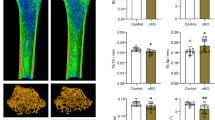
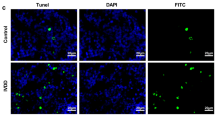
Abstract
Recent evidence suggests a role of type II collagen in Kashin-Beck disease (KBD) degeneration. We aimed to assess the abnormal expression of heat shock protein 47 (HSP47) which is associated with a decrease in type II collagen and an increase in cartilage degradation in KBD. Hand phalange cartilages were collected from KBD and healthy children. Rats were administered with T-2 toxin under the selenium (Se)-deficient diet. ATDC5 cells were seeded on bone matrix gelatin to construct engineered cartilaginous tissue. C28/I2 and ATDC5 cells and engineered tissue were exposed to different concentrations of T-2 toxin with or without Se. Cartilage degeneration was determined through histological evaluation. The distribution and expression of type II collagen and HSP47 were investigated through immunohistochemistry, western blotting, and real-time PCR. KBD cartilages showed increased chondronecrosis and extracellular matrix degradation in deep zone with decreased type II collagen and HSP47 expression. The low-Se + T-2 toxin animal group showed a significantly lower type II collagen expression along with decreased HSP47 expression. Decreased type II collagen and HSP47 in C28/I2 and ATDC5 cells induced by T-2 toxin showed a dose-dependent manner. Hyaline-like cartilage with zonal layers was developed in engineered cartilaginous tissues, with decreased type II collagen and HSP47 expression found in T-2 toxin-treated group. Se-supplementation partially antagonized the inhibitory effects of T-2 toxin in chondrocytes and cartilages. HSP47 plays a role in the degenerative changes of KBD and associated with T-2 toxin-induced decreased type II collagen expression, further promoting matrix degradation.





Similar content being viewed by others
References
Allander E (1994) Kashin-Beck disease. An analysis of research and public health activities based on a bibliography 1849-1992. Scand J Rheumatol Suppl 99:1–36. https://doi.org/10.3109/03009749409117126
Guo X, Ma WJ, Zhang F, Ren FL, Qu CJ, Lammi MJ (2014) Recent advances in the research of an endemic osteochondropathy in China: Kashin-Beck disease. Osteoarthr Cartil 22:1774–1783. https://doi.org/10.1016/j.joca.2014.07.023
Guo X (2001) Diagnostic, clinical and radiological characteristics of Kashin-Beck disease in Shaanxi Province, PR China. Int Orthop 25:147–150
Huang Q, Zhou ZK, Ma J, Li Y, Yang X, Shen B, Yang J, Kang PD, Pei FX (2015) The arthropathic and functional impairment features of adult Kashin-Beck disease patients in Aba Tibetan area in China. Osteoarthr Cartil 23:601–606. https://doi.org/10.1016/j.joca.2015.01.005
Song QQ, Liu H, Lian W, Wang Y, Sun LY, Gao M, Liu YQ, Deng HX, Deng Q, Liu N, Yu J (2018) The characteristics of positive and confusing hand X-ray signs in diagnosing Kashin-Beck disease in children in Chiina. Sci Rep 8:3277. https://doi.org/10.1038/s41598-018-21675-5
Zhang GQ, Liu JX, Yu JY, Shi YX, Zhang SJ (1989) Ultrastructure of chondrocytes in articular cartilage of Kashin-Beck disease. Chin J Endemiol 8:27–30
Zhang Y, He Y, Zhang D, Zhang M, Wang MY, Zhang Y, Ma T, Chen J (2018) Death of chondrocytes in Kashin-Beck disease: apoptosis, necrosis or necroptosis? Int J Exp Pathol 99:312–322. https://doi.org/10.1111/iep.12297
Wang KW, Yu J, Liu H, Liu YQ, Liu N, Cao Y, Zhang X, Sun D (2020) Endemic Kashin-Beck disease: a food-sourced osteoarthropathy. Semin Arthritis Rheum 50:366–372. https://doi.org/10.1016/j.semarthrit.2019.07.014
Guo YN, Li HR, Yang LS, Li YH, Wei BG, Wang WY et al (2017) Trace element levels in scalp hair of school children in Shigase, Tibet, an endemic area for Kashin-Beck disease(KBD). Biol Trace Elem Res 180:15–22. https://doi.org/10.1007/s12011-017-0988-0
