Abstract
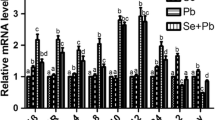
The aim of the present study was to investigate the effect of selenium (Se), polysaccharide of Atractylodes macrocephala Koidz. (PAMK), and the combination of Se and PAMK on the immune response, heat shock protein (HSP) levels under heat stress (HS) condition in chicken spleen. Two hundred chickens were randomly divided into two groups, the HS group and the control (Con) group. Then these chickens were treated with Se (0.3 mg/kg), PAMK (200 mg/kg) alone, and the combination of Se (0.3 mg/kg) and PAMK (200 mg/kg). The antioxidative enzymes, cytokines contents, and expression levels of HSP27 and HSP70 were examined in chicken spleen. The results indicated that HS induced higher levels of TNF-α, IL-4, HSP27, HSP70, and MDA levels but lower level of IFN-γ, IL-2, Gpx, and SOD in spleen (P < 0.05). These responses were ameliorated by the treatment of Se, PAMK alone, and the combination of Se and PAMK (P < 0.05 or not) The results showed that under common condition, Se and PAMK could improve the immune response by enhancing the levels of some cytokines to proper levels; however, under HS condition, Se and PAMK could change the abnormal levels of cytokines and oxidative damages to ameliorate the injury induced by HS. In addition, there existed synergistic effect on the modulation of these biomarkers in chicken spleen between Se and PAMK. So both Se and PAMK play important roles in regulating the immune function in chicken. Considering the synergistic effect on immune regulation of PAMK, this herb deserves further investigation to evaluate its role in improving the effect of traditional immune regulators.




Similar content being viewed by others
References
Geraert PA, Padilha JC, Guillaumin S (1996) Metabolic and endocrine changes induced by chronic heat exposure in broiler chickens: growth performance, body composition and energy retention. British J Nutr 75:195–204
Habibian M, Ghazi S, Moeini MM, Abdolmohammadi A (2013) Effects of dietary selenium and vitamin E on immune response and biological blood parameters of broilers reared under thermoneutral or heat stress conditions. Int J Biomet
Mashaly MM, Hendricks GL 3rd, Kalama MA et al (2004) Effect of heat stress on production parameters and immune responses of commercial laying hens. Poult Sci 83:889–894
Liu LL, He JH, Xie HB et al (2014) Resveratrol induces antioxidant and heat shock protein mRNA expression in response to heat stress in black-boned chickens. Poult Sci 93:54–62
Morrow-Tesch JL, McGlone JJ, Salak-Johnson JL (1994) Heat and social stress effects on pig immune measures. J Anim Sci 72:2599–2609
Starkie RL, Hargreaves M, Rolland J, Febbraio MA (2005) Heat stress, cytokines, and the immune response to exercise. Brain Behav Immun 19:404–412
Van Noort JM, Bsibsi M, Nacken P, Gerritsen WH, Amor S (2012) The link between small heat shock proteins and the immune system. Int J Biochem Cell Biol 44:1670–1679
Lindquist S, Craig EA (1988) The heat-shock proteins. Annu Rev Genet 22:631–677
Bao Y, Wang Q, Liu H, Lin Z (2011) A small HSP gene of bloody clam (Tegillarca granosa) involved in the immune response against Vibrio parahaemolyticus and lipopolysaccharide. Fish Shellfish Immun 30:729–733
Zhao FQ, Zhang ZW, Wang C et al (2013) The role of heat shock proteins in inflammatory injury induced by cold stress in chicken hearts. Cell Stress Chaperon 18:773–783
Ciocca DR, Arrigo AP, Calderwood SK (2013) Heat shock proteins and heat shock factor 1 in carcinogenesis and tumor development: an update. Arch Toxicol 87:19–48
Arthur JR, McKenzie RC, Beckett GJ (2003) Selenium in the immune system. J Nutr 133:1457S–1459S
Rayman MP (2000) The importance of selenium to human health. Lancet 356:233–241
Gan F, Ren F, Chen X et al (2013) Effects of selenium-enriched probiotics on heat shock protein mRNA levels in piglet under heat stress conditions. J Agric Food Chem 61:2385–2391
Mahmoud KZ, Edens FW (2005) Influence of organic selenium on hsp70 response of heat-stressed and enteropathogenic Escherichia coli-challenged broiler chickens (Gallus gallus). Comp Biochem Physiol C Toxicol Pharmacol 141:69–75
