniarska, E. Bar-Andziak, J. Chudek, N. Sloka, Association between vitamin D concentration and levels of sex hormones in an elderly Polish population with different genotypes of VDR polymorphisms (rs10735810, rs1544410, rs7975232, rs731236). Gene 559, 73–76 (2015). https://doi.org/10.1016/j.gene.2015.01.022
Y.J. Tak, J.G. Lee, Y.J. Kim, N.C. Park, S.S. Kim, S. Lee, B.M. Cho, E.H. Kong, D.W. Jung, Y.H. Yi, Serum 25-hydroxyvitamin D levels and testosterone deficiency in middle-aged Korean men: a cross-sectional study. Asian J. Androl. 17, 324–328 (2015). https://doi.org/10.4103/1008-682X.142137
N. Wang, B. Han, Q. Li, Y. Chen, Y. Chen, F. Xia, D. Lin, M.D. Jensen, Y. Lu, Vitamin D is associated with testosterone and hypogonadism in Chinese men: Results from a cross-sectional SPECT-China study. Reprod. Biol. Endocrinol. 13, 74 (2015). https://doi.org/10.1186/s12958-015-0068-2
G.M. Anic, D. Albanes, S. Rohrmann, N. Kanarek, W.G. Nelson, G. Bradwin, N. Rifai, K.A. McGlynn, E.A. Platz, A.M. Mondul, Association between serum 25-hydroxyvitamin D and serum sex steroid hormones among men in NHANES. Clin. Endocrinol. 85, 258–266 (2016). https://doi.org/10.1111/cen.13062
A. Barbonetti, M.R. Vassallo, G. Felzani, S. Francavilla, F. Francavilla, Association between 25(OH)-vitamin D and testosterone levels: Evidence from men with chronic spinal cord injury. J. Spinal Cord Med. 39, 246–252 (2016). https://doi.org/10.1179/2045772315Y.0000000050
R. Rafiq, N.M. van Schoor, E. Sohl, M.C. Zillikens, M.M. Oosterwerff, L. Schaap, P. Lips, R.T. de Jongh, Associations of vitamin D status and vitamin D-related polymorphisms with sex hormones in older men. J. Steroid Biochem. Mol. Biol. 164, 11–17 (2016). https://doi.org/10.1016/j.jsbmb.2015.11.013
S.G. Park, J.K. Yeo, D.Y. Cho, M.G. Park, Impact of metabolic status on the association of serum vitamin D with hypogonadism and lower urinary tract symptoms/benign prostatic hyperplasia. Aging Male 21, 55–59 (2018). https://doi.org/10.1080/13685538.2017.1311857
G. Tirabassi, M. Sudano, G. Salvio, M. Cutini, G. Muscogiuri, G. Corona, G. Balercia, Vitamin D and male sexual function: a transversal and longitudinal study. Int. J. Endocrinol. 3720813 (2018). https://doi.org/10.1155/2018/3720813
L. Ceglia, G.R. Chiu, S.S. Harris, A.B. Araujo, Serum 25-hydroxyvitamin D concentration and physical function in adult men. Clin. Endocrinol. 74, 370–376 (2011). https://doi.org/10.1111/j.1365-2265.2010.03926.x
C.H. Ramlau-Hansen, U.K. Moeller, J.P. Bonde, J. Olsen, A.M. Thulstrup, Are serum levels of vitamin D associated with semen quality? Results from a cross-sectional study in young healthy men. Fertil. Steril. 95, 1000–1004 (2011). https://doi.org/10.1016/j.fertnstert.2010.11.002
A.O. Hammoud, A.W. Meikle, C.M. Peterson, J. Stanford, M. Gibson, D.T. Carrell, Association of 25-hydroxy-vitamin D levels with semen and hormonal parameters. Asian J. Androl. 14, 855–859 (2012). https://doi.org/10.1038/aja.2012.77
R. Jorde, G. Grimnes, M.S. Hutchinson, M. Kjærgaard, E. Kamycheva, J. Svartberg, Supplementation with vitamin D does not increase serum testosterone levels in healthy males. Horm. Metab. Res. 45, 675–681 (2013). https://doi.org/10.1055/s-0033-1345139
