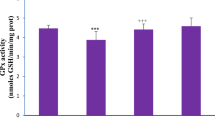
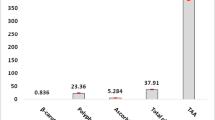
Abstract
Heavy metals (HM)in the environment have provoked global attention because of its deleterious effects. This study evaluated the protection offered by Zn or Se or both against HMM-induced alterations in the kidney. Male Sprague Dawley rats were distributed into 5 groups of 7 rats each. Group I served as normal control with unrestricted access to food and water. Group II received Cd, Pb, and As (HMM) per oral daily for 60 days while groups III and IV received HMM in addition to Zn and Se respectively for 60 days. Group V received both Zn and Se in addition to HMM for 60 days. Metal accumulation in feces was assayed at days 0, 30, and 60 while accumulation in the kidney and kidney weight were measured at day 60. Kidney function tests, NO, MDA, SOD, catalase, GSH, GPx, NO, IL-6, NF-Κb, TNFα, caspase 3, and histology were assessed. There is a significant increase in urea, creatinine, and bicarbonate ions while potassium ions decreased. There was significant increase in renal function biomarkers, MDA, NO, NF-Κb, TNFα, caspase 3, and IL-6 while SOD, catalase, GSH, and GPx decrease. Administration of HMM distorted the integrity of the rat kidney, and co-treatment with Zn or Se or both offered reasonable protection suggesting that Zn or Se could be used as an antidot against the deleterious effects of these metals.
Graphical Abstract







Similar content being viewed by others
Data Availability
All data have been provided.
References
Anyanwu BO, Ezejiofor AN, Igweze ZN, Orisakwe OE (2018) Heavy metal mixture exposure and effects in developing nations: an update. Toxics 6(4):65
WHO (2007) Health risks of heavy metals from long-range transboundary air pollution. World Health Organization. Regional Office for Europe
Lynch NR, Hoang TC, O'Brien TE (2016) Acute toxicity of binary-metal mixtures of copper, zinc, and nickel to Pimephales promelas: evidence of more-than-additive effect. Environ Toxicol Chem 35:446–457
Orr SE, Barnes MC, George HS, Joshee L, Jeon B, Scircle A, Black O, Cizdziel JV, Smith BE, Bridges CC (2018) Exposure to mixtures of mercury, cadmium, lead, and arsenic alters the disposition of single metals in tissues of Wistar rats. J Toxicol Environ Health Part A 81:1246–1256
Ara A, Usmani JA (2015) Lead toxicity: a review. Interdiscipl Toxicol 8:55
Jihen el H, Imed M, Fatima H, Abdelhamid K (2008) Protective effects of selenium (Se) and zinc (Zn) on cadmium (Cd) toxicity in the liver and kidney of the rat: histology and Cd accumulation. Food Chem Toxicol 46:3522–3527
Rădulescu A, Lundgren S (2019) A pharmacokinetic model of lead absorption and calcium competitive dynamics. Sci Rep 9:14225
Chen S-J, Yan X-J, Chen Z (2013) Arsenic in tissues, organs, and cells. In: Kretsinger RH, Uversky VN, Permyakov EA (eds) Encyclopedia of Metalloproteins. Springer New York, New York, NY, pp 135–138
Balali-Mood M, Naseri K, Tahergorabi Z, Khazdair MR, Sadeghi M (2021) Toxic mechanisms of five heavy metals: mercury, lead, chromium, cadmium, and arsenic. Front Pharmacol 12
Awoke JN, Orji OU, Aja PM, Ezeani NN, Aloke C, Obasi OD (2020) Ethanol leaf extract of Ruspolia hypocrateriformis abrogated hepatic redox imbalance and oxidative damage induced by heavy metal toxicity in rats. Arab J Chem 13:8133–8145
García-Niño WR, Pedraza-Chaverrí J (2014) Protective effect of curcumin against heavy metals-induced liver damage. Food Chem Toxicol 69:182–201
Valko M, Morris H, Cronin MT (2005) Metals, toxicity and oxidative stress. Curr Med Chem 12:1161–1208
