Abstract
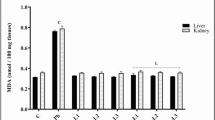
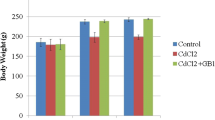
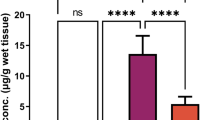
The current study investigates the potential alleviating activity of bulbs (B) and leaves (L) of Allium triquetrum aqueous extract (ATE) on repro-toxicity induced by lead acetate (Pb) in male Wistar rats administrated orally for 3 consecutive weeks. Eighteen groups of rats were divided into the control, Pb (500 mg/kg body weight/day), positive controls of B and L (2 g, 3 g, 4 g, 6 g/kg body weight/day), in addition to four mixtures of each of Pb-B (Pb-B1, Pb-B2, Pb-B3, Pb-B4) and Pb-L (Pb-L1, Pb-L2, Pb-L3, Pb-L4). The two extracts were subjected to phytochemical screening and HPLC analysis. Sperm characteristics were evaluated by CASA system, as well as the serum testosterone, testicular and epididymal levels of glutathione (GSH), glutathione peroxidase (GPx), and malondialdehyde (MDA). The phytochemical screening proved that bulbs’ and leaves’ extracts were rich in various compounds and the HPLC showed that leaves contain more tannins. Results revealed a significant decrease in the testicular and in the epididymal weights, sperm concentration, motility, testosterone, velocity, vitality, round cells, GSH, and GPx levels in the Pb-intoxicated rats compared to the control, with the exception of MDA concentration that was significantly increased. However, the co-administration of garlic extracts (Pb-B and Pb-L) exhibited a significant increase in all mentioned markers, except for the MDA level which was reduced. Likewise, Pb caused histological injuries in the testicular seminiferous of rats, while the co-administration of wild garlic has reduced such effect, especially in the higher doses. Both extracts of Pb-B and Pb-L have attenuated Pb toxicity in a dose-dependent manner. In conclusion, aqueous extracts of A. triquetrum have the potential to reduce Pb testicular injuries by boosting sperm characteristics and ameliorating oxidative stress markers.













Similar content being viewed by others
References
Otmani I, Abdennour C, Dridi A, Kahalerras L, Halima-Salem A (2019) Characteristics of the bitter and sweet honey from Algeria Mediterranean coast. Vet World 12(4):551–557. https://doi.org/10.14202/vetworld.2019.551-557
Lanzotti V, Romano A, Lanzuise S, Bonanomi S, Scala F (2012) Antifungal saponins from bulbs of white onion, Allium cepa L. Phytochemistry 74:133–139. https://doi.org/10.1016/j.phytochem.2011.11.008
Chowdhury R, Dutta A, Chaudhuri SR, Sharma N, Giri AK, Chaudhuri K (2008) In vitro and in vivo reduction of sodium arsenite induced toxicity by aqueous garlic extract. Food Chem Toxicol 46:740–751. https://doi.org/10.1016/j.fct.2007.09.108
Weiss N, Papatheodorou L, Morihara N, Hilge R, Ide N (2013) Aged garlic extract restores nitric oxide bioavailability in cultured human endothelial cells even under conditions of homocysteine elevation. J Ethnopharmacol 145:162–167. https://doi.org/10.1016/j.jep.2012.10.045
Rivlin RS (2001) Historical perspective on the use of garlic. J Nutr 131:951S-954S. https://doi.org/10.1093/jn/131.3.951S
Vijayakumar S, Malaikozhundan B, Saravanakumar K, Durán-Lara EF, Wang MH, Vaseeharan B (2019) Garlic clove extract assisted silver nanoparticle - antibacterial, antibiofilm, antihelminthic, anti-inflammatory, anticancer and ecotoxicity assessment. J PhotochemPhotobiol B 198:111558. https://doi.org/10.1016/j.jphotobiol.2019.111558
