Abstract
Purpose
Non-alcoholic fatty liver disease is highly associated with morbidity and mortality in population. Although studies have already demonstrated that the immune response plays a pivotal role in the development of non-alcoholic fatty liver disease, the comprehensive regulation is unclear. Therefore, present study was carried out to investigate the non-alcoholic fatty liver disease development under neutrophil depletion.
Methods
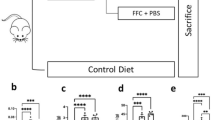
To achieve the aim of the study, C57BL/6 J mice were fed with high fat diet for 6 weeks before treated with neutrophil deplete antibody 1A8 or isotype control (200 μg/ mouse every week) for another 4 weeks.
Results
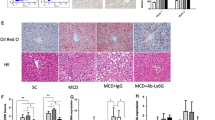
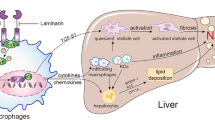
Treated with 1A8 antibody, obese mice exhibited better whole body metabolic parameters, including reduction of body weight gain and fasting blood glucose levels. Neutrophil depletion also effectively reduced hepatic structural disorders, dysfunction and lipid accumulation. Lipid β-oxidative markers, phosphorylated-AMP-activated protein kinase α and phosphorylated-acetyl-CoA carboxylase levels were increased in 1A8 antibody-treated obese mouse group. The mitochondrial number and function were also reversed after 1A8 antibody treatment, including increased mitochondrial number, reduced lipid oxidative damage and enhanced mitochondrial activity. Furthermore, the expression of inflammatory cytokines, tumor necrosis factor-α, interleukin-6, and monocyte chemoattractant protein-1 were obviously reduced after neutrophil depletion, accompanied with decreased F4/80 mRNA level and macrophage percentage in liver. The decreased NF-κB signaling activity was also involved in the beneficial effect of neutrophil depletion.
Conclusion
Taken together, neutrophil depletion could attenuate metabolic syndromes and hepatic dysfunction.





Similar content being viewed by others
References
P.L. Jansen, Non-alcoholic steatohepatitis. Eur. J. Gastroenterol. Hepatol. 16, 1079–1085 (2004)
C.A. Matteoni, Z.M. Younossi, T. Gramlich, N. Boparai, Y.C. Liu, A.J. McCullough, Nonalcoholic fatty liver disease: a spectrum of clinical and pathological severity. Gastroenterology 116, 1413–1419 (1999)
C.P. Day, O.F. James, Steatohepatitis: a tale of two “hits” ? Gastroenterology 114, 842–845 (1998)
A. Leroux, G. Ferrere, V. Godie, F. Cailleux, M.L. Renoud, F. Gaudin, S. Naveau, S. Prevot, S. Makhzami, G. Perlemuter, A.M. Cassard-Doulcier, Toxic lipids stored by Kupffer cells correlates with their pro-inflammatory phenotype at an early stage of steatohepatitis. J. Hepatol. 57, 141–149 (2012)
T. Miyake, S.M. Akbar, O. Yoshida, S. Chen, Y. Hiasa, B. Matsuura, M. Abe, M. Onji, Impaired dendritic cell functions disrupt antigen-specific adaptive immune responses in mice with nonalcoholic fatty liver disease. J. Gastroenterol. 45, 859–867 (2010)
R. Xu, H. Huang, Z. Zhang, F.S. Wang, The role of neutrophils in the development of liver diseases. Cell. Mol. Immunol. 11, 224–231 (2014b)
M. Donini, S. Fontana, G. Savoldi, W. Vermi, L. Tassone, F. Gentili, E. Zenaro, D. Ferrari, L.D. Notarangelo, F. Porta, F. Facchetti, L.D. Notarangelo, S. Dusi, R. Badolato, G-CSF treatment of severe congenital neutropenia reverses neutropenia but does not correct the underlying functional deficiency of the neutrophil in defending against microorganisms. Blood 109, 4716–4723 (2007)