Yan L-J, Allen DC (2021) Cadmium-induced kidney injury: oxidative damage as a unifying mechanism. Biomolecules 11:1575
Patra RC, Rautray AK, Swarup D (2011) Oxidative stress in lead and cadmium toxicity and its amelioration. Vet Med Int 2011:457327
Ajarem JS, Hegazy AK, Allam GA, Allam AA, Maodaa SN, Mahmoud AM (2022) Heavy metal accumulation, tissue injury, oxidative stress, and inflammation in dromedary camels living near petroleum industry sites in Saudi Arabia. Animals 12:707
Kovalchuk L, Mikshevich N (2017) Accumulation of heavy metals by small mammals the background and polluted territories of the Urals. Vestn Zool 51:325
Tak PP, Firestein GS (2001) NF-κB: a key role in inflammatory diseases. J Clin Invest 107:7–11
Zhong J, Lu P, Wu H, Liu Z, Sharifi-Rad J, Setzer WN, Suleria HA (2022, 2022) Current insights into phytochemistry, nutritional, and pharmacological properties of prosopis plants. Evid-based Complement Altern Med
Wang Y, Wang K, Han T, Zhang P, Chen X, Wu W, Feng Y, Yang H, Li M, Xie B, Guo P, Warren JL, Shi X, Wang S, Zhang Y (2020a) Exposure to multiple metals and prevalence for preeclampsia in Taiyuan, China. Environ Int 145:106098
Okoye EA, Bocca B, Ruggieri F, Ezejiofor AN, Nwaogazie IL, Domingo JL, Rovira J, Frazzoli C, Orisakwe OE (2021) Metal pollution of soil, plants, feed and food in the Niger Delta, Nigeria: health risk assessment through meat and fish consumption. Environ Res 198:111273
Maduabuchi JMU, Nzegwu CN, Adigba EO, Oragwu CI, Agbo FN, Agbata CA, Ani GC, Orisakwe OE (2008) Iron, manganese and nickel exposure from beverages in Nigeria: a public health concern? J Health Sci 54(3):335–338
Roberts II, Orisakwe OE (2011) Evaluation of potential dietary toxicity of heavy metals in some common Nigerian beverages: a look at antimony, tin and mercury. QSci Connect 2011(1):2
Orisakwe OE, Frazzoli C, Ilo CE, Oritsemuelebi B (2019) Public health burden of e-waste in Africa. J Health Pollut 9(22):190610
Yu H-T, Zhen J, Leng J-Y, Cai L, Ji H-L, Keller BB (2021) Zinc as a countermeasure for cadmium toxicity. Acta Pharmacol Sin 42:340–346
Olechnowicz J, Tinkov A, Skalny A, Suliburska J (2018) Zinc status is associated with inflammation, oxidative stress, lipid, and glucose metabolism. J Physiol Sci 68:19–31
Zhang D, Liu J, Gao J, Shahzad M, Han Z, Wang Z, Li J, Sjölinder H (2014) Zinc supplementation protects against cadmium accumulation and cytotoxicity in Madin-Darby bovine kidney cells. PLoS One 9:e103427
Ge J, Liu L-L, Cui Z-G, Talukder M, Lv M-W, Li J-Y, Li J-L (2021) Comparative study on protective effect of different selenium sources against cadmium-induced nephrotoxicity via regulating the transcriptions of selenoproteome. Ecotoxicol Environ Saf 215:112135
Vodela J, Renden J, Lenz S, McElhenney W, Kemppainen B (1997) Drinking water contaminants (arsenic, cadmium, lead, benzene, and trichloroethylene). 1. Interaction of contaminants with nutritional status on general performance and immune function in broiler chickens. Poult Sci 76:1474–1492
Fiati Kenston SS, Su H, Li Z, Kong L, Wang Y, Song X, Gu Y, Barber T, Aldinger J, Hua Q (2018) The systemic toxicity of heavy metal mixtures in rats. Toxicol Res 7:396–407
Anyanwu BO, Orish CN, Ezejiofor AN, Nwaogazie IL, Orisakwe OE (2020) Protective effect of costus afer aqueous leaf extract (CALE) on low-dose heavy metal mixture-induced alterations in serum lipid profile and hematological parameters of male Wistar Albino Rats. J Toxicol 2020:8850264
Cobbina SJ, Chen Y, Zhou Z, Wu X, Zhao T, Zhang Z, Feng W, Wang W, Li Q, Wu X, Yang L (2015) Toxicity assessment due to sub-chronic exposure to individual and mixtures of four toxic heavy metals. J Hazard Mater 294:109–120
