Abstract
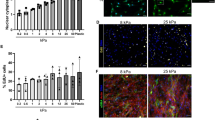
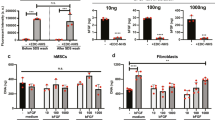
During myofiber regeneration, myoblasts are continuously subjected to shear stress. It is currently not known whether shear stress affects the regenerative capacity of myoblasts when extracellular matrix (ECM) stiffness increases (e.g. upon aging). Therefore, we aimed to assess (1) whether matrix stiffness and pulsating fluid shear stress affect myoblast proliferation and/or expression of differentiation-associated genes in myoblasts, and (2) whether matrix stiffness modulates the mechanoresponse of myoblasts to pulsating fluid shear stress. Myoblasts were seeded on matrigel-coated polyacrylamide gel matrices of varying stiffness, mimicking young (“soft”, 0.5 kPa) and old ECM (“stiff”, 20 kPa), as well as on matrigel-coated glass matrices with very high stiffness (40 ϺPa), and subjected to 1 h pulsating fluid shear stress (3 Pa/s or 4 Pa/s, 1 Hz). We found enhanced proliferation of myoblasts on stiff matrices, but reduced differentiation compared to myoblasts on soft matrices. Pulsating fluid shear stress significantly upregulated gene expression of proliferation-associated genes C-fos and Il-6, as well as expression of cytoskeletal α-actin in myoblasts seeded on glass. In contrast, pulsating fluid shear stress significantly downregulated gene expression of α-actin and Myogenin in myoblasts seeded on soft matrices. In conclusion, these results suggest that age and disease-associated increased ECM stiffness may contribute to declined regenerative capacity of myoblasts, by reducing their capacity to differentiate into new muscular tissue, at least in the absence of mechanical stimulation.



Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request. All graphs and plots corresponding to the datasets used in this study are included in this published article.
References
Charrier, E. E., Montel, L., Asnacios, A., Delort, F., Vicart, P., Gallet, F., & Hénon, S. (2018). The desmin network is a determinant of the cytoplasmic stiffness of myoblasts. Biology of the Cell, 110(4), 77–90. https://doi.org/10.1111/boc.201700040.
Domingues-Faria, C., Vasson, M. P., Goncalves-Mendes, N., Boirie, Y., & Walrand, S. (2016). Skeletal muscle regeneration and impact of aging and nutrition. Ageing Research Reviews, 26, 22–36. https://doi.org/10.1016/j.arr.2015.12.004.
Blau, H. M., Cosgrove, B. D., & Ho, A. T. V. (2015). The central role of muscle stem cells in regenerative failure with aging. Nature Medicine, 21(8), 854–862. https://doi.org/10.1038/nm.3918.
Zhang, H., Lin, S., Gao, T., Zhong, F., Cai, J., Sun, Y., & Ma, A. (2018). Association between sarcopenia and metabolic syndrome in middle-aged and older non-obese adults: A systematic review and meta-analysis. Nutrients, 10(3). https://doi.org/10.3390/nu10030364.
Rudnicki, M. A., Le Grand, F., McKinnell, I., & Kuang, S. (2008). The molecular regulation of muscle stem cell function. In Cold Spring Harbor Symposia on Quantitative Biology (Vol. 73, pp. 323–331). https://doi.org/10.1101/sqb.2008.73.064.
MAURO, A. (1961). Satellite cell of skeletal muscle fibers. The Journal of Biophysical and Biochemical Cytology, 9, 493–495. https://doi.org/10.1083/jcb.9.2.493.
Forbes, S. J., & Rosenthal, N. (2014). Preparing the ground for tissue regeneration: from mechanism to therapy. Nature Medicine, 20(8), 857–869. https://doi.org/10.1038/nm.3653.
