Abstract
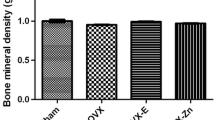
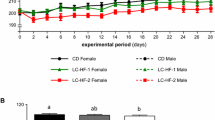
Zinc (Zn) deficiency impairs bone growth. However, the precise skeletal effects of varying levels of Zn deficiency and response to subsequent Zn repletion on the growing skeleton are incompletely understood. To address this gap in knowledge, we investigated the effects of dietary Zn ((severe deficiency (< 0.5 mg Zn/kg diet) and short-term Zn repletion (30 mg/kg diet), marginal deficiency (6 mg Zn/kg diet)) on bone mass, density, and cortical and cancellous bone microarchitecture in growing male Sprague Dawley rats. Marginal Zn intake for 42 days had no effect on bone mass or cortical and cancellous bone microarchitecture. Twenty-one days of severe Zn deficiency lowered serum osteocalcin and C terminal telopeptide of type I collagen (CTX-1), decreased tibial bone mineral content and density, and lowered cross-sectional volume, cortical volume, and cortical thickness in tibial diaphysis as compared to both Zn-adequate (30 mg/kg diet) and pair-fed controls. Severe Zn deficiency similarly lowered cancellous bone volume in proximal tibial metaphysis. Zn repletion (10 days) accelerated weight gain, indicative of catch-up growth, normalized CTX-1 and osteocalcin, but did not normalize bone mass (unadjusted and adjusted for body weight) or cortical and cancellous bone microarchitecture. In summary, severe but not marginal Zn deficiency in rapidly growing rats impaired acquisition of cortical and cancellous bone, resulting in abnormalities in bone microarchitecture. Zn repletion accelerated weight gain compared to Zn-adequate controls but absence of a compensatory increase in serum osteocalcin or bone mass suggests Zn repletion may be insufficient to fully counteract the detrimental effects of prior Zn deficiency on skeletal growth.







Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article. The datasets will also be made available to anyone upon request.
References
Ganss B, Jheon A (2004) Zinc finger transcription factors in skeletal development. Crit Rev Oral Biol Med 15(5):282–297. https://doi.org/10.1177/154411130401500504
King JC (2011) Zinc: an essential but elusive nutrient. Am J Clin Nutr 94(2):679S-S684. https://doi.org/10.3945/ajcn.110.005744
Krezel A, Maret W (2016) The biological inorganic chemistry of zinc ions. Arch Biochem Biophys 611:3–19. https://doi.org/10.1016/j.abb.2016.04.010
Livingstone C (2015) Zinc: physiology, deficiency, and parenteral nutrition. Nutr Clin Pract 30(3):371–382. https://doi.org/10.1177/0884533615570376
Hambidge KM, Krebs NF (2007) Zinc deficiency: a special challenge. J Nutr 137(4):1101–1105. https://doi.org/10.1093/jn/137.4.1101
Wessells KR, Brown KH (2012) Estimating the global prevalence of zinc deficiency: results based on zinc availability in national food supplies and the prevalence of stunting. PLoS ONE 7(11):e50568. https://doi.org/10.1371/journal.pone.0050568
Maxfield L, Shukla S, Crane JS (2022) Zinc Deficiency, in StatPearls. Treasure Island (FL)
Doherty CP, Crofton PM, Sarkar MA, Shakur MS, Wade JC, Kelnar CJ, Elmlinger MW, Ranke MB, Cutting WA (2002) Malnutrition, zinc supplementation and catch-up growth: changes in insulin-like growth factor I, its binding proteins, bone formation and collagen turnover. Clin Endocrinol (Oxf) 57(3):391–9. https://doi.org/10.1046/j.1365-2265.2002.01622.x
Rossi L, Migliaccio S, Corsi A, Marzia M, Bianco P, Teti A, Gambelli L, Cianfarani S, Paoletti F, Branca F (2001) Reduced growth and skeletal changes in zinc-deficient growing rats are due to impaired growth plate activity and inanition. J Nutr 131(4):1142–1146. https://doi.org/10.1093/jn/131.4.1142
Bruno RS, Song Y, Leonard SW, Mustacich DJ, Taylor AW, Traber MG, Ho E (2007) Dietary zinc restriction in rats alters antioxidant status and increases plasma F2 isoprostanes. J Nutr Biochem 18(8):509–518. https://doi.org/10.1016/j.jnutbio.2006.09.001
Hosea HJ, Taylor CG, Wood T, Mollard R, Weiler HA (2004) Zinc-deficient rats have more limited bone recovery during repletion than diet-restricted rats. Exp Biol Med (Maywood) 229(4):303–311. https://doi.org/10.1177/153537020422900404