Okolo KO, Orisakwe OE, Siminialayi IM (2017) Pleurotus tuber-regium mushrooms in the diet of rats ameliorates reproductive and testicular injury caused by carbon tetrachloride. Clin Phytosci 3:14
Draper HH, Hadley M (1990) Malondialdehyde determination as index of lipid Peroxidation. Methods in Enzymology, Academic Press, pp. 421-431
Nims RW, Darbyshire JF, Saavedra JE, Christodoulou D, Hanbauer I, Cox GW, Grisham MB, Laval F, Cook JA, Krishna MC, Wink DA (1995) Colorimetric methods for the determination of nitric oxide concentration in neutral aqueous solutions. Methods 7:48–54
Tipple TE, Rogers LK (2012) Methods for the determination of plasma or tissue glutathione levels. Developmental Toxicology, Springer, pp 315–324
Weydert CJ, Cullen JJ (2010) Measurement of superoxide dismutase, catalase and glutathione peroxidase in cultured cells and tissue. Nat Protoc 5:51–66
Kovesdy CP (2022) Epidemiology of chronic kidney disease: an update 2022. Kidney Int Suppl 12:7–11
Moody EC, Coca SG, Sanders AP (2018) Toxic metals and chronic kidney disease: a systematic review of recent literature. Curr Environ Health Rep 5:453–463
Shaikh ZA, Smith LM (1984) Biological indicators of cadmium exposure and toxicity. Experientia 40:36–43
Yabe J, Nakayama SMM, Ikenaka Y, Yohannes YB, Bortey-Sam N, Kabalo AN, Ntapisha J, Mizukawa H, Umemura T, Ishizuka M (2018) Lead and cadmium excretion in feces and urine of children from polluted townships near a lead-zinc mine in Kabwe, Zambia. Chemosphere 202:48–55
Chan Yoo Y, Ki Lee S, Yeol Yang J, Whan In S, Wook Kim K, Hyuck Chung K, Gyu Chung M, Young Choung S (2002) Organ distribution of heavy metals in autopsy material from Normal Korean. J Health Sci 48:186–194
O'Flaherty EJ (1998) Physiologically based models of metal kinetics. Crit Rev Toxicol 28:271–317
Ratnaike RN (2003) Acute and chronic arsenic toxicity. Postgrad Med J 79:391
Lihm H, Kim H, Chang H, Yoon M, Lee K, Choi J (2013) Vitamin C modulates lead excretion in rats. Anat Cell Biol 46:239–245
Brzóska MM, Moniuszko-Jakoniuk J (2001) Interactions between cadmium and zinc in the organism. Food Chem Toxicol 39:967–980
Donma O, Donma MM (2005) Cadmium, lead and phytochemicals. Med Hypotheses 65:699–702
Messaoudi I, El Heni J, Hammouda F, Saïd K, Kerkeni A (2009) Protective effects of selenium, zinc, or their combination on cadmium-induced oxidative stress in rat kidney. Biol Trace Elem Res 130:152–161
Ponomarenko O, La Porte PF, Singh SP, Langan G, Fleming DEB, Spallholz JE, Alauddin M, Ahsan H, Ahmed S, Gailer J, George GN, Pickering IJ (2017) Selenium-mediated arsenic excretion in mammals: a synchrotron-based study of whole-body distribution and tissue-specific chemistry. Metallomics 9:1585–1595
Tai Y-T, Chou S-H, Cheng C-Y, Ho C-T, Lin H-C, Jung S-M, Chu P-H, Ko F-H (2022) The preferential accumulation of cadmium ions among various tissues in mice. Toxicol Rep 9:111–119
Vesey DA (2010) Transport pathways for cadmium in the intestine and kidney proximal tubule: focus on the interaction with essential metals. Toxicol Lett 198:13–19
Rana MN, Tangpong J, Rahman MM (2018) Toxicodynamics of lead, cadmium, mercury and arsenic- induced kidney toxicity and treatment strategy: a mini review. Toxicol Rep 5:704–713
Sosa C, Guillen N, Lucea S, Sorribas V (2020) Effects of oral exposure to arsenite on arsenic metabolism and transport in rat kidney. Toxicol Lett 333:4–12
Waalkes MP (1986) Effect of dietary zinc deficiency on the accumulation of cadmium and metallothionein in selected tissues of the rat. J Toxicol Environ Health 18:301–313
