Abstract
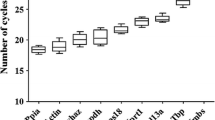
Normalization of the quantitative real-time PCR (RT-qPCR) data to the stably expressed reference genes is critically important for obtaining reliable results. However, all previous studies focused on F− toxicity for brain tissues used a single, non-validated reference gene, what might be a cause of contradictory or false results. The present study was designed to analyze the expression of a series of reference genes to select optimal ones for RT-qPCR analysis in cortex and hippocampus of rats chronically exposed to excessive fluoride (F−) amounts. Six-week-old male Wistar rats randomly assigned to four groups consumed regular tap water with 0.4 (control), 5, 20, and 50 ppm F− (NaF) for 12 months. The expression of six genes (Gapdh, Pgk1, Eef1a1, Ppia, Tbp, Helz) was compared by RT-qPCR in brain tissues from control and F−-exposed animals. The stability of candidate reference genes was evaluated by coefficient of variation (CV) analysis and RefFinder online program summarizing the results of four well-acknowledged statistical methods (Delta-Ct, BestKeeper, NormFinder, and GeNorm). In spite of some discrepancies in gene ranking between these algorisms, Pgk1, Eef1a1, and Ppia were found to be most valid in cortex, while Ppia, Eef1a1, and Helz showed the greatest expression stability in hippocampus. Tbp and Helz were identified as the least stable genes in cortex, whereas Gapdh and Tbp are unsuitable for hippocampus. These data indicate that reliable mRNA quantification in the cortex and hippocampus of F−-poisoned rats is possible using normalization to geometric mean of Pgk1+Eef1a1 or Ppia+Eef1a1 expression, respectively.




Similar content being viewed by others
Data Availability
Data will be made available on request.
References
Johnston NR, Strobel SA (2020) Principles of fluoride toxicity and the cellular response: a review. Arch Toxicol 94:1051–1069. https://doi.org/10.1007/s00204-020-02687-5
Srivastava S, Flora SJS (2020) Fluoride in drinking water and skeletal fluorosis: a review of the global impact. Curr Environ Health Rep 7:140–146. https://doi.org/10.1007/s40572-020-00270-9
Spencer AJ, Do LG, Mueller U, Baines J, Foley M, Peres MA (2018) Understanding optimum fluoride intake from population-level evidence. Adv Dent Res 29:144–156. https://doi.org/10.1177/0022034517750592
Sarkar S, Gill SS, Gupta GD, Verma SK (2022) Water toxicants: a comprehension on their health concerns, detection, and remediation. Environ Sci Pollut Res Int 29:53934–53953. https://doi.org/10.1007/s11356-022-20384-x
WHO (2020) 10 chemicals of public health concern (who.int)
Dec K, Lukomska A, Maciejewska D, Jakubczyk K, Baranowska-Bosiacka I, Chlubek D, Wasik A, Gutowska I (2017) The influence of fluorine on the disturbances of homeostasis in the central nervous system. Biol Trace Elem Res 177:224–234. https://doi.org/10.1007/s12011-016-0871-4
Agalakova NI, Nadei OV (2020) Inorganic fluoride and functions of brain. Crit Rev Toxicol 50:28–46. https://doi.org/10.1080/10408444.2020.1722061
Huggett J, Dheda K, Bustin S, Zumla A (2005) Real-time RT-PCR normalisation; strategies and considerations. Genes Immun 6:279–284. https://doi.org/10.1038/sj.gene.6364190
Nolan T, Hands RE, Bustin SA (2006) Quantification of mRNA using real-time RT-PCR. Nat Protoc 1:1559–1582. https://doi.org/10.1038/nprot.2006.236
Maurer-Morelli CV, de Vasconcellos JF, Reis-Pinto FC, Rocha CS, Domingues RR, Yasuda CL, Tedeschi H, De Oliveira E, Cendes F, Lopes-Cendes I (2012) A comparison between different reference genes for expression studies in human hippocampal tissue. J Neurosci Methods 208:44–47. https://doi.org/10.1016/j.jneumeth.2012.04.020
Jacob F, Guertler R, Naim S, Nixdorf S, Fedier A, Hacker NF, Heinzelmann-Schwarz V (2013) Careful selection of reference genes is required for reliable performance of RT-qPCR in human normal and cancer cell lines. PLoS One 8:e59180. https://doi.org/10.1371/journal.pone.0059180
Faibish D, Suzuki M, Bartlett JD (2016) Appropriate real-time PCR reference genes for fluoride treatment studies performed in vitro or in vivo. Arch Oral Biol 62:33–42. https://doi.org/10.1016/j.archoralbio.2015.11.004