Chen JH, Xue S, Li S, Wang ZL, Yang H, Wang W, Song D, Zhou X, Chen C (2012) Oxidant damage in Kashin-Beck disease and a rat Kashin-Beck disease model by employing T-2 toxin treatment under selenium deficient conditions. J Orthop Res 30:1229–1237. https://doi.org/10.1002/jor.22073
Guan F, Li S, Wang ZL, Yang H, Xue S, Wang W, Song D, Zhou X, Zhou W, Chen JH, Caterson B, Hughes C (2013) Histopathology of chondronecrosis development in knee articular cartilage in a rat model of Kashin-Beck disease using T-2 toxin and selenium deficiency conditions. Rheumatol Int 33:157–166. https://doi.org/10.1007/s00296-011-2335-7
Song QQ, Sun LY, Li CH, Liu YJ, Cui SL, Liu YQ, Cao YH, Pei JR, Wang Y, Lian W, Jiao Z, Deng Q, Yu J (2019) The urinary levels of CTX- II, C2C, PYD, and helix-II increased among adults with KBD: a cross-sectional study. J Orthop Surg Res 14:328. https://doi.org/10.1186/s13018-019-1392-6
Chang Y, Wang X, Sun Z, Jin Z, Chen M, Wang X, Lammi MJ, Guo X (2017) Inflammatory cytokine of IL-1β is involved in T-2 toxin-triggered chondrocyte injury and metabolism imbalance by the activation of Wnt/β-catenin signaling. Mol Immunol 91:195–201. https://doi.org/10.1016/j.molimm.2017.08.019
Chen JH, Luo MX, Wang W, Zhang ZT, He Y, Duance VC et al (2015) Altered proteolytic activity and expression of MMPs and aggrecanases and their inhibitors in Kashin-Beck disease. J Orthop Res 33:47–55. https://doi.org/10.1002/jor.22708
Wang W, Guo X, Chen JH, Xu P, Lammi MJ (2008) Morphology and phenotype expression of types I, II, III, and X collagen and MMP-13 of chondrocytes cultured from articular cartilage of Kashin-Beck disease. J Rheumatol 35:696–702
Zhou X, Yang H, Guan F, Xue S, Song D, Chen J, Wang Z (2015) T-2 toxin alters the levels of collagen II and its regulatory enzymes MMPs/TIMP-1 in a low-selenium rat model of Kashin-Beck disease. Biol Trace Elem Res 169:237–246. https://doi.org/10.1007/s12011-015-0408-2
Ito S, Nagata K (2018) Roles of the endoplasmic reticulum–resident, collagen-specific molecular chaperone Hsp47 in vertebrate cells and human disease. J Biol Chem 294:2133–2141. https://doi.org/10.1074/jbc.tm118.002812
Ito S, Nagata K (2017) Biology of Hsp47 (Serpin H1), a collagen-specific molecular chaperone. Semin Cell Dev Biol 62:142–151. https://doi.org/10.1016/j.semcdb.2016.11.005
Nagata K (2003) HSP47 as a collagen-specific molecular chaperone: function and expression in normal mouse development. Semin Cell Dev Biol 14:275–282. https://doi.org/10.1016/j.semcdb.2003.09.020
Matsuoka Y, Kubota H, Adachi E, Nagai N, Marutani T, Hosokawa N, Nagata K (2004) Insufficient folding of type IV collagen and formation of abnormal basement membrane-like structure in embryoid bodies derived from Hsp47-null embryonic stem cells. Mol Biol Cell 15:4467–4475. https://doi.org/10.1091/mbc.e04-01-0050
Masago Y, Hosoya A, Kawasaki K, Kawano S, Nasu A, Toguchida J, Fujita K, Nakamura H, Kondoh G, Nagata K (2012) The molecular chaperone Hsp47 is essential for cartilage and endochondral bone formation. J Cell Sci 125:1118–1128. https://doi.org/10.1242/jcs.089748
Chioran A, Duncan S, Catalano A, Brown TJ, Ringuette MJ (2017) Collagen IV trafficking: the inside-out and beyond story. Dev Biol 431:124–133. https://doi.org/10.1016/j.ydbio.2017.09.037
Sengupta P (2013) The laboratory rat: relating its age with Human’s. Int J Prev Med 4:624–630
Reeves PG (1997) Components of the AIN-93 diets as improvements in the AIN-76A diet. J Nutr 127:838S–841S. https://doi.org/10.1093/jn/127.5.838S