Jamieson JA, Stringer DM, Zahradka P, Taylor CG (2008) Dietary zinc attenuates renal lead deposition but metallothionein is not directly involved. Biometals 21:29–40
Sabolić I, Breljak D, Skarica M, Herak-Kramberger CM (2010) Role of metallothionein in cadmium traffic and toxicity in kidneys and other mammalian organs. Biometals 23:897–926
Garla R, Sharma N, Kaushal NS, Garg ML (2021) Effect of zinc on hepatic and renal tissues of chronically arsenic exposed rats: a biochemical and histopathological study. Biol Trace Elem Res 199:4237–4250
Wang Y, Zhao H, Nie X, Guo M, Jiang G, Xing M (2020b) Zinc application alleviates the adverse renal effects of arsenic stress in a protein quality control way in common carp. Environ Res 191:110063
Wahba ZZ, Coogan TP, Rhodes SW, Waalkes MP (1993) Protective effects of selenium on cadmium toxicity in rats: role of altered toxicokinetics and metallothionein. J Toxicol Environ Health 38:171–182
Flora SJ, Singh S, Tandon SK (1983) Role of selenium in protection against lead intoxication. Acta Pharmacol Toxicol 53:28–32
Park Y, Lee SJ (2022) Association of blood heavy metal levels and renal function in Korean adults. Int J Environ Res Public Health 19
Barbier O, Jacquillet G, Tauc M, Cougnon M, Poujeol P (2005) Effect of heavy metals on, and handling by, the kidney. Nephron Physiol 99:105–110
Patel K, Batura D (2020) An overview of hydronephrosis in adults. Br J Hosp Med 81:1–8
El-Nahas AR, Elsaadany MM, Taha DE, Elshal AM, El-Ghar MA, Ismail AM, Elsawy EA, Saleh HH, Wafa EW, Awadalla A (2017) A randomised controlled trial evaluating renal protective effects of selenium with vitamins A, C, E, verapamil, and losartan against extracorporeal shockwave lithotripsy-induced renal injury. BJU Int 119:142–147
Andjelkovic M, Buha Djordjevic A, Antonijevic E, Antonijevic B, Stanic M, Kotur-Stevuljevic J, Spasojevic-Kalimanovska V, Jovanovic M, Boricic N, Wallace D (2019) Toxic effect of acute cadmium and lead exposure in rat blood, liver, and kidney. Int J Environ Res Public Health 16:274
ElMahdy MK, Zaki MO, Al-Karmalawy AA, Abdo W, Alnasser SM, Antar SA (2022) Glimepiride ameliorates renal toxicity induced by cadmium in mice: modulation of Jun N terminal kinase (JNK)/nuclear factor kappa B (NF-κB) and phosphatidylinositol 3-kinases (PI3K)/protein kinase (AKT) pathways. Life Sci:121184
Gabr SA, Alghadir AH, Ghoniem GA (2019) Biological activities of ginger against cadmium-induced renal toxicity. Saudi J Biol Sci 26:382–389
Fatema K, Shoily SS, Ahsan T, Haidar Z, Sumit AF, Sajib AA (2021) Effects of arsenic and heavy metals on metabolic pathways in cells of human origin: similarities and differences. Toxicol Rep 8:1109–1120
Hasanein P, Emamjomeh A (2019) Chapter 28 - Beneficial effects of natural compounds on heavy metal–induced hepatotoxicity. In: Watson RR, Preedy VR (eds) Dietary Interventions in Liver Disease. Academic Press, pp 345–355
Bao RK, Zheng SF, Wang XY (2017) Selenium protects against cadmium-induced kidney apoptosis in chickens by activating the PI3K/AKT/Bcl-2 signaling pathway. Environ Sci Pollut Res Int 24:20342–20353
Thomas DD (2015) Breathing new life into nitric oxide signaling: a brief overview of the interplay between oxygen and nitric oxide. Redox Biol 5:225–233
Exner R, Wessner B, Manhart N, Roth E (2000) Therapeutic potential of glutathione. Wien Klin Wochenschr 112:610–616
Fujii J, Homma T, Osaki T (2022) Superoxide radicals in the execution of cell death. Antioxidants 11(3):501
Chen F, Shi X (2002) Signaling from toxic metals to NF-kappaB and beyond: not just a matter of reactive oxygen species. Environ Health Perspect 110(Suppl 5):807–811