Dumont, N. A., Wang, Y. X., & Rudnicki, M. A. (2015). Intrinsic and extrinsic mechanisms regulating satellite cell function. Development (Cambridge), 142(9), 1572–1581. https://doi.org/10.1242/dev.114223.
Le Grand, F., & Rudnicki, M. A. (2007). Skeletal muscle satellite cells and adult myogenesis. Current Opinion in Cell Biology. https://doi.org/10.1016/j.ceb.2007.09.012.
Collins, C. A., Olsen, I., Zammit, P. S., Heslop, L., Petrie, A., Partridge, T. A., & Morgan, J. E. (2005). Stem cell function, self-renewal, and behavioral heterogeneity of cells from the adult muscle satellite cell niche. Cell, 122(2), 289–301. https://doi.org/10.1016/j.cell.2005.05.010.
Morgan, J. E., & Partridge, T. A. (2003). Muscle satellite cells. International Journal of Biochemistry and Cell Biology, 35(8), 1151–1156. https://doi.org/10.1016/S1357-2725(03)00042-6.
Yucesoy, C. A., Koopman, B. H. F. J. M., Baan, G. C., Grootenboer, H. J., & Huijing, P. A. (2003). Effects of inter- and extramuscular myofascial force transmission on adjacent synergistic muscles: Assessment by experiments and finite-element modeling. Journal of Biomechanics, 36(12), 1797–1811. https://doi.org/10.1016/S0021-9290(03)00230-6.
Maas, H., Baan, G. C., Huijing, P. A., Yucesoy, C. A., Koopman, B. H. F. J. M., & Grootenboer, H. J. (2003). The relative position of EDL muscle affects the length of sarcomeres within muscle fibers: experimental results and finite-element modeling. Journal of Biomechanical Engineering, 125(5), 745–753. https://doi.org/10.1115/1.1615619.
Yucesoy, C. A., Koopman, B. H. F. J. M., Baan, G. C., Grootenboer, H. J., & Huijing, P. A. (2003). Extramuscular myofascial force transmission: Experiments and finite element modeling. Archives of Physiology and Biochemistry, 111(4), 377–388. https://doi.org/10.1080/13813450312331337630.
Boers, H. E., Haroon, M., Le Grand, F., Bakker, A. D., Klein-Nulend, J., & Jaspers, R. T. (2018). Mechanosensitivity of aged muscle stem cells. Journal of Orthopaedic Research, 36(2), 632–641. https://doi.org/10.1002/jor.23797.
Haroon, M., Klein-Nulend, J., Bakker, A. D., Jin, J., Offringa, C., Le Grand, F., … Jaspers, R. T. (2021). Myofiber stretch induces tensile and shear deformation of muscle stem cells in their native niche. Biophysical Journal, 120(13), 2665–2678. https://doi.org/10.1016/j.bpj.2021.05.021.
D’Andrea, P., Civita, D., Cok, M., Ulloa Severino, L., Vita, F., Scaini, D., & Bandiera, A. (2017). Myoblast adhesion, proliferation and differentiation on human elastin-like polypeptide (HELP) hydrogels. Journal of Applied Biomaterials and Functional Materials, 15(1), 43–53. https://doi.org/10.5301/jabfm.5000331.
Dumont, N. A., Bentzinger, C. F., Sincennes, M. C., & Rudnicki, M. A. (2015). Satellite cells and skeletal muscle regeneration. Comprehensive Physiology, 5(3), 1027–1059. https://doi.org/10.1002/cphy.c140068.
Dumont, N. A., Wang, Y. X., Von Maltzahn, J., Pasut, A., Bentzinger, C. F., Brun, C. E., & Rudnicki, M. A. (2015). Dystrophin expression in muscle stem cells regulates their polarity and asymmetric division. Nature Medicine, 21(12), 1455–1463. https://doi.org/10.1038/nm.3990.
