Abstract
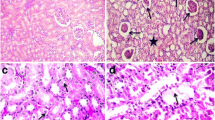
The examination chemical factors including industrial toxins and heavy metals seem to be crucial during the prepubertal period. In order to investigate the effects of prepubertal exposure to toxic doses of Cd on liver, hematological, and biochemical parameters in the serum, 16 female rats weaned on postnatal day (PND) 21 were randomly divided into four groups with four rats in each (n = 4). The treatments were as follows: control (0.5 mL distilled water), 25, 50, and 75 mg/kg/day received cadmium chloride (CdCl2). The CdCl2 were administered orally from PND 21 days until observed first vaginal opening (VO). The result showed that the treatment of 75 mg/kg CdCl2 dramatically increased the serum level of LDL (P < 0.0001) and LDL/HDL ratio (P = 0.0004). Conversely, treatment of 75 mg/kg CdCl2 considerably decreased the serum level of HDL in comparison with control group (P = 0.0002). Nevertheless, the rats that received different doses of CdCl2 showed no significant differences in Glu, TG, and TC compared to control group. Number of RBC and Hb of rats treated with 75 mg/kg CdCl2 were significantly less than the other groups (P < 0.0001), whereas a number of WBCs in rats treated with 75 mg/kg CdCl2 (5.27 ± 0.13 103/μL) showed significant difference (P < 0.0001) compared to control group (4.23 ± 0.09 103/μL). Histopathological exams showed nodular accumulation of lymphocytes in the liver (lymphocytic hepatitis) of rats, treated with 75 mg/kg CdCl2. These results showed that CdCl2 could cause change in serum lipidome and hematological parameters. What is more, exposure to Cd triggers liver injury and cardiovascular disease during the prepubertal period.





Similar content being viewed by others
References
Agarwal S, Zaman T, Murat Tuzcu E, Kapadia SR (2011) Heavy metals and cardiovascular disease: results from the National Health and nutrition examination survey (NHANES) 1999-2006. Angiology 62:422–429
Andjelkovic M, Buha Djordjevic A, Antonijevic E, Antonijevic B, Stanic M, Kotur-Stevuljevic J, Spasojevic-Kalimanovska V, Jovanovic M, Boricic N, Wallace D, Bulat Z (2019) Toxic effect of acute cadmium and Lead exposure in rat blood, liver, and kidney. Int J Environ Res Public Health 16:274
Arroyo V, Flores K, Ortiz L et al (2012) Liver and cadmium toxicity. J Drug Metab Toxicol S 5
Atsdr S (2012) Toxicological profile for chromium. Agency for Toxic Substances and Disease Registry. Public Health Service, US Department of Health and Human Services. http://www.atsdr.cdc.gov/toxprofiles/tp.asp
Badisa VL, Latinwo LM, Odewumi CO et al (2007) Mechanism of DNA damage by cadmium and interplay of antioxidant enzymes and agents. Environmental Toxicology: An International Journal 22:144–151
Bhattacharyya M (2000) Biochemical pathways in cadmium toxicity. Molecular biology and toxicology of metals:34–74
Bizoń A, Milnerowicz H (2017) The effect of passive and active exposure to tobacco smoke on lipid profile parameters and the activity of certain membrane enzymes in the blood of women in the first trimester of pregnancy. Environ Toxicol Pharmacol 53:74–80
Brzóska MM (2012) Low-level chronic exposure to cadmium enhances the risk of long bone fractures: a study on a female rat model of human lifetime exposure. J Appl Toxicol 32:34–44
Brzóska MM, Galażyn-Sidorczuk M, Dzwilewska I (2013) Ethanol consumption modifies the body turnover of cadmium: a study in a rat model of human exposure. J Appl Toxicol 33:784–798
Chapatwala K, Hobson M, Desaiah D et al (1982) Effect of cadmium on hepatic and renal gluconeogenic enzymes in female rats. Toxicol Lett 12:27–34
Chapatwala K, Rajanna E, Desaiah D (1980) Cadmium induced changes in gluconeogenic enzymes in rat kidney and liver. Drug Chem Toxicol 3:407–420
