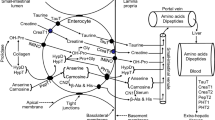
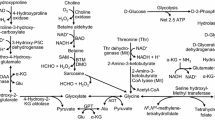
Abstract
This work determines the effect nopal consumption at different maturity stages (60, 200, 400, and 600 g) as the only calcium source in bone metabolism. The apparent mineral absorption, the biomarkers of bone metabolism, the bone mineral density at different femoral regions, and crystal properties of the bone were evaluated during the growth stage. The Ca absorption was increased with the rat age in most of the experimental groups, while Mg supplementation decreased intestinal absorption probably due to a saturation process. Intestinal Ca and Mg absorption showed an opposite trend; this result suggests that both ions can compete for vitamin D absorption sites. The percentage of absorption of K was lower in the groups fed with Nopal; nevertheless, due to supplementation, the net absorption was higher than the control group. In all groups, osteocalcin levels decreased with the rat age. Nopal consumption increased osteocalcin levels during the adolescence stage in comparison to the control group. Amino N-terminal propeptide of type I procollagen levels increased in puberty and adolescence in all groups compared to the control group. Bone mineral density in different femoral regions was lower in the groups fed with nopal at early maturity stages (N-60 and N-200) than the groups fed with nopal at late maturity stages (N-400 and N-600). The crystal size of hydroxyapatite exhibited changes for all the groups, indicating the inclusion of mono and divalent ions in calcium replacement. On this basis, the nopal at late maturity stage contributed to bone formation.




Similar content being viewed by others
References
Boskey AL, Robey PG (2013) The composition of bone: relationships to bone fragility and antiosteoportotic drug effects. Bone Key Reports 447(2):1–11. https://doi.org/10.1038/bonekey.2013.181
Rogers A, Hannon RA, Eastell R (2000) Biochemical markers as predictors of rates of bone loss after menopause. J Bone Miner Res 15(7):1398–1404. https://doi.org/10.1359/jbmr.2000.15.7.1398
McKee MD, Cole WG (2012) Bone matrix and mineralization. In: Pediatric Bone, pp 9–37. https://doi.org/10.1016/B978-0-12-382040-2.10002-4
Londoño-Restrepo SM, Ramirez-Gutierrez CF, Del Real A, Rubio-Rosas E, Rodriguez-Garcia ME (2016) Study of bovine hydroxyapatite obtained by calcination at low heating rates and cooled in furnace air. J Mater Sci 51(9):4431–4441. https://doi.org/10.1007/s10853-016-9755-4
Matsunaga K, Murata H (2009) Formation energies of substitutional sodium and potassium in hydroxyapatite. Mater Trans 50(5):1041–1045. https://doi.org/10.2320/matertrans.MC200819
Tucker KL, Hannan MT, Chen H, Cupples LA, Wilson PW, Kiel DP (1999) Potassium, magnesium, and fruit and vegetable intakes are associated with greater bone mineral density in elderly men and women. Am J Clin Nutr 69(4):727–736. https://doi.org/10.1111/j.1532-5415.2005.53561.x
Dawson-Hughes B, Harris SS, Palermo NJ, Gilhooly CH, Shea MK, Fielding RA, Ceglia L (2015) Potassium bicarbonate supplementation lowers bone turnover and calcium excretion in older men and women: a randomized dose-finding trial. J Bone Miner Res 30(11):2103–2111. https://doi.org/10.1002/jbmr.2554
Jimenez-Mendoza D, Espinosa-Arbelaez DG, Giraldo-Betancur AL, Hernandez-Urbiola MI, Vargas-Vazquez D, Rodriguez-Garcia ME (2011) Single x-ray transmission system for bone mineral density determination. Rev Sci Instrum 82:125105. https://doi.org/10.1063/1.3666864