Yu, L., Ma, X., Sun, J., Tong, J., Shi, L., Sun, L., & Zhang, J. (2017). Fluid shear stress induces osteoblast differentiation and arrests the cell cycle at the G0 phase via the ERK1/2 pathway. Molecular Medicine Reports, 6(16), 8699–8708. https://doi.org/10.3892/mmr.2017.7720.
Ahsan, T., & Nerem, R. M. (2010). Fluid shear stress promotes an endothelial-like phenotype during the early differentiation of embryonic stem cells. Tissue Engineering - Part A, 16(11), 3547–3553. https://doi.org/10.1089/ten.tea.2010.0014.
Nishii, K., Brodin, E., Renshaw, T., Weesner, R., Moran, E., Soker, S., & Sparks, J. L. (2018). Shear stress upregulates regeneration-related immediate early genes in liver progenitors in 3D ECM-like microenvironments. Journal of Cellular Physiology, 233(5), 4272–4281. https://doi.org/10.1002/jcp.26246.
Wood, L. K., Kayupov, E., Gumucio, J. P., Mendias, C. L., Claflin, D. R., & Brooks, S. V. (2014). Intrinsic stiffness of extracellular matrix increases with age in skeletal muscles of mice. Journal of Applied Physiology, 117(4), 363–369. https://doi.org/10.1152/japplphysiol.00256.2014.
Alnaqeeb, M. A., Al Zaid, N. S., & Goldspink, G. (1984). Connective tissue changes and physical properties of developing and ageing skeletal muscle. Journal of anatomy, 139(4), 677–89. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/6526719%0A or http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=PMC1164979.
Grounds, M. D., Sorokin, L., & White, J. (2005). Strength at the extracellular matrix-muscle interface. Scandinavian Journal of Medicine and Science in Sports, 15(6), 381–391. https://doi.org/10.1111/j.1600-0838.2005.00467.x.
Lacraz, G., Rouleau, A. J., Couture, V., Söllrald, T., Drouin, G., Veillette, N., … Grenier, G. (2015). Increased stiffness in aged skeletal muscle impairs muscle progenitor cell proliferative activity. PLoS ONE, 10(8). https://doi.org/10.1371/journal.pone.0136217.
Hynes, R. O. (2002). Integrins: Bidirectional, allosteric signaling machines. Cell, 110(6), 673–687. https://doi.org/10.1016/S0092-8674(02)00971-6.
Yeh, Y. C., Ling, J. Y., Chen, W. C., Lin, H. H., & Tang, M. J. (2017). Mechanotransduction of matrix stiffness in regulation of focal adhesion size and number: Reciprocal regulation of caveolin-1 and β1 integrin. Scientific Reports, 7(1). https://doi.org/10.1038/s41598-017-14932-6.
Sun, Z., Guo, S. S., & Fässler, R. (2016). Integrin-mediated mechanotransduction. Journal of Cell Biology, 215(4), 445–456. https://doi.org/10.1083/jcb.201609037.
Guo, M., Pegoraro, A. F., Mao, A., Zhou, E. H., Arany, P. R., Han, Y., & Weitz, D. A. (2017). Cell volume change through water efflux impacts cell stiffness and stem cell fate. Proceedings of the National Academy of Sciences of the United States of America, 114(41), E8618–E8627. https://doi.org/10.1073/pnas.1705179114.
Boontheekul, T., Hill, E. E., Kong, H. J., & Mooney, D. J. (2007). Regulating myoblast phenotype through controlled gel stiffness and degradation. Tissue Engineering, 13(7), 1431–1442. https://doi.org/10.1089/ten.2006.0356.
Yaffe, D., & Saxel, O. (1977). Serial passaging and differentiation of myogenic cells isolated from dystrophic mouse muscle. Nature, 270(5639), 725–727. https://doi.org/10.1038/270725a0.
Colin-York, H., Eggeling, C., & Fritzsche, M. (2017). Dissection of mechanical force in living cells by super-resolved traction force microscopy. Nature Protocols, 12(4), 783–796. https://doi.org/10.1038/nprot.2017.009.