Chen JP (2012) Decontamination of heavy metals: processes, mechanisms, and applications. Crc Press
Ding D, Li X, Qiu J et al. (2014) Serum lipids, apolipoproteins, and mortality among coronary artery disease patients. BioMed research international 2014
Duffus JH (2002) " heavy metals" a meaningless term? (IUPAC technical report). Pure Appl Chem 74:793–807
El-Boshy ME, Risha EF, Abdelhamid FM et al (2015) Protective effects of selenium against cadmium induced hematological disturbances, immunosuppressive, oxidative stress and hepatorenal damage in rats. J Trace Elem Med Biol 29:104–110
Enomoto M, Adachi H, Hirai Y et al (2011) LDL-C/HDL-C ratio predicts carotid intima-media thickness progression better than HDL-C or LDL-C alone. J Lipids:2011
Ferraro PM, Costanzi S, Naticchia A, Sturniolo A, Gambaro G (2010) Low level exposure to cadmium increases the risk of chronic kidney disease: analysis of the NHANES 1999-2006. BMC Public Health 10:304
Franssen D, Ioannou YS, Alvarez-Real A et al (2014) Pubertal timing after neonatal diethylstilbestrol exposure in female rats: neuroendocrine vs peripheral effects and additive role of prenatal food restriction. Reprod Toxicol 44:63–72
Gadelha ICN, Fernandes De Macedo M, Oloris SCS et al (2014) Gossypol promotes degeneration of ovarian follicles in rats. Sci World J:2014
Giesbrecht GF, Ejaredar M, Liu J et al (2017) Prenatal bisphenol a exposure and dysregulation of infant hypothalamic-pituitary-adrenal axis function: findings from the APrON cohort study. Environ Health 16:47
Gill TS, Epple A (1993) Stress-related changes in the hematological profile of the American eel (Anguilla rostrata). Ecotoxicol Environ Saf 25:227–235
Go Y-M, Sutliff RL, Chandler JD, Khalidur R, Kang BY, Anania FA, Orr M, Hao L, Fowler BA, Jones DP (2015) Low-dose cadmium causes metabolic and genetic dysregulation associated with fatty liver disease in mice. Toxicol Sci 147:524–534
Graczyk A, Radomska K, Dlugaszek M (2000) Synergizm i antagonizm miedzy biopierwiastkami i metalami toksycznymi. Ochrona Środowiska i Zasobów Naturalnych:39–45
Hambach R, Lison D, D’haese P et al (2013) Co-exposure to lead increases the renal response to low levels of cadmium in metallurgy workers. Toxicol Lett 222:233–238
Han X, He Y, Zeng G, Wang Y, Sun W, Liu J, Sun Y, Yu J (2017) Intracerebroventricular injection of RFRP-3 delays puberty onset and stimulates growth hormone secretion in female rats. Reprod Biol Endocrinol 15:35
Hoshyari E, Pourkhabbaz A, Mansouri B (2012) Contaminations of metal in tissues of Siberian gull Larusheuglini: gender, age, and tissue differences. Bull Environ Contam Toxicol 89:102–106
Hounkpatin A, Edorh P, Guédénon P et al (2013) Haematological evaluation of Wistar rats exposed to chronic doses of cadmium, mercury and combined cadmium and mercury. Afr J Biotechnol 12
Ibraheem AS, Seleem AA, El-Sayed MF et al (2016) Single or combined cadmium and aluminum intoxication of mice liver and kidney with possible effect of zinc. The Journal of Basic & Applied Zoology 77:91–101
Ibrahim MA, Almaeen AH, El Moneim MA et al (2018) Cadmium-induced hematological, renal, and hepatic toxicity: the amelioration by spirulina platensis. The Saudi Journal of Forensic Medicine and Sciences 1:5
Järup L (2003) Hazards of heavy metal contamination. Br Med Bull 68:167–182
Jung U, Choi M-S (2014) Obesity and its metabolic complications: the role of adipokines and the relationship between obesity, inflammation, insulin resistance, dyslipidemia and nonalcoholic fatty liver disease. Int J Mol Sci 15:6184–6223
Jurczuk M, Brzóska MM, Moniuszko-Jakoniuk J et al (2004) Antioxidant enzymes activity and lipid peroxidation in liver and kidney of rats exposed to cadmium and ethanol. Food Chem Toxicol 42:429–438