B. McDonald, E.F. McAvoy, F. Lam, V. Gill, C. de la Motte, R.C. Savani, P. Kubes, Interaction of CD44 and hyaluronan is the dominant mechanism for neutrophil sequestration in inflamed liver sinusoids. J. Exp. Med. 205, 915–927 (2008)
N. Alkhouri, G. Morris-Stiff, C. Campbell, R. Lopez, T.A. Tamimi, L. Yerian, N.N. Zein, A.E. Feldstein, Neutrophil to lymphocyte ratio: a new marker for predicting steatohepatitis and fibrosis in patients with nonalcoholic fatty liver disease. Liver Int. 32, 297–302 (2012)
C.Z. Larter, M.M. Yeh, W.G. Haigh, D.M. Van Rooyen, J. Brooling, D. Heydet, C.J. Nolan, N.C. Teoh, G.C. Farrell, Dietary modification dampens liver inflammation and fibrosis in obesity-related fatty liver disease. Obesity 21, 1189–1199 (2013)
E.J. Park, J.H. Lee, G.Y. Yu, G. He, S.R. Ali, R.G. Holzer, C.H. Osterreicher, H. Takahashi, M. Karin, Dietary and genetic obesity promote liver inflammation and tumorigenesis by enhancing IL-6 and TNF expression. Cell 140, 197–208 (2010)
R. Ibusuki, H. Uto, S. Arima, S. Mawatari, Y. Setoguchi, Y. Iwashita, S. Hashimoto, T. Maeda, S. Tanoue, S. Kanmura, M. Oketani, A. Ido, H. Tsubouchi, Transgenic expression of human neutrophil peptide-1 enhances hepatic fibrosis in mice fed a choline-deficient, l-amino acid-defined diet. Liver Int. 33, 1549–1556 (2013)
M.P. Murphy, How mitochondria produce reactive oxygen species. Biochem. J. 417, 1–13 (2009)
C. Koliaki, J. Szendroedi, K. Kaul, T. Jelenik, P. Nowotny, F. Jankowiak, C. Herder, M. Carstensen, M. Krausch, W.T. Knoefel, M. Schlensak, M. Roden, Adaptation of hepatic mitochondrial function in humans with non-alcoholic fatty liver is lost in steatohepatitis. Cell Metab. 21, 739–746 (2015)
J. Chida, K. Yamane, T. Takei, H. Kido, An efficient extraction method for quantitation of adenosine triphosphate in mammalian tissues and cells. Anal. Chim. Acta 727, 8–12 (2012)
K.E. Duberley, A.Y. Abramov, A. Chalasani, S.J. Heales, S. Rahman, I.P. Hargreaves, Human neuronal coenzyme Q10 deficiency results in global loss of mitochondrial respiratory chain activity, increased mitochondrial oxidative stress and reversal of ATP synthase activity: implications for pathogenesis and treatment. J. Inherit. Metab. Dis. 36, 63–73 (2013)
M.A. Selak, J.P. de Chadarevian, J.J. Melvin, W.D. Grover, L. Salganicoff, E.M. Kaye, Mitochondrial activity in Pompe’s disease. Pediatr. Neurol. 23, 54–57 (2000)
Z.F. Ba, P. Wang, D.J. Koo, W.G. Cioffi, K.I. Bland, I.H. Chaudry, Alterations in tissue oxygen consumption and extraction after trauma and hemorrhagic shock. Crit. Care Med. 28, 2837–2842 (2000)
G. Hofhaus, R.M. Shakeley, G. Attardi, Use of polarography to detect respiration defects in cell cultures. Methods Enzymol. 264, 476–483 (1996)
J. Xu, K. Cao, Y. Li, X. Zou, C. Chen, I.M. Szeto, Z. Dong, Y. Zhao, Y. Shi, J. Wang, J. Liu, Z. Feng, Bitter gourd inhibits the development of obesity-associated fatty liver in C57BL/6 mice fed a high-fat diet. J. Nutr. 144, 475–483 (2014a)
A. Carobene, F. Braga, T. Roraas, S. Sandberg, W.A. Bartlett, A systematic review of data on biological variation for alanine aminotransferase, aspartate aminotransferase and gamma-glutamyl transferase. Clin. Chem. Lab. Med. 51, 1997–2007 (2013)