Amagase H (2006) Clarifying the real bioactive constituents of garlic. In: Rivlin R, Budoff M, Amagase H (eds) Symposium of significance of garlic and its constituents in cancer and cardiovascular disease; 9–11 April 2005. Georgetown University, Washington, DC. J Nutr 136:716S–725S
Morakinyo AO, Oloyo AK, Raji Y, Adegoke OA (2008) Effects of aqueous extract of garlic (Allium sativum) on testicular functions in the rats. Niger J Health Biomed Sci 7:26–30. https://doi.org/10.4314/njhbs.v7i2.11672
Josling P (2005) ALLICIN: The heart of garlic nature’s aid to healing the human body. Chicago
Yang JS, Kok LF, Lin YH, Kuo TC, Yang JL, Lin CC, Chen GW, Huang WW, Ho HC, Chung JG (2006) Diallyl disulfide inhibits WEHI-3 leukemia cells in vivo. Anticancer Res 26:219–225 (PMID: 16475702)
Lanzotti V, Scala F, Bonanomi G (2014) Compounds from Allium species with cytotoxic and antimicrobial activity. Phytochem Rev 13:769–791. https://doi.org/10.1007/s11101-014-9366-0
Drif F, Abdennour C, Ciğerci İH, Muddassir AM, Mansouri O, Messarah M (2019) Preliminary assessment of stress and genotoxicity biomarkers in bivalve molluscs from the Gulf of Annaba, Algeria. Bull Environ ContamToxicol 102:555–559. https://doi.org/10.1007/s00128-019-02583-4
Senapati SK, Dey S, Dwived SK, Swarup D (2001) Effect of garlic (Allium sativum L.) extract on tissue lead level in rats. J Ethnopharmacol 76:229–232. https://doi.org/10.1016/s0378-8741(01)00237-9
Andjelkovic M, Djordjevic AB, Antonijevic E, Antonijevic B, Stanic M, Kotur-Stevuljevic J, Spasojevic-Kalimanovska V, Jovanovic M, Boricic N, Wallace D, Bulat Z (2019) Toxic effect of acute cadmium and lead exposure in rat blood, liver, and kidney. Int J Environ Res Public Health 16:274. https://doi.org/10.3390/ijerph16020274
El-Magd MA, Kahilo KA, Nasr NE, Kamal T, Shukry M, Saleh AA (2016) A potential mechanism associated with lead-induced testicular toxicity in rats. Andrologia 49:e12750. https://doi.org/10.1111/and.12750
Allouche L, Hamadouche M, Touabti A (2009) Chronic effects of low lead levels on sperm quality, gonadotropins and testosterone in albino rats. Exp Toxicol Pathol 61:503–510. https://doi.org/10.1016/j.etp.2008.12.003
Berredjem R, Mansouri O, Abdennour C, Boulakoud MK, Khelili K (2014) What is the benefit of garlic supplementation to Pb intoxicated rats? Adv Environ Biol 8:268–275
Chowdhury AR (2009) Recent advances in heavy metals induced effect on male reproductive function-a retrospective. Al Ameen J Med Sci 2:37–42
Sainath SB, Meena R, Supriya C, Reddy KP, Reddy PS (2011) Protective role of Centella asiatica on lead-induced oxidative stress and suppressed reproductive health in male rats. Environ Toxicol Pharmacol 32:146–154. https://doi.org/10.1016/j.etap.2011.04.005
Iavicoli I, Fontana L, Bergamaschi A (2009) The effects of metals as endocrine disruptors. J Toxicol Environ Health Part B 12:206–223. https://doi.org/10.1080/10937400902902062
Liguori G, Pelagalli A, Assisi L, Squillacioti C, Costagliola A, Mirabella N (2018) Effects of orexins on 17β-estradiol synthesis and P450 aromatase modulation in the testis of alpaca (Vicugna pacos). Anim Reprod Sci 192:313–320. https://doi.org/10.1016/j.anireprosci.2018.03.032
Elgawish RAR, Abdelrazek HMA (2014) Effects of lead acetate on testicular function and caspase-3 expression with respect to the protective effect of cinnamon in albino rats. Toxicol Rep 1:795–801. https://doi.org/10.1016/j.toxrep.2014.10.010