Liu LL, Zhang JL, Zhang ZW et al (2014) Protective roles of selenium on nitric oxide-mediated apoptosis of immune organs induced by cadmium in chickens. Biol Trace Elem Res
Zhang ZW, Zhang JL, Gao YH et al (2013) Effect of oxygen free radicals and nitric oxide on apoptosis of immune organ induced by selenium deficiency in chickens. Biometals 26:355–365
Liao X, Lu L, Li S et al (2012) Effects of selenium source and level on growth performance, tissue selenium concentrations, antioxidation, and immune functions of heat-stressed broilers. Biol Trace Elem Res 150:158–165
Li R, Sakwiwatkul K, Yutao L, Hu S (2009) Enhancement of the immune responses to vaccination against foot-and-mouth disease in mice by oral administration of an extract made from Rhizoma Atractylodis Macrocephalae (RAM). Vaccine 27:2094–2098
Li LL, Yin FG, Zhang B et al (2011) Dietary supplementation with Atractylodes macrocephala Koidz polysaccharides ameliorate metabolic status and improve immune function in early-weaned pigs. Livest Sci 142:33–41
Du CY, Choi RC, Zheng KY et al (2013) Yu Ping Feng San, an ancient Chinese herbal decoction containing Astragali Radix, Atractylodis Macrocephalae Rhizoma and Saposhnikoviae Radix, regulates the release of cytokines in murine macrophages. PLoS One 8:e78622
Rotruck JT, Pope AL, Ganther HE et al (1973) Selenium: biochemical role as a component of glutathione peroxidase. Science 179:588–590
Esterbauer H, Cheeseman KH (1990) Determination of aldehydic lipid peroxidation products: malonaldehyde and 4-hydroxynonenal. Methods Enzymol 186:407–421
Yao HD, Wu Q, Zhang ZW et al (2013) Selenoprotein W serves as an antioxidant in chicken myoblasts. Biochim Biophys Acta 1830:3112–3120
Peirson SN, Butler JN, Foster RG (2003) Experimental validation of novel and conventional approaches to quantitative real-time PCR data analysis. Nucleic Acids Res 31:e73
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Ellis RJ (1990) The molecular chaperone concept. Semin Cell Biol 1:1–9
Nathan DB, Heller ED, Perek M (1976) The effect of short heat stress upon leucocyte count, plasma corticosterone level, plasma and leucocyte ascorbic acid content. Br Poult Sci 17:481–485
Puthpongsiriporn U, Scheideler SE, Sell JL, Beck MM (2001) Effects of vitamin E and C supplementation on performance, in vitro lymphocyte proliferation, and antioxidant status of laying hens during heat stress. Poult Sci 80:1190–1200
Lee JC, Lee KY, Son YO et al (2007) Stimulating effects on mouse splenocytes of glycoproteins from the herbal medicine Atractylodes macrocephala Koidz. Phytomedicine 14:390–395
Mahmoud KZ, Edens FW (2003) Influence of selenium sources on age-related and mild heat stress-related changes of blood and liver glutathione redox cycle in broiler chickens (Gallus domesticus). Comp Biochem Physiol Biochem Mole Biol 136:921–934
Lin H, De Vos D, Decuypere E, Buyse J (2008) Dynamic changes in parameters of redox balance after mild heat stress in aged laying hens (Gallus gallus domesticus). Comp Biochem Physiol Toxicol Pharmacol 147:30–35
Acknowledgments
This research was funded by the Aid Program for Science and Technology Innovative Research Team in Higher Educational Institutions of Hunan Province. The project was supported by Hunan Provincial Natural Science Foundation of China (grant no. 12JJ8014), Zhujiang Science and Technology Star of Guangzhou (grant no. 2011 J2200026), Training Program for Outstanding Young Teachers in Higher Education of Guangdong Province (grant no. Yq2013098), and Key Program of Guangdong Technology Plan (grant no. 2012A020200010).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Xu, D., Li, W., Huang, Y. et al. The Effect of Selenium and Polysaccharide of Atractylodes macrocephala Koidz. (PAMK) on Immune Response in Chicken Spleen Under Heat Stress. Biol Trace Elem Res 160, 232–237 (2014). https://doi.org/10.1007/s12011-014-0056-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-014-0056-y