A. Herms, M. Bosch, B.J. Reddy, N.L. Schieber, A. Fajardo, C. Ruperez, A. Fernandez-Vidal, C. Ferguson, C. Rentero, F. Tebar, C. Enrich, R.G. Parton, S.P. Gross, A. Pol, AMPK activation promotes lipid droplet dispersion on detyrosinated microtubules to increase mitochondrial fatty acid oxidation. Nat. Commun. 6, 7176 (2015)
X. Fu, R.M. Chin, L. Vergnes, H. Hwang, G. Deng, Y. Xing, M.Y. Pai, S. Li, L. Ta, F. Fazlollahi, C. Chen, R.M. Prins, M.A. Teitell, D.A. Nathanson, A. Lai, K.F. Faull, M. Jiang, S.G. Clarke, T.F. Cloughesy, T.G. Graeber, D. Braas, H.R. Christofk, M.E. Jung, K. Reue, J. Huang, 2-Hydroxyglutarate inhibits ATP synthase and mTOR signaling. Cell Metab. 22, 508–515 (2015)
K. Miura, L. Yang, N. van Rooijen, H. Ohnishi, E. Seki, Hepatic recruitment of macrophages promotes nonalcoholic steatohepatitis through CCR2. Am. J. Physiol. Gastrointest. Liver Physiol. 302, G1310–G1321 (2012)
V. Bieghs, P.C. Rensen, M.H. Hofker, R. Shiri-Sverdlov, NASH and atherosclerosis are two aspects of a shared disease: central role for macrophages. Atherosclerosis 220, 287–293 (2012)
K.C. McGrath, X.H. Li, P.T. Whitworth, R. Kasz, J.T. Tan, S.V. McLennan, D.S. Celermajer, P.J. Barter, K.A. Rye, A.K. Heather, High density lipoproteins improve insulin sensitivity in high-fat diet-fed mice by suppressing hepatic inflammation. J. Lipid Res. 55, 421–430 (2014)
E. Passos, C.D. Pereira, I.O. Goncalves, S. Rocha-Rodrigues, N. Silva, J.T. Guimaraes, D. Neves, A. Ascensao, J. Magalhaes, M.J. Martins, Role of physical exercise on hepatic insulin, glucocorticoid and inflammatory signaling pathways in an animal model of non-alcoholic steatohepatitis. Life Sci. 123, 51–60 (2015)
P. Filipazzi, R. Valenti, V. Huber, L. Pilla, P. Canese, M. Iero, C. Castelli, L. Mariani, G. Parmiani, L. Rivoltini, Identification of a new subset of myeloid suppressor cells in peripheral blood of melanoma patients with modulation by a granulocyte-macrophage colony-stimulation factor-based antitumor vaccine. J. Clin. Oncol. 25, 2546–2553 (2007)
K. Wing, S. Sakaguchi, Regulatory T cells exert checks and balances on self tolerance and autoimmunity. Nat. Immunol. 11, 7–13 (2010)
R. Bottino, L.A. Fernandez, C. Ricordi, R. Lehmann, M.F. Tsan, R. Oliver, L. Inverardi, Transplantation of allogeneic islets of Langerhans in the rat liver: effects of macrophage depletion on graft survival and microenvironment activation. Diabetes 47, 316–323 (1998)
R.I. Tepper, R.L. Coffman, P. Leder, An eosinophil-dependent mechanism for the antitumor effect of interleukin-4. Science 257, 548–551 (1992)
J.M. Daley, A.A. Thomay, M.D. Connolly, J.S. Reichner, J.E. Albina, Use of Ly6G-specific monoclonal antibody to deplete neutrophils in mice. J. Leukoc. Biol. 83, 64–70 (2008)
D.E. Dulek, D.C. Newcomb, K. Goleniewska, J. Cephus, W. Zhou, S. Reiss, S. Toki, F. Ye, R. Zaynagetdinov, T.P. Sherrill, T.S. Blackwell, M.L. Moore, K.L. Boyd, J.K. Kolls, R.S. Peebles Jr., Allergic airway inflammation decreases lung bacterial burden following acute Klebsiella pneumoniae infection in a neutrophil- and CCL8-dependent manner. Infect. Immun. 82, 3723–3739 (2014)