Limón-Pacheco J, Gonsebatt ME (2009) The role of antioxidants and antioxidant-related enzymes in protective responses to environmentally induced oxidative stress. Mutat Res 674:137–147. https://doi.org/10.1016/j.mrgentox.2008.09.015
He Y, Zou Q, Chen H, Weng S, Luo T, Zeng W (2016) Lead inhibits human sperm functions by reducing the levels of intracellular calcium, cAMP, and tyrosine phosphorylation. Tohoku J Exp Med 238:295–303. https://doi.org/10.1620/tjem.238.295
Dorostghoal M, Seyyednejad SM, Jabari A (2013) Protective effects of Fumaria parviflora L on lead-induced testicular toxicity in male rats. Andrologia 46:437–446. https://doi.org/10.1111/and.12100
Corea G, Fattorusso E, Lanzotti V (2003) Saponins and flavonoids of Allium triquetrum. J Nat Prod 66:1405–1411. https://doi.org/10.1021/np030226q
Drevius LO, Eriksson H (1966) Osmotic swelling of mammalian spermatozoa. Exp Cell Res 42:136–156. https://doi.org/10.1016/0014-4827(66)90327-2
Jayendran RS, Van Der Ven HH, Zaneveld LJ (1992) The hypo-osmotic swelling test: an update. Arch Androl 29:105–116. https://doi.org/10.3109/01485019208987714
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1006/abio.1976.9999
Weckbecker G, Cory JG (1988) Ribonucleotide reductase activity and growth of glutathione-depleted mouse leukemia L1210 cells in vitro. Cancer Lett 40:257–264. https://doi.org/10.1016/0304-3835(88)90084-5
Flohé L, Günzler WA (1984) [12] Assays of glutathione peroxidase. Methods Enzymol 105:114–120. Academic Press. https://doi.org/10.1016/S0076-6879(84)05015-1
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Godínez-Solís Y, Solís-Heredia MJ, Roa-Espitia A, Parra-Forero Y, Hernández-González EO, Hernández-Ochoa I, Quintanilla-Vega B (2019) Low concentrations of lead decrease the sperm fertilization ability by altering the acrosome reaction in mice. Toxicol Appl Pharmacol 380:114694. https://doi.org/10.1016/j.taap.2019.114694
Fihri AF, Al-Waili NS, El-Haskoury R, Bakour M, Amarti A, Ansari MJ, Lyoussi B (2016) Protective effect of Morocco carob honey against lead-induced anemia and hepato-renal toxicity. Cell PhysiolBiochem 39:115–122. https://doi.org/10.1159/000445610
Hamadouche NA, Sadi N, Kharoubi O, Slimani M, Aoues A (2013) The protective effect of vitamin E against genotoxicity of lead acetate intraperitoneal administration in male rat. Not Sci Biol 5: 412–419. https://doi.org/10.15835/nsb549125
Wang L, Xun P, Zhao Y, Wang X, Qian L, Chen F (2008) Effects of lead exposure on sperm concentrations and testes weight in male rats: a meta-regression analysis. J Toxicol Environ Health A 71:454–463. https://doi.org/10.1080/15287390701839331
Flora SJS, Agrawal S (2017) Arsenic, cadmium, and lead. In: Gupta RC (ed) Reproductive and developmental toxicology. Academic Press, Elsevier, USA, pp 537–566. https://doi.org/10.1016/B978-0-12-804239-7.00031-7
Ouarda M (2011) Abdennour C (2016) Evaluation of the therapeutic efficiency of raw garlic on reproduction of domestic rabbits under lead induced toxicity. Ann Biol Res 2:389–393
Shubina OS, Dudenkova NA (2016) The effect of lead on the process of spermatogenesis in sex glands of male albino rats. Vet World 9:1129–1134. https://doi.org/10.14202/vetworld.2016.1129-1134