E. Lerchbaum, S. Pilz, C. Trummer, T. Rabe, M. Schenk, A.C. Heijboer, B. Obermayer-Pietsch, Serum vitamin D levels and hypogonadism in men. Andrology 2, 748–754 (2014). https://doi.org/10.1111/j.2047-2927.2014.00247.x
M. Blomberg Jensen, J. Gerner Lawaetz, A.M. Andersson, J.H. Petersen, L. Nordkap, A.K. Bang, P. Ekbom, U.N. Joensen, L. Prætorius, P. Lundstrøm, V.H. Boujida, B. Lanske, A. Juul, N. Jørgensen, Vitamin D deficiency and low ionized calcium are linked with semen quality and sex steroid levels in infertile men. Hum. Reprod. 31, 1875–1885 (2016). https://doi.org/10.1093/humrep/dew152
S. Abbasihormozi, A. Kouhkan, A.R. Alizadeh, A.H. Shahverdi, M.H. Nasr-Esfahani, M.A. Sadighi Gilani, R. Salman Yazdi, A. Matinibehzad, Z. Zolfaghari, Association of vitamin D status with semen quality and reproductive hormones in Iranian subfertile men. Andrology 5, 113–118 (2017). https://doi.org/10.1111/andr.12280
M.Y. Sim, S.H. Kim, K.M. Kim, Seasonal variations and correlations between Vitamin D and total testosterone levels. Korean J. Fam. Med. 38, 270–275 (2017). https://doi.org/10.4082/kjfm.2017.38.5.270
D. Zhao, P. Ouyang, I.H. de Boer, P.L. Lutsey, Y.M. Farag, E. Guallar, D.S. Siscovick, W.S. Post, R.R. Kalyani, K.L. Billups, E.D. Michos, Serum vitamin D and sex hormones levels in men and women: The Multi-Ethnic Study of Atherosclerosis (MESA). Maturitas 96, 95–102 (2017). https://doi.org/10.1016/j.maturitas.2016.11.017
A. Rudnicka, E. Adoamnei, J.A. Noguera-Velasco, J. Vioque, F. Cañizares-Hernández, J. Mendiola, N. Jørgensen, J.E. Chavarro, S.H. Swan, A.M. Torres-Cantero, Vitamin D status is not associated with reproductive parameters in young Spanish men. Andrology 8, 323–331 (2020). https://doi.org/10.1111/andr.12690
L. Shamseer, D. Moher, M. Clarke, D. Ghersi, A. Liberati, M. Petticrew, P. Shekelle, L.A. Stewart, Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: elaboration and explanation. BMJ 349, g7647 (2015). https://doi.org/10.1136/bmj.g7647
M.F. Holick, N.C. Binkley, H.A. Bischoff-Ferrari, C.M. Gordon, D.A. Hanley, R.P. Heaney, M.H. Murad, C.M. Weaver, Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 96, 1911–1930 (2011). https://doi.org/10.1210/jc.2011-0385
M. Bland, Estimating mean and standard deviation from the sample size, three quartiles, minimum, and maximum estimating mean and standard deviation from the sample size, three quartiles, minimum, and maximum. Int. J. Stat. Med. Res. 4, 57–64 (2015). https://doi.org/10.6000/1929-6029.2015.04.01.6
J.J. Deeks, J. Dinnes, R. D’Amico, A.J. Sowden, C. Sakarovitch, F. Song, M. Petticrew, D.G. Altman, Evaluating non randomized intervention studies. Health Technol. Assess 7, 1–173 (2003). https://doi.org/10.3310/hta7270
J.P. Higgins, S.G. Thompson, J.J. Deeks, D.G. Altman, Measuring inconsistency in meta-analyses. BMJ 32, 557–560 (2003). https://doi.org/10.1136/bmj.327.7414.557