Ge Y, Chen L, Yin Z, Song X, Ruan T, Hua L, Liu J, Wang J, Ning H (2018) Fluoride-induced alterations of synapse-related proteins in the cerebral cortex of ICR offspring mouse brain. Chemosphere 201:874–883. https://doi.org/10.1016/j.chemosphere.2018.02.167
Łukomska A, Baranowska-Bosiacka I, Dec K, Pilutin A, Tarnowski M, Jakubczyk K, Żwierełło W, Skórka-Majewicz M, Chlubek D, Gutowska I (2020) Changes in gene and protein expression of metalloproteinase-2 and -9 and their inhibitors TIMP2 and TIMP3 in different parts of fluoride-exposed rat brain. Int J Mol Sci 22:391. https://doi.org/10.3390/ijms22010391
Li X, Zhang J, Niu R, Manthari RK, Yang K, Wang J (2019) Effect of fluoride exposure on anxiety- and depression-like behavior in mouse. Chemosphere 215:454–460. https://doi.org/10.1016/j.chemosphere.2018.10.070
Niu R, Chen H, Manthari RK, Sun Z, Wang J, Zhang J, Wang J (2018) Effects of fluoride on synapse morphology and myelin damage in mouse hippocampus. Chemosphere 194:628–633. https://doi.org/10.1016/j.chemosphere.2017.12.027
Wei N, Dong YT, Deng J, Wang Y, Qi XL, Yu WF, Xiao Y, Zhou JJ, Guan ZZ (2018) Changed expressions of N-methyl-d-aspartate receptors in the brains of rats and primary neurons exposed to high level of fluoride. J Trace Elem Med Biol 45:31–40. https://doi.org/10.1016/j.jtemb.2017.09.020
Nadei OV, Khvorova IA, Agalakova NI (2020) Cognitive decline of rats with chronic fluorosis is associated with alterations in hippocampal calpain signaling. Biol Trace Elem Res 197:495–506. https://doi.org/10.1007/s12011-019-01993-z
Bustelo M, Bruno MA, Loidl CF, Rey-Funes M, Steinbusch HWM, Gavilanes AWD, van den Hove DLA (2020) Statistical differences resulting from selection of stable reference genes after hypoxia and hypothermia in the neonatal rat brain. PLoS One 15:e0233387. https://doi.org/10.1371/journal.pone.0233387
Sundaram VK, Sampathkumar NK, Massaad C, Grenier J (2019) Optimal use of statistical methods to validate reference gene stability in longitudinal studies. PLoS One 14:e0219440. https://doi.org/10.1371/journal.pone.0219440
Schwarz AP, Kovalenko AA, Malygina DA, Postnikova TY, Zubareva OE, Zaitsev AV (2020) Reference gene validation in the brain regions of young rats after pentylenetetrazole-induced seizures. Biomedicines 8:239. https://doi.org/10.3390/biomedicines8080239
Nutrient requirements of laboratory animals: fourth revised edition, 1995. National Academy Press, Washington
Lin W, Burks CA, Hansen DR, Kinnamon SC, Gilbertson TA (2004) Taste receptor cells express pH-sensitive leak K+ channels. J Neurophysiol 92:2909–2919. https://doi.org/10.1152/jn.01198.2003
Kolson DR, Wan J, Wu J, Dehoff M, Brandebura AN, Qian J, Mathers PH, Spirou GA (2016) Temporal patterns of gene expression during calyx of held development. Dev Neurobiol 76:166–189. https://doi.org/10.1002/dneu.22306
Langnaese K, John R, Schweizer H, Ebmeyer U, Keilhoff G (2008) Selection of reference genes for quantitative real-time PCR in a rat asphyxial cardiac arrest model. BMC Mol Biol 9:53. https://doi.org/10.1186/1471-2199-9-53
Meng XW, Jin XH, Wei X, Wang LN, Yang JP, Ji FH (2019) Low-affinity neurotrophin receptor p75 of brain-derived neurotrophic factor contributes to cancer-induced bone pain by upregulating mTOR signaling. Exp Ther Med 18:4379–4387. https://doi.org/10.3892/etm.2019.8097
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 25(4):402–408
Bu J, Zhao J, Liu M (2016) Expression stabilities of candidate reference genes for RT-qPCR in Chinese Jujube (Ziziphus jujuba mill.) under a variety of conditions. PloS One 11:e0154212. https://doi.org/10.1371/journal.pone.0154212
Wang H, Zhang X, Liu Q, Liu X, Ding S (2017) Selection and evaluation of new reference genes for RT-qPCR analysis in Epinephelus akaara based on transcriptome data. PLoS One 12:e0171646. https://doi.org/10.1371/journal.pone.0171646
Zhou D, Li G, Moore KB 3rd, Xie Y, Peterson KA, Schaefer HF 3rd (2018) Noncovalent Interactions between molecular hydrogen and the alkali fluorides: H-H···F-M (M = Li, Na, K, Rb, Cs). High level theoretical predictions and SAPT analysis. J Chem Theory Comput 14:5118–5127. https://doi.org/10.1021/acs.jctc.8b00461