Castellini C, Mourvaki E, Sartini B, Cardinali R, Moretti E, Collodelb G, Fortanerc S, Sabbionic E, Renieri T (2009) In vitro toxic effects of metal compounds on kinetic traits and ultra structure of rabbit spermatozoa. ReprodToxicol 27:46–54. https://doi.org/10.1016/j.reprotox.2008.12.003
Corpas I, Castillo M, Marquina D, Benito MJ (2002) Lead intoxication in gestational and lactation periods alters the development of male reproductive organs. Ecotox Environ Safe 53:259–266. https://doi.org/10.1006/eesa.2002.2230
He X, Wu J, Yuan L, Lin F, Yi J, Li J, Yuan H, Shi J, Yuan T, Zhang S, Fan Y (2017) Lead induces apoptosis in mouse TM3 Leydig cells through the Fas/FasL death receptor pathway. Environ Toxicol Pharmacol 56:99–105. https://doi.org/10.1016/j.etap.2017.08.034
Fahim MA, Tariq S, Adeghate E (2013) Vitamin E modifies the ultrastructure of testis and epididymis in mice exposed to lead intoxication. Ann Anat 195:272–277. https://doi.org/10.1016/j.aanat.2012.11.001
Johanisson E, Campana A, Luthi R, de Agostini A (2000) Evaluation of ‘round cells’ in semen analysis: a comparative study. Hum Rep Update 6:404–412. https://doi.org/10.1093/humupd/6.4.404
Palermo GD, Neri QV, Cozzubbo T, Cheung S, Pereira N, Rosenwaks Z (2016) Shedding light on the nature of seminal round cells. PLOS ONE 11:e0151640. https://doi.org/10.1371/journal.pone.0151640
Barraud-Lange V, Pont JC, Ziyyat A, Pocate KP, Sifer C, Cedrin-Durnerin I, Fechtali B, Ducot B, Wolf JP (2011) Seminal leukocytes are Good Samaritans for spermatozoa. FertilSteril 96:1315–1319. https://doi.org/10.1016/j.fertnstert.2011.09.035
Batra N, Nehru B, Bansal MP (2011) Influence of lead and zinc on rat male reproduction at ‘biochemical and histopathological levels.’ J Appl Toxicol 21:507–512. https://doi.org/10.1002/jat.796
Naha N, Chowdhury AR (2006) Inorganic lead exposure in battery and paint factory: effect on human sperm structure and functional activity. J UOEH 28:157–171. https://doi.org/10.7888/juoeh.28.157
Nasr NE, Elmadawy MA, Almadaly EA, Abdo W, Zamel MM (2017) Garlic powder attenuates apoptosis associated with lead acetate-induced testicular damage in adult male rats. Alex J Vet Sci 54:70–78. https://doi.org/10.5455/ajvs.268149
Yang JM, Arnush M, Chen QY, Wu XD, Pang B, Jiang XZ (2003) Cadmium-induced damage to primary cultures of rat Leydig cells. ReprodToxicol 17:553–560. https://doi.org/10.1016/S0890-6238(03)00100-X
Soleimanzadeh A, Kian M, Moradi S, Mahmoudi S (2020) Carob (Ceratonia siliqua L.) fruit hydro-alcoholic extract alleviates reproductive toxicity of lead in male mice: evidence on sperm parameters, sex hormones, oxidative stress biomarkers and expression of Nrf2 and iNOS. Avicenna J Phytomed 10:35–49. https://doi.org/10.22038/AJP.2019.13513
Li N, Hou Y, Ma D, Jing W, Dahms HU, Wang L (2015) Lead accumulation, oxidative damage and histopathological alteration in testes and accessory glands of freshwater crab, Sinopotamonhenanense, induced by acute lead exposure. Ecotoxicol Environ Saf 117:20–27. https://doi.org/10.1016/j.ecoenv.2015.03.019
Bechara EJH (2004) Lead poisoning and oxidative stress. In: Institute of de Quimica, Universidade de Sao Paulo, Brazil. SFRR`s 12th biennial meeting progamme and abstracts; 5–9 May 2004; Crown Plaza PanamericanoHotel Buenos Aires Argentina. Free Radic Biol Med 36:S9–46. p S22
Manna P, Sinha M, Sil PC (2009) Taurine plays a beneficial role against cadmium-induced oxidative renal dysfunction. Amino Acids 36:417–428. https://doi.org/10.1007/s00726-008-0094-x