Tse, J. R., & Engler, A. J. (2010). Preparation of hydrogel substrates with tunable mechanical properties. Current Protocols in Cell Biology. https://doi.org/10.1002/0471143030.cb1016s47.
Bacabac, R. G., Smith, T. H., Heethaar, R. M., Van Loon, J. J. W. A., Pourquie, M. J. M. B., Nieuwstadt, F. T. M., & Klein-Nulend, J. (2002). Characteristics of the parallel-plate flow chamber for mechanical stimulation of bone cells under microgravity. In European Space Agency, (Special Publication) ESA SP (pp. 83–84).
Razavi, M. A., Wong, J., Akkera, M., Shalaby, M., Shalaby, H., Sholl, A., & Lee, G. S. (2020). Nuclear morphometry in indeterminate thyroid nodules. Gland Surgery, 9(2), 238–244. https://doi.org/10.21037/gs.2020.02.02.
Shiihara, H., Terasaki, H., Sonoda, S., Kakiuchi, N., Yamaji, H., Yamaoka, S., … Sakamoto, T. (2020). Association of foveal avascular zone with the metamorphopsia in epiretinal membrane. Scientific Reports, 10(1). https://doi.org/10.1038/s41598-020-74190-x.
Panda, A. C., Abdelmohsen, K., Yoon, J.-H., Martindale, J. L., Yang, X., Curtis, J., & Gorospe, M. (2014). RNA-binding protein AUF1 promotes myogenesis by regulating MEF2C expression levels. Molecular and Cellular Biology, 34(16), 3106–3119. https://doi.org/10.1128/mcb.00423-14.
Kassar-Duchossoy, L., Gayraud-Morel, B., Gomès, D., Rocancourt, D., Buckingham, M., Shinin, V., & Tajbakhsh, S. (2004). Mrf4 determines skeletal muscle identity in Myf5:Myod double-mutant mice. Nature, 431(7007), 466–471. https://doi.org/10.1038/nature02876.
Gissel, H. (2006). The role of Ca2+ in muscle cell damage. Annals of the New York Academy of Sciences. https://doi.org/10.1196/annals.1363.013.
Glogauer, M., Arora, P., Yao, G., Sokholov, I., Ferrier, J., & McCulloch, C. A. G. (1997). Calcium ions and tyrosine phosphorylation interact coordinately with actin to regulate cytoprotective responses to stretching. Journal of Cell Science, 110(1), 11–21. https://doi.org/10.1242/jcs.110.1.11.
Ogilvie, R. W., Armstrong, R. B., Baird, K. E., & Bottoms, C. L. (1988). Lesions in the rat soleus muscle following eccentrically biased exercise. American Journal of Anatomy, 182(4), 335–346. https://doi.org/10.1002/aja.1001820405.
Fridén, J., & Lieber, R. L. (1998). Segmental muscle fiber lesions after repetitive eccentric contractions. Cell and Tissue Research, 293(1), 165–171. https://doi.org/10.1007/s004410051108.
Sciorati, C., Rigamonti, E., Manfredi, A. A., & Rovere-Querini, P. (2016). Cell death, clearance and immunity in the skeletal muscle. Cell Death and Differentiation, 23(6), 927–937. https://doi.org/10.1038/cdd.2015.171.
Bechshøft, C. J. L., Jensen, S. M., Schjerling, P., Andersen, J. L., Svensson, R. B., Eriksen, C. S., & Mackey, A. L. (2019). Age and prior exercise in vivo determine the subsequent in vitro molecular profile of myoblasts and nonmyogenic cells derived from human skeletal muscle. American Journal of Physiology - Cell Physiology, 316(6), C898–C912. https://doi.org/10.1152/ajpcell.00049.2019.