A. Barbonetti, A. Martorella, E. Minaldi, S. D’Andrea, D. Bardhi, C. Castellini, F. Francavilla, S. Francavilla, Testicular cancer in infertile men with and without testicular microlithiasis: a systematic review and meta-analysis of case-control studies. Front. Endocrinol. 10, 164 (2019). https://doi.org/10.3389/fendo.2019.00164
S. D’Andrea, F. Pallotti, G. Senofonte, C. Castellini, D. Paoli, F. Lombardo, A. Lenzi, S. Francavilla, F. Francavilla, A. Barbonetti, Polymorphic cytosine-adenine-guanine repeat length of androgen receptor gene and gender incongruence in trans women: a systematic review and meta-analysis of case-control studies. J. Sex. Med. 17, 543–550 (2020). https://doi.org/10.1016/j.jsxm.2019.12.010
E. Minaldi, S. D’Andrea, C. Castellini, A. Martorella, F. Francavilla, S. Francavilla, A. Barbonetti, Thyroid autoimmunity and risk of post-partum depression: a systematic review and meta-analysis of longitudinal studies. J. Endocrinol. Invest. 43, 271–277 (2020). https://doi.org/10.1007/s40618-019-01120-8
J.A. Sterne, M. Egger, Funnel plots for detecting bias in meta-analysis: guidelines on choice of axis. J. Clin. Epidemiol. 54, 1046–1055 (2001). https://doi.org/10.1016/S0895-4356(01)00377-8
S. Duval, R. Tweedie, Trim and fill: a simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 56, 455–463 (2000). https://doi.org/10.1111/j.0006-341X.2000.00455.x
J. MacLaughlin, M.F. Holick, Aging decreases the capacity of human skin to produce vitamin D3. J. Endocrinol. Invest. 76, 1536–1538 (1985). https://doi.org/10.1172/JCI112134
J.M. Zmuda, J.A. Cauley, A. Kriska, N.W. Glynn, J.P. Gutai, L.H. Kuller, Longitudinal relation between endogenous testosterone and cardiovascular disease risk factors in middle-aged men. A 13-year follow-up of former multiple risk factor intervention trial participants. Am. J. Epidemiol. 146, 609–617 (1997). https://doi.org/10.1093/oxfordjournals.aje.a009326
S.M. Harman, E.J. Metter, J.D. Tobin, J. Pearson, M.R. Blackman, Longitudinal effects of aging on serum total and free testosterone levels in healthy men. Baltimore Longitudinal Study of Aging. J. Clin. Endocrinol. Metab. 86, 724–731 (2001). https://doi.org/10.1210/jcem.86.2.7219
H.A. Feldman, C. Longcope, C.A. Derby, C.B. Johannes, A.B. Araujo, A.D. Coviello, W.J. Bremner, J.B. McKinlay, Age trends in the level of serum testosterone and other hormones in middle-aged men: longitudinal results from the Massachusetts male aging study. J. Clin. Endocrinol. Metab. 87, 589–598 (2002). https://doi.org/10.1210/jcem.87.2.8201
J.M. Kaufman, A. Vermeulen, The decline of androgen levels in elderly men and its clinical and therapeutic implications. Endocr. Rev. 26, 833–876 (2005). https://doi.org/10.1210/er.20040013
E.M. Camacho, I.T. Huhtaniemi, T.W. O’Neill, J.D. Finn, S.R. Pye, D.M. Lee, A. Tajar, G. Bartfai, S. Boonen, F.F. Casanueva, G. Forti, A. Giwercman, T.S. Han, K. Kula, B. Keevil, M.E. Lean, N. Pendleton, M. Punab, D. Vanderschueren, F.C.W. Wu, Age-associated changes in hypothalamic-pituitary-testicular function in middle-aged and older men are modified by weight change and lifestyle factors: longitudinal results from the European Male Ageing Study. Eur. J. Endocrinol. 168, 445–455 (2013). https://doi.org/10.1530/EJE-12-0890