Jurczuk M, Moniuszko-Jakoniuk J, Brzóska M et al (2003) Superoxidase dismutase and catalase activities and malondialdehyde concentration in the brain rats co-exposed to lead or cadmium and ethanol. Pol J Environ Stud 12:199–203
Kinouchi R, Matsuzaki T, Iwasa T, Gereltsetseg G, Nakazawa H, Kunimi K, Kuwahara A, Yasui T, Irahara M (2012) Prepubertal exposure to glucocorticoid delays puberty independent of the hypothalamic Kiss1-GnRH system in female rats. Int J Dev Neurosci 30:596–601
Kojima M, Masui T, Nemoto K, Degawa M (2004) Lead nitrate-induced development of hypercholesterolemia in rats: sterol-independent gene regulation of hepatic enzymes responsible for cholesterol homeostasis. Toxicol Lett 154:35–44
Kucuk A, Uslu AU, Icli A et al (2017) The LDL/HDL ratio and atherosclerosis in ankylosing spondylitis. Z Rheumatol 76:58–63
Larregle EV, Varas SM, Oliveros LB, Martinez LD, Antón R, Marchevsky E, Giménez MS (2008) Lipid metabolism in liver of rat exposed to cadmium. Food Chem Toxicol 46:1786–1792
Liju VB, Jeena K, Kuttan R (2013) Acute and subchronic toxicity as well as mutagenic evaluation of essential oil from turmeric (Curcuma longa L). Food Chem Toxicol 53:52–61
Lucia M, André J-M, Gonzalez P, Baudrimont M, Bernadet MD, Gontier K, Maury-Brachet R, Guy G, Davail S (2010) Effect of dietary cadmium on lipid metabolism and storage of aquatic bird Cairina moschata. Ecotoxicology 19:163–170
Marcondes F, Bianchi F, Tanno A (2002) Determination of the estrous cycle phases of rats: some helpful considerations. Braz J Biol 62:609–614
Mehbub M, Lei J, Franco C, Zhang W (2014) Marine sponge derived natural products between 2001 and 2010: trends and opportunities for discovery of bioactives. Marine drugs 12:4539–4577
Menke A, Muntner P, Silbergeld EK et al (2008) Cadmium levels in urine and mortality among US adults. Environ Health Perspect 117:190–196
Murugavel P, Pari L (2007) Diallyl tetrasulfide protects cadmium-induced alterations in lipids and plasma lipoproteins in rats. Nutr Res 27:356–361
Ng DS (2013) Lecithin cholesterol acyltransferase deficiency protects from diet-induced insulin resistance and obesity—novel insights from mouse models. Vitamins & Hormones, Elsevier, pp 259–270
Ojeda SR, Skinner MK (2006) Puberty in the rat. In: Knobil and Neill's physiology of reproduction. Elsevier Inc
Olisekodiaka M, Igbeneghu C, Onuegbu A et al (2012) Lipid, lipoproteins, total antioxidant status and organ changes in rats administered high doses of cadmium chloride. Med Princ Pract 21:156–159
Pamir N, Hutchins P, Ronsein G, Vaisar T, Reardon CA, Getz GS, Lusis AJ, Heinecke JW (2016) Proteomic analysis of HDL from inbred mouse strains implicates APOE associated with HDL in reduced cholesterol efflux capacity via the ABCA1 pathway. J Lipid Res 57:246–257
Parasuraman S, Raveendran R, Kesavan R (2010) Blood sample collection in small laboratory animals. J Pharmacol Pharmacother 1:87
Ramaiah L, Bounous DI, Elmore SA (2013) Hematopoietic system. In: Haschek and Rousseaux's handbook of Toxicologic pathology. Elsevier, Third Edition, pp 1863–1933
Ramirez DC, Gimenez MS (2002) Lipid modification in mouse peritoneal macrophages after chronic cadmium exposure. Toxicology 172:1–12
Rogalska J, Brzóska MM, Roszczenko A, Moniuszko-Jakoniuk J (2009) Enhanced zinc consumption prevents cadmium-induced alterations in lipid metabolism in male rats. Chem Biol Interact 177:142–152
Samarghandian S, Azimi-Nezhad M, Shabestari MM, Azad FJ, Farkhondeh T, Bafandeh F (2015) Effect of chronic exposure to cadmium on serum lipid, lipoprotein and oxidative stress indices in male rats. Interdiscip Toxicol 8:151–154
Sarmiento-Ortega VE, Treviño S, Flores-Hernández JÁ, Aguilar-Alonso P, Moroni-González D, Aburto-Luna V, Diaz A, Brambila E (2017) Changes on serum and hepatic lipidome after a chronic cadmium exposure in Wistar rats. Arch Biochem Biophys 635:52–59