Z.G. Fridlender, J. Sun, S. Kim, V. Kapoor, G. Cheng, L. Ling, G.S. Worthen, S.M. Albelda, Polarization of tumor-associated neutrophil phenotype by TGF-beta: “N1” versus “N2” TAN. Cancer Cell 16, 183–194 (2009)
C.D. Williams, M.L. Bajt, M.R. Sharpe, M.R. McGill, A. Farhood, H. Jaeschke, Neutrophil activation during acetaminophen hepatotoxicity and repair in mice and humans. Toxicol. Appl. Pharmacol. 275, 122–133 (2014)
A.P. de Vries, P. Ruggenenti, X.Z. Ruan, M. Praga, J.M. Cruzado, I.M. Bajema, V.D. D’Agati, H.J. Lamb, D. Pongrac Barlovic, R. Hojs, M. Abbate, R. Rodriquez, C.E. Mogensen, E. Porrini; Diabesity E-EWG, Fatty kidney: emerging role of ectopic lipid in obesity-related renal disease. Lancet Diabetes Endocrinol. 2, 417–426 (2014)
L. Martinez-Rubio, S. Morais, O. Evensen, S. Wadsworth, J.G. Vecino, K. Ruohonen, J.G. Bell, D.R. Tocher, Effect of functional feeds on fatty acid and eicosanoid metabolism in liver and head kidney of Atlantic salmon (Salmo salar L.) with experimentally induced heart and skeletal muscle inflammation. Fish Shellfish. Immunol. 34, 1533–1545 (2013)
A.W. Bell, Lipid metabolism in liver and selected tissues and in the whole body of ruminant animals. Prog. Lipid Res. 18, 117–164 (1979)
W. Otten, C. Wirth, P.A. Iaizzo, H.M. Eichinger, A high omega 3 fatty acid diet alters fatty acid composition of heart, liver, kidney, adipose tissue and skeletal muscle in swine. Ann. Nutr. Metab. 37, 134–141 (1993)
F. Nassir, J.A. Ibdah, Role of mitochondria in nonalcoholic fatty liver disease. Int. J. Mol. Sci. 15, 8713–8742 (2014)
A.B. Santamarina, M. Carvalho-Silva, L.M. Gomes, M.H. Okuda, A.A. Santana, E.L. Streck, M. Seelaender, C.M. Oller do Nascimento, E.B. Ribeiro, F.S. Lira, L.M. Oyama, Decaffeinated green tea extract rich in epigallocatechin-3-gallate prevents fatty liver disease by increased activities of mitochondrial respiratory chain complexes in diet-induced obesity mice. J. Nutr. Biochem 26, 1348–1356 (2015)
J. Ye, Mechanisms of insulin resistance in obesity. Front. Med. 7, 14–24 (2013)
S.S. Rensen, V. Bieghs, S. Xanthoulea, E. Arfianti, J.A. Bakker, R. Shiri-Sverdlov, M.H. Hofker, J.W. Greve, W.A. Buurman, Neutrophil-derived myeloperoxidase aggravates non-alcoholic steatohepatitis in low-density lipoprotein receptor-deficient mice. PLoS One 7, e52411 (2012)
S. Talukdar, Y. Oh da, G. Bandyopadhyay, D. Li, J. Xu, J. McNelis, M. Lu, P. Li, Q. Yan, Y. Zhu, J. Ofrecio, M. Lin, M.B. Brenner, J.M. Olefsky, Neutrophils mediate insulin resistance in mice fed a high-fat diet through secreted elastase. Nat. Med. 18, 1407–1412 (2012)
Acknowledgements
This study was supported by the National Natural Science Foundation of China (81571395, 81373075 and 81371748).
Funding
This study was funded by the National Natural Science Foundation of China (81571395, 81373075 and 81371748).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
Procedures involving the animal experiments were approved by the Wenzhou Medical University Animal Policy and Welfare Committee.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Ou, R., Liu, J., Lv, M. et al. Neutrophil depletion improves diet-induced non-alcoholic fatty liver disease in mice. Endocrine 57, 72–82 (2017). https://doi.org/10.1007/s12020-017-1323-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-017-1323-4