A. Karagiannis, F. Harsoulis, Gonadal dysfunction in systemic diseases. Eur. J. Endocrinol. 152, 501–513 (2005). https://doi.org/10.1530/eje.1.01886
T. van der Poll, J.A. Romijn, E. Endert, H.P. Sauerwein, Effects of tumor necrosis factor on the hypothalamic-pituitary-testicular axis in healthy men. Metabolism 42, 303–307 (1993). https://doi.org/10.1016/0026-0495(93)90078-3
C.Y. Hong, J.H. Park, R.S. Ahn, S.Y. Im, H.S. Choi, J. Soh, S.H. Mellon, K. Lee, Molecular mechanism of suppression of testicular steroidogenesis by proinflammatory cytokine tumor necrosis factor alpha. Mol. Cell. Biol. 24, 2593–25604 (2004). https://doi.org/10.1128/MCB.24.7.2593-2604.2004
J. Veldhuis, R. Yang, F. Roelfsema, P. Takahashi, Proinflammatory cytokine infusion attenuates LH’s feedforward on testosterone secretion: modulation by age. J. Clin. Endocrinol. Metab. 101, 539–549 (2016). https://doi.org/10.1210/jc.2015-3611
R. Haring, S.E. Baumeister, H. Völzke, M. Dörr, T. Kocher, M. Nauck, H. Wallaschofski, Prospective inverse associations of sex hormone concentrations in men with biomarkers of inflammation and oxidative stress. J. Androl. 33, 944–950 (2012). https://doi.org/10.2164/jandrol.111.015065
T. Ahern, A. Swiecicka, R.J. Eendebak, E.L. Carter, J.D. Finn, S.R. Pye, T.W. O’Neill, L. Antonio, B. Keevil, G. Bartfai, F.F. Casanueva, G. Forti, A. Giwercman, T.S. Han, K. Kula, M.E.J. Lean, N. Pendleton, M. Punab, G. Rastrelli, M.K. Rutter, D. Vanderschueren, I.T. Huhtaniemi, F.C.W. Wu, Natural history, risk factors and clinical features of primary hypogonadism in ageing men: longitudinal data from the European Male Ageing Study. Clin. Endocrinol. 85, 891–901 (2016). https://doi.org/10.1111/cen.13152
M. Pereira-Santos, P.R. Costa, A.M. Assis, C.A. Santos, D.B. Santos, Obesity and vitamin D deficiency: a systematic review and meta-analysis. Obes. Rev. 16, 341–349 (2015). https://doi.org/10.1111/obr.12239
A.G. Need, H.A. Morris, M. Horowitz, C. Nordin, Effects of skin thickness, age, body fat, and sunlight on serum 25-hydroxyvitamin D. Am. J. Clin. Nutr. 58, 882–885 (1993). https://doi.org/10.1093/ajcn/58.6.882
J. Wortsman, L.Y. Matsuoka, T.C. Chen, Z. Lu, M.F. Holick, Decreased bioavailability of vitamin D in obesity. Am. J. Clin. Nutr. 72, 690–693 (2000). https://doi.org/10.1093/ajcn/72.3.690
AndreaDi Nisio, LucaDe Toni, Iva Sabovic, MariaSanta Rocca, VincenzoDe Filippis, Giuseppe Opocher, Bruno Azzena, Roberto Vettor, Mario Plebani, Carlo Foresta, Impaired release of Vitamin D in dysfunctional adipose tissue: new cues on Vitamin D supplementation in obesity. J. Clin. Endocrinol. Metab. 102, 2564–2574 (2017). https://doi.org/10.1210/jc.2016-3591
J.E. Compston, S. Vedi, J.E. Ledger, A. Webb, J.C. Gazet, T.R. Pilkington, Vitamin D status and bone histomorphometry in gross obesity. Am. J. Clin. Nutr. 34, 2359–2363 (1981). https://doi.org/10.1093/ajcn/34.11.2359
E. Hyppönen, C. Power, Hypovitaminosis D in British adults at age 45 y: nationwide cohort study of dietary and lifestyle pre-dictors. Am. J. Clin. Nutr. 85, 860–868 (2007). https://doi.org/10.1093/ajcn/85.3.860