Carballo CB, Nakagawa Y, Sekiya I, Rodeo SA (2017) Basic science of articular cartilage. Clin Sports Med 36:413–425. https://doi.org/10.1016/j.csm.2017.02.001
Evans HB, Ayad S, Abedin MZ, Hopkins S, Morgan K, Walton KW, Weiss JB, Holt PJ (1983) Localisation of collagen types and fibronectin in cartilage by immunofluorescence. Ann Rheum Dis 42:575–581. https://doi.org/10.1136/ard.42.5.575
Eyre DR (2004) Collagens and cartilage matrix homeostasis. Clin Orthop Relat Res 427:S118–S122. https://doi.org/10.1097/01.blo.0000144855.48640.b9
Garnero P, Ayral X, Rousseau JC, Christgau S, Sandell LJ, Dougados M, Delmas PD (2002) Uncoupling of type II collagen synthesis and degradation predicts progression of joint damage in patients with knee osteoarthritis. Arthritis Rheum 46:2613–2624. https://doi.org/10.1002/art.10576
Wang XX, Cai L (2018) Expression level of proteoglycan, collagen and type II collagen in osteoarthritis rat model is promoted and degradation of cartilage is prevented by glucosamine methyl ester. Eur Rev Med Pharmacol Sci 22:3609–3616. https://doi.org/10.26355/eurrev_201806_15188
Alam J, Jantan I, Kumolosasi E, Nafiah MA, Mesaik MA (2018) Suppressive effects of the standardized extract of Phyllanthus amarus on type II collagen-induced rheumatoid arthritis in Sprague Dawley rats. Curr Pharm Biotechnol 19:1156–1169. https://doi.org/10.2174/1389201020666181211124954
Firner S, Zaucke F, Michael J, Dargel J, Schiwy-Bochat KH, Heilig J, Rothschild MA, Eysel P, Brüggemann GP, Niehoff A (2017) Extracellular distribution of collagen II and perifibrillar adapter proteins in healthy and osteoarthritic human knee joint cartilage. J Histochem Cytochem 65:593–606. https://doi.org/10.1369/0022155417729154
Cheng B, Zhang Y, Tong B, Yin H (2017) Influence of selenium on the production of T-2 toxin by Fusarium poae. Biol Trace Elem Res 178:147–152. https://doi.org/10.1007/s12011-016-0900-3
Khan ES, Sankaran S, Paez JI, Muth C, Han MKL, del Campo A (2019) Photoactivatable Hsp47: a tool to regulate collagen secretion and assembly. Adv Sci 6(9):1801982. https://doi.org/10.1002/advs.201801982
Marutani T, Yamamoto A, Nagai N, Kubota H, Nagata K (2004) Accumulation of type IV collagen in dilated ER leads to apoptosis in Hsp47-knockout mouse embryos via induction of CHOP. J Cell Sci 117:5913–5922
Li X, Jin L, Balian G, Laurencin CT, Greg Anderson D (2006) Demineralized bone matrix gelatin as scaffold for osteochondral tissue engineering. Biomaterials 27:2426–2433. https://doi.org/10.1016/j.biomaterials.2005.11.040
Pei Y, Fan JJ, Zhang XQ, Zhang ZY, Yu M (2014) Repairing the osteochondral defect in goat with the tissue-engineered osteochondral graft preconstructed in a double-chamber stirring bioreactor. Biomed Res Int 2014:219203–219211. https://doi.org/10.1155/2014/219203
Nagai N, Hosokawa M, Itohara S, Adachi E, Matsushita T, Hosokawa N, Nagata K (2000) Embryonic lethality of molecular chaperone hsp47 knockout mice is associated with defects in collagen biosynthesis. J Cell Biol 150:1499–1506. https://doi.org/10.1083/jcb.150.6.1499
Funding
This study was supported by the National Natural Science Foundation of China (No. 81872565, No. 81573102, No. 81273006).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interests
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(PDF 139 kb)
Rights and permissions
About this article
Cite this article
Zhang, M., Wang, M., Wang, H. et al. Decreased Expression of Heat Shock Protein 47 Is Associated with T-2 Toxin and Low Selenium-Induced Matrix Degradation in Cartilages of Kashin-Beck Disease. Biol Trace Elem Res 199, 944–954 (2021). https://doi.org/10.1007/s12011-020-02237-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02237-1