Zhang H, Li L, Wang Y, Dong F, Chen X, Liu F, Xu D, Yi F, Kapron CM, Liu J (2016) NF-κB signaling maintains the survival of cadmium-exposed human renal glomerular endothelial cells. Int J Mol Med 38:417–422
Lee SA, Cozzi M, Bush EL, Rabb H (2018) Distant organ dysfunction in acute kidney injury: a review. Am J Kidney Dis 72:846–856
Buha A, Baralić K, Djukic-Cosic D, Bulat Z, Tinkov A, Panieri E, Saso L (2021) The role of toxic metals and metalloids in Nrf2 signaling. Antioxidants 10:630
El-Boshy ME, Risha EF, Abdelhamid FM, Mubarak MS, Hadda TB (2015) Protective effects of selenium against cadmium induced hematological disturbances, immunosuppressive, oxidative stress and hepatorenal damage in rats. J Trace Elem Med Biol 29:104–110
Di Leo V, D'Incà R, Barollo M, Tropea A, Fries W, Mazzon E, Irato P, Cecchetto A, Sturniolo GC (2001) Effect of zinc supplementation on trace elements and intestinal metallothionein concentrations in experimental colitis in the rat. Dig Liver Dis 33:135–139
Swindell WR (2011) Metallothionein and the biology of aging. Ageing Res Rev 10:132–145
Wang W-C, Mao H, Ma D-D, Yang W-X (2014) Characteristics, functions, and applications of metallothionein in aquatic vertebrates. Front Mar Sci 1
Liu L, Yang B, Cheng Y, Lin H (2015) Ameliorative effects of selenium on cadmium-induced oxidative stress and endoplasmic reticulum stress in the chicken kidney. Biol Trace Elem Res 167:308–319
Skalny AA, Tinkov AA, Medvedeva YS, Alchinova IB, Karganov MY, Skalny AV, Nikonorov AA (2015) Effect of short-term zinc supplementation on zinc and selenium tissue distribution and serum antioxidant enzymes. Acta Sci Pol Technol Aliment 14:269–276
Zwolak I (2020) The role of selenium in arsenic and cadmium toxicity: an updated review of scientific literature. Biol Trace Elem Res 193:44–63
Minich WB (2022) Selenium metabolism and biosynthesis of selenoproteins in the human body. Biochemistry 87:S168–S177
Xu C, Sun S, Johnson T, Qi R, Zhang S, Zhang J, Yang K (2021) The glutathione peroxidase Gpx4 prevents lipid peroxidation and ferroptosis to sustain Treg cell activation and suppression of antitumor immunity. Cell Rep 35:109235
Lentini P, Zanoli L, Granata A, Signorelli SS, Castellino P, Dell'Aquila R (2017) Kidney and heavy metals - the role of environmental exposure (Review). Mol Med Rep 15:3413–3419
Paugam-Burtz C, Levesque E, Louvet A, Thabut D, Amathieu R, Bureau C, Camus C, Chanques G, Faure S, Ferrandière M, Francoz C, Galbois A, Gustot T, Ichai C, Ichai P, Jaber S, Lescot T, Moreau R, Roullet S et al (2020) Management of liver failure in general intensive care unit. Anaesth Crit Care Pain Med 39:143–161
Author information
Authors and Affiliations
Contributions
HO, ANE, CNO Bench work, Data Sampling and data acquisition; KOO, AC, AC Data analyses and drafting of manuscript, OEO Conceptualization and drafting of manuscript.
Corresponding author
Ethics declarations
Ethical Approval
All the procedures involving the animals and the experimental protocol followed guidelines for the safe use of animals in research and were approved by the university of Port Harcourt animal research committee (UPH/CEREMAD/REC/MM72/093).
Consent to Participate
Not applicable.
Consent for Publication
All authors have given their consent for publication.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ozoani, H., Ezejiofor, A.N., Okolo, K.O. et al. Ameliorative Effects of Zn and Se Supplementation on Heavy Metal Mixture Burden via Increased Renal Metal Excretion and Restoration of Redoxo-Inflammatory Alterations. Biol Trace Elem Res 202, 643–658 (2024). https://doi.org/10.1007/s12011-023-03709-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-023-03709-w