Pupo G, Gouverneur V (2022) Hydrogen bonding phase-transfer catalysis with alkali metal fluorides and beyond. J Am Chem Soc 144:5200–5213. https://doi.org/10.1021/jacs.2c00190
Qin J, Chai G, Brewer JM, Lovelace LL, Lebioda L (2006) Fluoride inhibition of enolase: crystal structure and thermodynamics. Biochemistry 45:793–800. https://doi.org/10.1021/bi051558s
Agalakova NI, Gusev GP (2012) Molecular mechanisms of cytotoxicity and apoptosis induced by inorganic fluoride. ISRN Cell Biol 403835. https://doi.org/10.5402/2012/403835
Ren C, Li HH, Zhang CY, Song XC (2022) Effects of chronic fluorosis on the brain. Ecotoxicol Environ Saf 244:114021. https://doi.org/10.1016/j.ecoenv.2022.114021
Ottappilakkil H, Babu S, Balasubramanian S, Manoharan S, Perumal E (2022) Fluoride induced neurobehavioral impairments in experimental animals: a brief review. Biol Trace Elem Res. https://doi.org/10.1007/s12011-022-03242-2
Pernot F, Dorandeu F, Beaup C, Peinnequin A (2010) Selection of reference genes for real-time quantitative reverse transcription-polymerase chain reaction in hippocampal structure in a murine model of temporal lobe epilepsy with focal seizures. J Neurosci Res 88:1000–1008. https://doi.org/10.1002/jnr.22282
Sadangi C, Rosenow F, Norwood BA (2017) Validation of reference genes for quantitative gene expression analysis in experimental epilepsy. J Neurosci Res 95:2357–2366. https://doi.org/10.1002/jnr.24089
Zhou J, Zhang X, Ren J, Wang P, Zhang J, Wei Z, Tian Y (2016) Validation of preference genes for quantitative real-time PCR in valproic acid rat models of autism. Mol Biol Rep 43:837–847. https://doi.org/10.1007/s11033-016-4015-x
Swijsen A, Nelissen K, Janssen D, Rigo JM, Hoogland G (2012) Validation of reference genes for quantitative real-time PCR studies in the dentate gyrus after experimental febrile seizures. BMC Res Notes 5:685. https://doi.org/10.1186/1756-0500-5-685
Zhang Q, Zhang M, Li J, Xiao H, Wu D, Guo Q, Zhang Y, Wang H, Li S, Liao S (2018) Selection and validation of reference genes for RT-PCR expression analysis of candidate genes involved in morphine-induced conditioned place preference mice. J Mol Neurosci 66:587–594. https://doi.org/10.1007/s12031-018-1198-8
Ran LY, Xiang J, Zeng XX, He WW, Dong YT, Yu WF, Qi XL, Xiao Y, Cao K, Zou J, Guan ZZ (2023) The influence of NQO2 on the dysfunctional autophagy and oxidative stress induced in the hippocampus of rats and in SH-SY5Y cells by fluoride. CNS Neurosci Ther. https://doi.org/10.1111/cns.14090
Yıldız MO, Çelik H, Caglayan C, Kandemir FM, Gür C, Bayav İ, Genç A, Kandemir Ö (2022) Neuromodulatory effects of hesperidin against sodium fluoride-induced neurotoxicity in rats: Involvement of neuroinflammation, endoplasmic reticulum stress, apoptosis and autophagy. Neurotoxicology 90:197–204. https://doi.org/10.1016/j.neuro.2022.04.002
Alieva AK, Filatova EV, Rudenok MM, Slominsky PA, Shadrina MI (2021) Housekeeping genes for Parkinson's disease in humans and mice. Cells 10:2252. https://doi.org/10.3390/cells10092252
de Araújo MA, Marques TE, Taniele-Silva J, Souza FM, de Andrade TG, Garcia-Cairasco N, Paçó-Larson ML, Gitaí DL (2014) Identification of endogenous reference genes for the analysis of microRNA expression in the hippocampus of the pilocarpine-induced model of mesial temporal lobe epilepsy. PLoS One 9:e100529. https://doi.org/10.1371/journal.pone.0100529
Funding
The work was performed within the state assignment of Federal Agency of Scientific Organizations of Russia (theme no. 075-01052-22-00). The experiments were carried out using the equipment of the Center for Joint Usage of the Sechenov Institute of Evolutionary Physiology and Biochemistry.
Author information
Authors and Affiliations
Contributions
Olga Nadei: methodology, validation, investigation, formal analysis, writing — review and editing; Natalia Agalakova: conceptualization, resources, writing – original draft, writing — review and editing.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nadei, O.V., Agalakova, N.I. Optimal Reference Genes for RT-qPCR Experiments in Hippocampus and Cortex of Rats Chronically Exposed to Excessive Fluoride. Biol Trace Elem Res 202, 199–209 (2024). https://doi.org/10.1007/s12011-023-03646-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-023-03646-8