Moskaug JØ, Carlsen H, Myhrstad MC, Blomhoff R (2005) Polyphenols and glutathione synthesis regulation. Am J Clin Nutr 81:277S-283S. https://doi.org/10.1093/ajcn/81.1.277S
Menacer A, Boukhatem MN, Benhelal A, Saïdi F (2017) In vitro antioxidant activity of different extracts of Algerian Allium plant (Allium triquetrum L.). Rev des Bio-Resources 7:80–91. https://doi.org/10.12816/0045885
Abdelhamid FM, Mahgoub HA, Ateya AI (2020) Ameliorative effect of curcumin against lead acetate–induced hemato-biochemical alterations, hepatotoxicity, and testicular oxidative damage in rats. Environ Sci Pollut Res Int 27(10):10950–10965. https://doi.org/10.1007/s11356-020-07718-3
Offor SJ, Mbagwu HOC, Orisakwe OE (2019) Improvement of lead acetate-induced testicular injury and sperm quality deterioration by Solanum anomalum Thonn. Ex. Schumach Fruit Extracts in Albino Rats. J Fam Reprod Health 13(2):98–108
Sharma P, Khan IA, Singh R (2018) Curcumin and quercetin ameliorated cypermethrin and deltamethrin-induced reproductive system impairment in male Wistar rats by upregulating the activity of pituitary gonadal hormones and steroidogenic enzymes. Int J Fertil Steril 12:72. https://doi.org/10.22074/ijfs.2018.5160
Odumosu FM, Olokodana O, Huthman IO, Akinyemi RA, Adesanya OA, Adefule AK (2013) Effect of onion, garlic and Aridan aqueous extract on hormone influenced testis weight of adult Wistar rats. Int J Chem Pharm Res 2:9
Kasuga S, Uda N, Kyo E, Ushijima M, Morihara N, Itakura Y (2001) Pharmacologic activities of aged garlic extract in comparison with other garlic preparations. J Nutr 131:1080S-S1084. https://doi.org/10.1093/jn/131.3.1080S
Nasr AY (2017) The impact of aged garlic extract on adriamycin-induced testicular changes in adult male Wistar rats. Acta Histochem 119(6):648–662. https://doi.org/10.1016/j.acthis.2017.07.006
Acknowledgements
The authors would like to thank the General Directorate of Scientific Research and Technological Development (DGRSDT) for supporting this project (award number 04/2016). Special thanks are given to Dr. T. Hamel (plant identification), Dr. M. Zebour, Dr. Z. Gouasmi, and Pr. Y. Hadef (HPLC analysis), University of Badji Mokhtar-Annaba. Thanks are also given to the Laboratory of Pathological Anatomy, EL-Hadjar, Annaba Hospital, for histological studies.
Funding
The author(s) received financial support for bench fees, but not for authorship, and/or for publication of this article.
Author information
Authors and Affiliations
Contributions
L Kahalerras drafted the manuscript, managed the experimental work, and contributed to the analysis and interpretation of results; I Otmani participated in the experimental work; C Abdennour corrected and revised the manuscript. All authors read and gave the final approval of manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Experiments were carried out according to the international animal handling of Helsinki Declaration of 2008 and to the National Ethical Committee of Animal Sciences.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kahalerras, L., Otmani, I. & Abdennour, C. Wild Garlic Allium triquetrum L. Alleviates Lead Acetate-Induced Testicular Injuries in Rats. Biol Trace Elem Res 200, 2205–2222 (2022). https://doi.org/10.1007/s12011-021-02818-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-021-02818-8