Ovesen J, Moller-Madsen B, Thomsen JS, Danscher G, Mosekilde L (2001) The positive effects of zinc on skeletal strength in growing rats. Bone 29(6):565–570. https://doi.org/10.1016/s8756-3282(01)00616-0
Song Y, Leonard SW, Traber MG, Ho E (2009) Zinc deficiency affects DNA damage, oxidative stress, antioxidant defenses, and DNA repair in rats. J Nutr 139(9):1626–1631. https://doi.org/10.3945/jn.109.106369
Zhang Y, Cheng Y, Hong Y, Li S (2003) Zinc deficiency on pathological changes of femur epiphyseal growth plate in rats. Wei Sheng Yan Jiu 32(1):16–19
Salgueiro MJ, Torti H, Meseri E, Weill R, Orlandini J, Urriza R, Zubillaga M, Janjetic M, Barrado A, Boccio J (2006) Dietary zinc effects on zinc, calcium, and magnesium content in bones of growing rats. Biol Trace Elem Res 110(1):73–78. https://doi.org/10.1385/BTER:110:1:73
Windisch W, Wher U, Rambeck W, Erben R (2002) Effect of Zn deficiency and subsequent Zn repletion on bone mineral composition and markers of bone tissue metabolism in 65Zn-labelled, young-adult rats. J Anim Physiol Anim Nutr (Berl) 86(7–8):214–221. https://doi.org/10.1046/j.1439-0396.2002.00377.x
Eberle J, Schmidmayer S, Erben RG, Stangassinger M, Roth HP (1999) Skeletal effects of zinc deficiency in growing rats. J Trace Elem Med Biol 13(1–2):21–26. https://doi.org/10.1016/S0946-672X(99)80019-4
Maki K, Nishioka T, Nishida I, Ushijima S, Kimura M (2002) Effect of zinc on rat mandibles during growth. Am J Orthod Dentofacial Orthop 122(4):410–413. https://doi.org/10.1067/mod.2002.126152
Scrimgeour AG, Stahl CH, McClung JP, Marchitelli LJ, Young AJ (2007) Moderate zinc deficiency negatively affects biomechanical properties of rat tibiae independently of body composition. J Nutr Biochem 18(12):813–819. https://doi.org/10.1016/j.jnutbio.2006.12.018
Erben RG, Lausmann K, Roschger P, Schuler C, Skalicky M, Klaushofer K, Windisch W (2009) Long-term marginal zinc supply is not detrimental to the skeleton of aged female rats. J Nutr 139(4):703–709. https://doi.org/10.3945/jn.108.099077
Golub MS, Gershwin ME, Hurley LS, Saito WY, Hendrickx AG (1984) Studies of marginal zinc deprivation in rhesus monkeys. IV. Growth of infants in the first year. Am J Clin Nutr 40(6):1192–202. https://doi.org/10.1093/ajcn/40.6.1192
Sandstead HH (1995) Is zinc deficiency a public health problem? Nutrition 11(1 Suppl):87–92
Boersma B, Wit JM (1997) Catch-up growth. Endocr Rev 18(5):646–661. https://doi.org/10.1210/edrv.18.5.0313
Wit JM, Boersma B (2002) Catch-up growth: definition, mechanisms, and models. J Pediatr Endocrinol Metab 15:1229–1241
Kim JT, Baek SH, Lee SH, Park EK, Kim EC, Kwun IS, Shin HI (2009) Zinc-deficient diet decreases fetal long bone growth through decreased bone matrix formation in mice. J Med Food 12(1):118–123. https://doi.org/10.1089/jmf.2007.0647
Lowe NM, Woodhouse LR, Wee J, King JC (1999) Short-term zinc kinetics in pregnant rats fed marginal zinc diets. J Nutr 129(5):1020–1025. https://doi.org/10.1093/jn/129.5.1020
Sun JY, Jing MY, Wang JF, Zi NT, Fu LJ, Lu MQ, Pan L (2006) Effect of zinc on biochemical parameters and changes in related gene expression assessed by cDNA microarrays in pituitary of growing rats. Nutrition 22(2):187–196. https://doi.org/10.1016/j.nut.2005.07.007
Wong CP, Magnusson KR, Sharpton TJ, Ho E (2021) Effects of zinc status on age-related T cell dysfunction and chronic inflammation. Biometals 34(2):291–301. https://doi.org/10.1007/s10534-020-00279-5
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate - a practical and powerful approach to multiple testing. J R Stat Soc Ser B-Stat Methodol 57(1):289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x
Iwaniec UT, Turner RT (2016) Influence of body weight on bone mass, architecture and turnover. J Endocrinol 230(3):R115–R130. https://doi.org/10.1530/JOE-16-0089