Fernández-Ortega M, Bioquímica DD, De MF, De PU (2008) Consumo de fuentes de calcio en adolescentes mujeres en Panamá. Arch Latinoam Nutr 58:286–291 doi:0004–0622
Barquera S, Hernández-Barrera L, Campos-Nonato I, Espinosa J, Flores M, Rivera JA (2009) Energy and nutrient consumption in adults: analysis of the mexican national health and nutrition survey. Salud Publica Mex 51(4):S562–S573. https://doi.org/10.1590/S0036-36342009001000011
Aguilera-Barreiro de L M, Rivera-Marquez JA, Trujillo-Arriaga HM, Tamayo-Orozco JA, Barreira-Mercado E, Rodrıguez-García ME (2013) Intake of dehydrated nopal (opuntia ficus indica) improves bone mineral density and calciuria in adult Mexican women. Food Nutr Res 57(1):1–9. https://doi.org/10.3402/fnr.v57i0.19106
Frati-Munari AC, Altamirano-Bustamante E, Rodriguez-Barcenas N, Ariza-Andraca R, Lopez-Ledesma R (1989) Hypoglycemic action of Opuntia streptacantha Lemaire: study using raw extracts. Arch Invest Med 20(4):321–325
Rojas-Molina IJ, Gutiérrez-Cortez E, Bah M, Rojas-Molina A, Ibarra-Alvarado C, Rivera E, Del Real A, Aguilera-Barreiro A (2015) Characterization of calcium compounds in Opuntia ficus indica as a source of calcium for human diet. J Chemother 7:1–8. https://doi.org/10.1155/2015/710328
Reeves PG, Nielsen FH, Fahey JGC (1993) AIN-93 purified diets for laboratory rodents, final report of the American Institute of Nutrition ad hoc writing committee on reformulation of the AIN-76A rodent diet. J Nutr 123:1939–1951
Sengupta P (2013) The laboratory rat: relating its age with human’s. Lab rat relat its age with human’s, vol 4, pp 624–630 doi:23930179
Zafar TA, Weaver CM, Zhao YD, Martin BR, Wastney ME (2004) Nondigestible oligosaccharides increase calcium absorption and suppress bone resorption in ovariectomized rats. J Nutr 134(2):399–402
Hernández-Becerra E, Gutiérrez-Cortez E, Del Real A, Rojas-Molina A, Rodriguez-Garcia ME, Rubio E, Quintero-Garcia M, Rojas-Molina I (2017) Bone mineral density, mechanical, microstructural properties and mineral content of the femur in growing rats fed with cactus opuntia ficus indica (L.) mill. (Cactaceae) cladodes as calcium source in diet. Nutrients 9(2):108–125. https://doi.org/10.3390/nu9020108
Lobo AR, Colli C, Filisetti TMCC (2006) Fructooligosaccharides improve bone mass and biomechanical properties in rats. Nutr Res 26:413–420. https://doi.org/10.1016/j.nutres.2006.06.019
Choi IS, Jung ES, Choi YE, Cho YK, Yang EM, Kim CJ (2013) Random urinary calcium/creatinine ratio for screening hypercalciuria in children with hematuria. Ann Lab Med 33(6):401–405. https://doi.org/10.3343/alm.2013.33.6.401
Londoño-Restrepo SM, Jeronimo-Cruz R, Rubio-Rosas E, Rodriguez-García ME (2018) The effect of cyclic heat treatment on the physicochemical properties of bio hydroxyapatite from bovine bone. J Mater Sci Mater Med 29:52–67. https://doi.org/10.1007/s10856-018-6061-5
Holzwarth U, Gibson N (2011) The Scherrer equation versus the 'Debye-Scherrer equation. Nat Nanotechnol 6(9):534
Contreras-Padilla M, Pérez-Torrero E, Hernández-Urbiola MI, Hernandez-Quevedo G, Del Real A, Rivera-Muñoz EM, Rodriguez-Garcia ME (2011) Evaluation of oxalates and calcium in nopal pads (Opuntia ficus-indica var. Redonda) at different maturity stages. J Food Compos Anal 24(1):38–43. https://doi.org/10.1016/j.jfca.2010.03.028