Yeung, T., Georges, P. C., Flanagan, L. A., Marg, B., Ortiz, M., Funaki, M., & Janmey, P. A. (2005). Effects of substrate stiffness on cell morphology, cytoskeletal structure, and adhesion. Cell Motility and the Cytoskeleton, 60(1), 24–34. https://doi.org/10.1002/cm.20041.
Shih, Y. R. V., Tseng, K. F., Lai, H. Y., Lin, C. H., & Lee, O. K. (2011). Matrix stiffness regulation of integrin-mediated mechanotransduction during osteogenic differentiation of human mesenchymal stem cells. Journal of Bone and Mineral Research, 26(4), 730–738. https://doi.org/10.1002/jbmr.278.
Kohn, J. C., Zhou, D. W., Bordeleau, F., Zhou, A. L., Mason, B. N., Mitchell, M. J., & Reinhart-King, C. A. (2015). Cooperative effects of matrix stiffness and fluid shear stress on endothelial cell behavior. Biophysical Journal, 108(3), 471–478. https://doi.org/10.1016/j.bpj.2014.12.023.
Serrano, A. L., Baeza-Raja, B., Perdiguero, E., Jardí, M., & Muñoz-Cánoves, P. (2008). Interleukin-6 is an essential regulator of satellite cell-mediated skeletal muscle hypertrophy. Cell Metabolism, 7(1), 33–44. https://doi.org/10.1016/j.cmet.2007.11.011.
Li, L., Chambard, J. C., Karin, M., & Olson, E. N. (1992). Fos and Jun repress transcriptional activation by myogenin and MyoD: The amino terminus of Jun can mediate repression. Genes and Development, 6(4), 676–689. https://doi.org/10.1101/gad.6.4.676.
Brown, J. R., Nigh, E., Lee, R. J., Ye, H., Thompson, M. A., Saudou, F., & Greenberg, M. E. (1998). Fos family members induce cell cycle entry by activating cyclin D1. Molecular and Cellular Biology, 18(9), 5609–5619. https://doi.org/10.1128/mcb.18.9.5609.
Daury, L., Busson, M., Tourkine, N., Casas, F., Cassar-Malek, I., Wrutniak-Cabello, C., & Cabello, G. (2001). Opposing functions of ATF2 and Fos-like transcription factors in c-Jun-mediated myogenin expression and terminal differentiation of avian myoblasts. Oncogene, 20(55), 7998–8008. https://doi.org/10.1038/sj.onc.1204967.
Guigó, R., Dermitzakis, E. T., Agarwal, P., Ponting, C. P., Parra, G., Reymond, A., … Brent, M. R. (2003). Comparison of mouse and human genomes followed by experimental verification yields an estimated 1,019 additional genes. Proceedings of the National Academy of Sciences of the United States of America, 100(3), 1140–1145. https://doi.org/10.1073/pnas.0337561100.
Chargé, S. B. P., & Rudnicki, M. A. (2004). Cellular and molecular regulation of muscle regeneration. Physiological Reviews, 84(1), 209–238. https://doi.org/10.1152/physrev.00019.2003.
Yin, H., Price, F., & Rudnicki, M. A. (2013). Satellite cells and the muscle stem cell niche. Physiological Reviews, 93(1), 23–67. https://doi.org/10.1152/physrev.00043.2011.
Acknowledgements
We thank Carla Offringa, Guus Baan and Gerard de Wit from the Laboratory for Myology of the Vrije Universiteit Amsterdam for technical support with experiments. We thank Mohammad Haroon for his technical support on PFF experiments.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Victor van Santen. The first draft of the manuscript was written by Victor van Santen and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
van Santen, V.J.B., Klein-Nulend, J., Bakker, A.D. et al. Stiff matrices enhance myoblast proliferation, reduce differentiation, and alter the response to fluid shear stress in vitro. Cell Biochem Biophys 80, 161–170 (2022). https://doi.org/10.1007/s12013-021-01050-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-021-01050-4