S. Migliaccio, A. Di Nisio, C. Mele, L. Scappaticcio, S. Savastano, A. Colao, Obesity and hypovitaminosis D: Causality or casualty? Int. J. Obes. Suppl. 9, 20–31 (2019). https://doi.org/10.1038/s41367-019-0010-8
G. Corona, G. Rastrelli, L. Vignozzi, E. Mannucci, M. Maggi, Testosterone, cardiovascular disease and the metabolic syndrome. Best Pract. Res. Clin. Endocrinol. Metab. 25, 337–353 (2011). https://doi.org/10.1016/j.beem.2010.07.002
A. Di Nisio, I. Sabovic, L. De Toni, M. Santa Rocca, S. Dall’Acqua, B. Azzena, M. De Rocco Ponce, C. Foresta, Testosterone is sequestered in dysfunctional adipose tissue, modifying androgen-responsive genes. Int. J. Obes. 44, 1617–1625 (2020). https://doi.org/10.1038/s41366-020-0568-9
G. Schneider, M.A. Kirschner, R. Berkowitz, N.H. Ertel, Increased estrogen production in obese men. J. Clin. Endocrinol. Metab. 48, 633–638 (1979). https://doi.org/10.1210/jcem-48-4-633
C.J. Bagatell, K.D. Dahl, W.J. Bremner, The direct pituitary effect of testosterone to inhibit gonadotropin secretion in men is partially mediated by aromatization to estradiol. J. Androl. 15, 15–21 (1994). https://doi.org/10.1002/j.1939-4640.1994.tb01674.x
V.A. Giagulli, J.M. Kaufman, A. Vermeulen, Pathogenesis of the decreased androgen levels in obese men. J. Clin. Endocrinol. Metab. 79, 997–1000 (1994). https://doi.org/10.1210/jcem.79.4.7962311
S. Bhasin, W.E. Taylor, R. Singh, J. Artaza, I. Sinha-Hikim, R. Jasuja, H. Choi, N.F. Gonzalez-Cadavid, The mechanisms of androgen effects on body composition: mesenchymal pluripotent cell as the target of androgen action. J. Gerontol. A Biol. Sci. Med. Sci. 58, M1103–M1110 (2003). https://doi.org/10.1093/gerona/58.12.M1103
D.E. Laaksonen, L. Niskanen, K. Punnonen, K. Nyyssönen, T.P. Tuomainen, V.P. Valkonen, R. Salonen, J.T. Salonen, Testosterone and sex hormone-binding globulin predict the metabolic syndrome and diabetes in middle-aged men. Diabetes Care 27, 1036–1041 (2004). https://doi.org/10.2337/diacare.27.5.1036
L. Maïmoun, C. Fattal, C. Sultan, Bone remodeling and calcium homeostasis in patients with spinal cord injury: a review. Metabolism 60, 1655–1663 (2011). https://doi.org/10.1016/j.metabol.2011.04.005
W.A. Bauman, M.F. Fountaine, A.M. Spungen, Age-related prevalence of low testosterone in men with spinal cord injury. J. Spinal Cord Med. 37, 32–39 (2014). https://doi.org/10.1179/2045772313Y.0000000122
A. Barbonetti, M.R. Vassallo, F. Pacca, F. Cavallo, M. Costanzo, G. Felzani, S. Francavilla, F. Francavilla, Correlates of low testosterone in men with chronic spinal cord injury. Andrology 2, 721–728 (2014). https://doi.org/10.1111/j.2047-2927.2014.00235.x
A. Barbonetti, M.R.C. Vassallo, M. Cotugno, G. Felzani, S. Francavilla, F. Francavilla, Low testosterone and non-alcoholic fatty liver disease: Evidence for their independent association in men with chronic spinal cord injury. J. Spinal Cord Med. 39, 443–449 (2016). https://doi.org/10.1179/2045772314Y.0000000288