Hunt JR, Hunt CD, Zito CA, Idso JP, Johnson LK (2008) Calcium requirements of growing rats based on bone mass, structure, or biomechanical strength are similar. J Nutr 138(8):1462–1468. https://doi.org/10.1093/jn/138.8.1462
Pineda-Gomez P, Hernández-Becerra P, Rojas-Molina I, Rosales-Rivera A, Rodríguez-Garcia ME (2018) The effect of calcium deficiency on bone properties in growing rats. Curr Nutr Food Sci 14:1–9. https://doi.org/10.2174/1573401314666180919142102
Toba Y, Masuyama R, Kato K, Takada Y, Aoe S, Suzuki K (1999) Effects of dietary magnesium level on calcium absorption in growing male rats. Nutr Res 19(5):783–793. https://doi.org/10.1016/S0271-5317(99)00040-8
Fine KD, Santa-Ana CA, Porter JL, Fordtran JS (1991) Intestinal absorption of magnesium from food and supplements. J Clin Invest 88(2):396–402. https://doi.org/10.1172/JCI115317
Bae YJ, Kim MH (2013) The effects of mg supplementation in diets with different calcium levels on the bone status and bone metabolism in growing female rats. Biol Trace Elem Res 155(3):431–438. https://doi.org/10.1007/s12011-013-9798-1
Zhang Y, Lai WP, Leung PC, Wu CF, Yao XS, Wong MS (2006) Effects of fructus ligustri lucidi extract on bone turnover and calcium balance in ovariectomized rats. Biol Pharm Bull 29(2):291–296. https://doi.org/10.1016/j.atherosclerosis.2005.07.029
Lee YS, Choi CW, Kim JJ, Ganapathi A, Udayakumar R, Kim SC (2009) Determination of mineral content in methanolic safflower (Carthamus tinctorius L.) seed extract and its effect on osteoblast markers. Int J Mol Sci 10:292–305. https://doi.org/10.3390/ijms10010292
Figueroa-Pérez MG, Pérez-Ramírez IF, Paredes-López O, Mondragón-Jacobo C, Reynoso-Camacho R (2016) Phytochemical composition and in vitro analysis of nopal ( O. ficus-indica ) cladodes at different stages of maturity. Int J Food Prop 2:912–1742. https://doi.org/10.1080/10942912.2016.1206126
Hale LV, Galvin RJ, Risteli J, Ma YL, Harvey AK, Yang X, Cain RL, Zeng Q, Frolik CA, Sato M, SChmidt AL, Geiser AG (2007) PINP: a serum biomarker of bone formation in the rat. Bone 40:1103–1109. https://doi.org/10.1016/j.bone.2006.11.027
Weaver CM, Alekel DL, Ward WE, Ronis MJ (2012) Flavonoid intake and bone health. J Nutr Gerontol Geriatr 31(3):239–253. https://doi.org/10.1080/21551197.2012.698220
Rey C, Combes C, Drouet1 C, Glimcher M (2008) Bone mineral: update on chemical composition and structure. Osteoporos Int 20(12):2155–2167. https://doi.org/10.1007/s00198-009-0860-y.Bone
Acknowledgments
We want to thank M. en I. Alicia del Real-López and Dra. Beatriz Millan Malo for their technical support.
Funding
This work was supported by CONACYT (Ciencia Basica, convocation 2011-02, Project No. 167769). Ezequiel Hernandez Becerra was financially supported by CONACyT Mexico for his postdoctoral position at CFATA-UNAM. This project was financed by PAPIIT-Universidad Nacional Autónoma de Mexico, project number IN112317.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed in studies involving animals were in accordance with the ethical standards of Mexican current regulation (NOM-062-ZOO-1999).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hernandez-Becerra, E., Mendoza-Avila, M., Jiménez-Mendoza, D. et al. Effect of Nopal (Opuntia ficus indica) Consumption at Different Maturity Stages as an Only Calcium Source on Bone Mineral Metabolism in Growing Rats. Biol Trace Elem Res 194, 168–176 (2020). https://doi.org/10.1007/s12011-019-01752-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-019-01752-0