Schmidt IU, Dobnig H, Turner RT (1995) Intermittent parathyroid hormone treatment increases osteoblast number, steady state messenger ribonucleic acid levels for osteocalcin, and bone formation in tibial metaphysis of hypophysectomized female rats. Endocrinology 136(11):5127–5134. https://doi.org/10.1210/endo.136.11.7588250
Kidder LS, Schmidt IU, Evans GL, Turner RT (1997) Effects of growth hormone and low dose estrogen on bone growth and turnover in long bones of hypophysectomized rats. Calcif Tissue Int 61(4):327–335. https://doi.org/10.1007/s002239900343
Turner RT, Rosen CJ, Iwaniec UT (2010) Effects of alcohol on skeletal response to growth hormone in hypophysectomized rats. Bone 46(3):806–812. https://doi.org/10.1016/j.bone.2009.10.027
Golub MS, Gershwin ME, Hurley LS, Hendrickx AG (1988) Studies of marginal zinc deprivation in rhesus monkeys. VIII. Effects in early adolescence. Am J Clin Nutr 47(6):1046–51. https://doi.org/10.1093/ajcn/47.6.1046
Wang X, Fosmire GJ, Gay CV, Leach RM Jr (2002) Short-term zinc deficiency inhibits chondrocyte proliferation and induces cell apoptosis in the epiphyseal growth plate of young chickens. J Nutr 132(4):665–673. https://doi.org/10.1093/jn/132.4.665
Leek JC, Keen CL, Vogler JB, Golub MS, Hurley LS, Hendrickx AG, Gershwin ME (1988) Long-term marginal zinc deprivation in rhesus monkeys. IV. Effects on skeletal growth and mineralization. Am J Clin Nutr 47(5):889–95. https://doi.org/10.1093/ajcn/47.5.889
Nakano M, Nakamura Y, Miyazaki A, Takahashi J (2021) Zinc pharmacotherapy for elderly osteoporotic patients with zinc deficiency in a clinical setting. Nutrients 13(6):1814. https://doi.org/10.3390/nu13061814
Relea P, Revilla M, Ripoll E, Arribas I, Villa LF, Rico H (1995) Zinc, biochemical markers of nutrition, and type I osteoporosis. Age Ageing 24(4):303–307. https://doi.org/10.1093/ageing/24.4.303
Ho E, Ames BN (2002) Low intracellular zinc induces oxidative DNA damage, disrupts p53, NFkappa B, and AP1 DNA binding, and affects DNA repair in a rat glioma cell line. Proc Natl Acad Sci U S A 99(26):16770–16775. https://doi.org/10.1073/pnas.222679399
Sun X, Zhou X, Du L, Liu W, Liu Y, Hudson LG, Liu KJ (2014) Arsenite binding-induced zinc loss from PARP-1 is equivalent to zinc deficiency in reducing PARP-1 activity, leading to inhibition of DNA repair. Toxicol Appl Pharmacol 274(2):313–318. https://doi.org/10.1016/j.taap.2013.11.010
Skrovanek S, DiGuilio K, Bailey R, Huntington W, Urbas R, Mayilvaganan B, Mercogliano G, Mullin JM (2014) Zinc and gastrointestinal disease. World J Gastrointest Pathophysiol 5(4):496–513. https://doi.org/10.4291/wjgp.v5.i4.496
Huang T, Yan G, Guan M (2020) Zinc homeostasis in bone: zinc transporters and bone diseases. Int J Mol Sci 21(4):1236. https://doi.org/10.3390/ijms21041236
Jovanovic M, Schmidt FN, Guterman-Ram G, Khayyeri H, Hiram-Bab S, Orenbuch A, Katchkovsky S, Aflalo A, Isaksson H, Busse B, Jahn K, Levaot N (2018) Perturbed bone composition and integrity with disorganized osteoblast function in zinc receptor/Gpr39-deficient mice. FASEB J 32(5):2507–2518. https://doi.org/10.1096/fj.201700661RR
Prasad AS, Halsted JA, Nadimi M (1961) Syndrome of iron deficiency anemia, hepatosplenomegaly, hypogonadism, dwarfism and geophagia. Am J Med 31:532–546. https://doi.org/10.1016/0002-9343(61)90137-1
Salgueiro J, Leonardi N, Zubillaga M, Weill R, Goldman C, Calmanovici G, Barrado A, Sarrasague MM, Boccio J (2005) Normal growth rate in rats is recovered after a period of zinc deficiency by restoration of zinc supply by means of a zinc-fortified Petit Suisse cheese. Biol Trace Elem Res 104(3):269–273. https://doi.org/10.1385/BTER:104:3:269
Sandstead HH, Strobel DA, Logan GM Jr, Marks EO, Jacob RA (1978) Zinc deficiency in pregnant rhesus monkeys: effects on behavior of infants. Am J Clin Nutr 31(5):844–849. https://doi.org/10.1093/ajcn/31.5.844
Jayalakshmi S, Platel K (2016) Supplemental levels of iron and calcium interfere with repletion of zinc status in zinc-deficient animals. Food Funct 7(5):2288–2293. https://doi.org/10.1039/c6fo00134c