Satarug S, Baker JR, Urbenjapol S, Haswell-Elkins M, Reilly PEB, Williams DJ, Moore MR (2003) A global perspective on cadmium pollution and toxicity in non-occupationally exposed population. Toxicol Lett 137:65–83
Schwartz GG, Il’yasova D, Ivanova A (2003) Urinary cadmium, impaired fasting glucose, and diabetes in the NHANES III. Diabetes Care 26:468–470
Smyth CE, Knee R, Wilkinson M, Murphy PR (1997) Decline in basic fibroblast growth factor (FGF-2) mRNA expression in female rat hypothalamus at puberty. J Neuroendocrinol 9:151–159
Syers J, Gochfeld M (2000) Environmental cadmium in the food chain: sources, pathways, and risks. In: Proceedings of the SCOPE Workshop, Brussels. p 13–16
Takiguchi M, Yoshihara SI (2006) New aspects of cadmium as endocrine disruptor. Environmental Sciences: an international journal of environmental physiology and toxicology 13:107–116
Tellez-Plaza M, Navas-Acien A, Menke A, Crainiceanu CM, Pastor-Barriuso R, Guallar E (2012) Cadmium exposure and all-cause and cardiovascular mortality in the US general population. Environ Health Perspect 120:1017–1022
Tinkov AA, Filippini T, Ajsuvakova OP et al (2017) The role of cadmium in obesity and diabetes. Sci Total Environ 601:741–755
Tomaszewska E, Dobrowolski P, Winiarska-Mieczan A, Kwiecień M, Tomczyk A, Muszyński S (2017) The effect of tannic acid on the bone tissue of adult male Wistar rats exposed to cadmium and lead. Exp Toxicol Pathol 69:131–141
Toth D, Wonger H (2003) Clinical chemistry textbook. Oxford University Press, Oxford
Umemura T (2000) Experimental reproduction of itai-itai disease, a chronic cadmium poisoning of humans, in rats and monkeys. Jpn J Vet Res 48:15–28
Winiarska-Mieczan A (2013) Protective effect of tannic acid on the brain of adult rats exposed to cadmium and lead. Environ Toxicol Pharmacol 36:9–18
Yamano T, Shimizu M, Noda T (1998) Comparative effects of repeated administration of cadmium on kidney, spleen, thymus, and bone marrow in 2-, 4-, and 8-month-old male Wistar rats. Toxicol Sci 46:393–402
Yang R, Wang Y-M, Zhang L-S, Zhang L, Zhao ZM, Zhao J, Peng SQ (2015) Delay of the onset of puberty in female rats by prepubertal exposure to T-2 toxin. Toxins 7:4668–4683
Yang R, Wang Y, Zhang L et al (2016) Prepubertal exposure to T-2 toxin advances pubertal onset and development in female rats via promoting the onset of hypothalamic–pituitary–gonadal axis function. Hum Exp Toxicol 35:1276–1285
Yildirim S, Celikezen FC, Oto G, Sengul E, Bulduk M, Tasdemir M, Ali Cinar D (2018) An investigation of protective effects of litium borate on blood and histopathological parameters in acute cadmium-induced rats. Biol Trace Elem Res 182:287–294
Yuan G, Dai S, Yin Z et al (2014) Toxicological assessment of combined lead and cadmium: acute and sub-chronic toxicity study in rats. Food Chem Toxicol 65:260–268
Yuhas E, Miya T, Schnell R (1979) Dose-related alterations in growth and mineral disposition by chronic oral cadmium administration in the male rat. Toxicology 12:19–29
Acknowledgments
The authors are grateful to members of the Center of Comparative and Experimental Medicine, Shiraz University of Medical Sciences, Shiraz.
Funding
The Shiraz University provided financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interests.
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Saedi, S., Jafarzadeh Shirazi, M.R., Totonchi, M. et al. Effect of Prepubertal Exposure to CdCl2 on the Liver, Hematological, and Biochemical Parameters in Female Rats; an Experimental Study. Biol Trace Elem Res 194, 472–481 (2020). https://doi.org/10.1007/s12011-019-01800-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-019-01800-9