S.D. Sullivan, M.S. Nash, E. Tefera, E. Tinsley, M.R. Blackman, S. Groah, Prevalence and etiology of hypogonadism in young men with chronic spinal cord injury: a cross-sectional analysis from two university-based rehabilitation centers. PM R 9, 751–760 (2017). https://doi.org/10.1016/j.pmrj.2016.11.005
W.A. Bauman, Y.G. Zhong, E. Schwartz, Vitamin D deficiency in veterans with chronic spinal cord injury. Metabolism 44, 1612–1616 (1995). https://doi.org/10.1016/00260495(95)90083-7
A. Barbonetti, S. D’Andrea, J. Samavat, A. Martorella, G. Felzani, S. Francavilla, M. Luconi, F. Francavilla, Can the positive association of osteocalcin with testosterone be unmasked when the preeminent hypothalamic-pituitary regulation of testosterone production is impaired? The model of spinal cord injury. J. Endocrinol. Invest. 42, 167–173 (2019). https://doi.org/10.1007/s40618-018-0897-x
S. Pilz, S. Frisch, H. Koertke, J. Kuhn, J. Dreier, B. Obermayer-Pietsch, E. Wehr, A. Zittermann, Effect of vitamin D supplementation on testosterone levels in men. Horm. Metab. Res. 43, 223–225 (2011). https://doi.org/10.1055/s-0030-1269854
O. Canguven, R.A. Talib, W. El Ansari, D.J. Yassin, A. Al Naimi, Vitamin D treatment improves levels of sexual hormones, metabolic parameters and erectile function in middle-aged vitamin D deficient men. Aging Male 20, 9–16 (2017). https://doi.org/10.1080/13685538.2016.1271783
E. Hosseini Marnani, M. Mollahosseini, A. Gheflati, A. Ghadiri-Anari, A. Nadjarzadeh, The effect of vitamin D supplementation on the androgenic profile in men: a systematic review and meta-analysis of clinical trials. Andrologia 51, e13343 (2019). https://doi.org/10.1111/and.13343
M.J. Bolland, A.B. Grey, R.W. Ames, B.H. Mason, A.M. Horne, G.D. Gamble, I.R. Reid, The effects of seasonal variation of 25-hydroxyvitamin D and fat mass on a diagnosis of vitamin D sufficiency. Am. J. Clin. Nutr. 86, 959–964 (2007). https://doi.org/10.1093/ajcn/86.4.959
R. Rosecrans, J.C. Dohnal, Seasonal vitamin D changes and the impact on health risk assessment. Clin. Biochem. 47, 670–672 (2014). https://doi.org/10.1016/j.clinbiochem.2014.02.004
Funding
This work was supported by the Ministero dell’Istruzione, Università e Ricerca (MIUR), PRIN 2017.
Author contributions
S.D.A. systematic literature search, evaluation of selected studies for eligibility, quality assessment of the studies; A.M. systematic literature search, evaluation of selected studies for eligibility, quality assessment of the studies; F.C. evaluation of selected studies for eligibility, data extraction; C.C. data extraction, data analysis; E.M. systematic literature search; M.T. data extraction; A.P. data extraction; S.F. quality assessment of the studies, revising the article for intellectual content; F.F. revising the article for intellectual content; A.B. conception and design, data analysis, drafting the article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
D’Andrea, S., Martorella, A., Coccia, F. et al. Relationship of Vitamin D status with testosterone levels: a systematic review and meta-analysis. Endocrine 72, 49–61 (2021). https://doi.org/10.1007/s12020-020-02482-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-020-02482-3