Hunt CD, Halas ES, Eberhardt MJ (1988) Long-term effects of lactational zinc deficiency on bone mineral composition in rats fed a commercially modified Luecke diet. Biol Trace Elem Res 16(2):97–113. https://doi.org/10.1007/BF02797095
Park JH, Grandjean CJ, Antonson DL, Vanderhoof JA (1986) Effects of isolated zinc deficiency on the composition of skeletal muscle, liver and bone during growth in rats. J Nutr 116(4):610–617. https://doi.org/10.1093/jn/116.4.610
Roth HP, Kirchgessner M (1977) Content of zinc, copper, iron, manganese and calcium in bone and liver of rats during zinc depletion and repletion. Zentralbl Veterinarmed A 24(3):177–188
Golub MS, Takeuchi PT, Keen CL, Gershwin ME, Hendrickx AG, Lonnerdal B (1994) Modulation of behavioral performance of prepubertal monkeys by moderate dietary zinc deprivation. Am J Clin Nutr 60(2):238–243. https://doi.org/10.1093/ajcn/60.2.238
Golub MS, Keen CL, Gershwin ME (2000) Moderate zinc-iron deprivation influences behavior but not growth in adolescent rhesus monkeys. J Nutr 130(2S Suppl):354S-357S. https://doi.org/10.1093/jn/130.2.354S
Fong L, Tan K, Tran C, Cool J, Scherer MA, Elovaris R, Coyle P, Foster BK, Rofe AM, Xian CJ (2009) Interaction of dietary zinc and intracellular binding protein metallothionein in postnatal bone growth. Bone 44(6):1151–1162. https://doi.org/10.1016/j.bone.2009.02.011
Leek JC, Vogler JB, Gershwin ME, Golub MS, Hurley LS, Hendrickx AG (1984) Studies of marginal zinc deprivation in rhesus monkeys. V. Fetal and infant skeletal effects. Am J Clin Nutr 40(6):1203–12. https://doi.org/10.1093/ajcn/40.6.1203
Golub MS, Keen CL, Gershwin ME, Styne DM, Takeuchi PT, Ontell F, Walter RM, Hendrickx AG (1996) Adolescent growth and maturation in zinc-deprived rhesus monkeys. Am J Clin Nutr 64(3):274–282. https://doi.org/10.1093/ajcn/64.3.274
Jamieson JA, Taylor CG, Weiler HA (2006) Marginal zinc deficiency exacerbates bone lead accumulation and high dietary zinc attenuates lead accumulation at the expense of bone density in growing rats. Toxicol Sci 92(1):286–294. https://doi.org/10.1093/toxsci/kfj201
Zhou JR, Canar MM, Erdman JW Jr (1993) Bone zinc is poorly released in young, growing rats fed marginally zinc-restricted diet. J Nutr 123(8):1383–1388. https://doi.org/10.1093/jn/123.8.1383
Sibonga JD, Zhang M, Evans GL, Westerlind KC, Cavolina JM, Morey-Holton E, Turner RT (2000) Effects of spaceflight and simulated weightlessness on longitudinal bone growth. Bone 27(4):535–540. https://doi.org/10.1016/s8756-3282(00)00352-5
Funding
This work was supported by the National Institute of Food and Agriculture—Agricultural Experimental Station Multi-state W4002 and Oregon Agricultural Experiment Station (OR00735).
Author information
Authors and Affiliations
Contributions
Study conception and design was performed by Emily Ho. Material preparation and data collection was completed by Kenneth Philbrick, Dawn Olson, Carmen Wong, and Yang Song. Data analysis was completed by Adam Branscum and Laura Beaver and figure preparation was done by Laura Beaver. The first draft of the manuscript was written by Laura Beaver and all authors commented on previous versions of the manuscript. Urszula Iwaniec takes responsibility for the integrity of the data. All authors read and approved the final manuscript version.
Corresponding author
Ethics declarations
Ethics Approval
All procedures involving animals were in compliance with the PHS policy on humane care and use of animals in research (2015), and ethical approval was granted by the institutional animal care and use committee of Oregon State University (Corvallis, Oregon, USA).
Competing Interests
The authors declare no competing interests.
Disclaimer
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Food and Agriculture.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Beaver, L.M., Song, Y., Philbrick, K.A. et al. Severe Zinc Deficiency Impairs Accrual of Bone in Rapidly Growing Rats That Is Partially Corrected Following Short-term Zinc Repletion. Biol Trace Elem Res 201, 3834–3849 (2023). https://doi.org/10.1007/s12011-022-03456-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-022-03456-4