Abstract
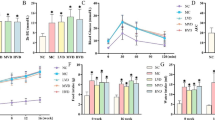
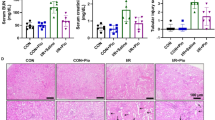
Diabetes-induced chronic kidney diseases are widespread and decrease the quality of life for millions of affected individuals in China. To date, no therapies effectively alleviate these conditions. Triptolide, a traditionally used Chinese medicine, has shown promise in treating renal diseases. Here, the study aimed to decipher the exact mechanism by which it functions. It was hypothesized that triptolide might prevent the epithelial-mesenchymal transition (EMT) of podocytes by activating the kindlin-2 and TGF-β/Smad pathways. Triptolide or telmisartan was intragastrically administered to 9-week-old db/db and dm/dm mice with diabetic nephropathy (DN) for 12 weeks. In addition, biochemical parameters and body weight were detected. WT-1, nephrin, podocin, E-cadherin, and α-SMA were determined by immunohistochemistry in the renal tissues of treated mice. Protein and mRNA expression of podocyte EMT markers, kindlin-2 and TGF-β/Smad, were analyzed to elucidate the underlying mechanism. It was observed that triptolide treatment relieved structural injuries and functional variations in diabetic mice. It also increased the protein and mRNA levels of nephrin, podocin, and E-cadherin and decreased the expression of α-SMA in diabetic mice. The protein and mRNA expressions of TGF-β1, p-SMAD3, and kindlin-2 decreased in diabetic kidneys following triptolide treatment. The findings demonstrated that triptolide might protect podocytes during DN by inhibiting podocyte EMT through inactivation of kindlin-2, combined with the downregulation of P-SMAD3 in the TGF-β/Smad signaling pathway.






Similar content being viewed by others
Data Availability
Not applicable.
References
Zhang, L., Long, J., Jiang, W., Shi, Y., & Wang, H. (2016). Trends in chronic kidney disease in China. New England Journal of Medicine, 375, 905–906.
Adeshara, K. A., Diwan, A. G., & Tupe, R. S. (2015). Diabetes and complications: Cellular signaling pathways, current understanding and targeted therapies. Current Drug Targets, 17(11), 1309–28.
Maezawa, Y., Takemoto, M., & Yokote, K. (2015). Cell biology of diabetic nephropathy: Roles of endothelial cells, tubulointerstitial cells and podocytes. Journal of Diabetes Investigation, 6, 3–15.
Liu, Y. (2004). Epithelial to mesenchymal transition in renal fibrogenesis: Pathologic significance, molecular mechanism, and therapeutic intervention. Journal of the American Society of Nephrology, 15, 1–12.
Haoran, D., Qingquan, L., & Baoli, L. (2017). Research progress on mechanism of podocyte depletion in diabetic nephropathy. Journal of Diabetes Research, 2017, 1–10.
Lu, Q., Ji, X. J., Zhou, Y. X., Yao, X. Q., Liu, Y. Q., Zhang, F., et al. (2015). Quercetin inhibits the mTORC1/p70S6K signaling-mediated renal tubular epithelial–mesenchymal transition and renal fibrosis in diabetic nephropathy. Pharmacological Research, 99, 237–47.
Ivonne, L., & Gunter, W. (2015). Epithelial-to-mesenchymal transition in diabetic nephropathy: Fact or fiction? Cells, 4, 631–52.
Liu, L., Fu, W., Xu, J., Shao, L., & Wang, Y. (2015). Effect of BMP7 on podocyte transdifferentiation and Smad7 expression induced by hyperglycemia. Clinical Nephrology, 84, 95.
Dai, H. Y., Zheng, M., Tang, R. N., Ni, J., Ma, K. L., Li, Q., et al. (2011). Effects of angiotensin receptor blocker on phenotypic alterations of podocytes in early diabetic nephropathy. The American Journal of the Medical Sciences, 341, 207–214.
Haiyan, X., Xu, W., Mingming, L., & Xueyuan, H. (2017). Tangzhiqing granules alleviate podocyte epithelial-mesenchymal transition in kidney of diabetic rats. Evidence-Based Complementray and Alternative Medicine, 2017, 1–9.
Qu, H., Tu, Y., Shi, X., Larjava, H., Saleem, M. A., Shattil, S. J., et al. (2011). Kindlin-2 regulates podocyte adhesion and fibronectin matrix deposition through interactions with phosphoinositides and integrins. Journal of Cell Science, 124, 879–891.
Wei, X., Xia, Y., Li, F., Tang, Y., Nie, J., Liu, Y., et al. (2013). Kindlin-2 mediates activation of TGF-β/Smad signaling and renal fibrosis. Journal of the American Society of Nephrology Jasn, 24, 1387–1398.
Ge, Y., Xie, H., Li, S., Jin, B., & Liu, Z. (2013). Treatment of diabetic nephropathy with Tripterygium wilfordii Hook F extract: A prospective, randomized, controlled clinical trial. Journal of Translational Medicine, 11, 134.
Liu, S., Li, X., Li, H., Liang, Q., & Chen, J. (2015). Comparison of tripterygium wilfordii multiglycosides and tacrolimus in the treatment of idiopathic membranous nephropathy: A prospective cohort study. BMC Nephrology, 16, 200.
Hao, L., Zhi-Hong, L., & Zhao-Hong, C. (2000). Triptolide: A potent inhibitor of NF-kappa B in T-lymphocytes. Acta Pharmacologica Sinica, 21, 782–786.
Qiu, D., & Kao, D. P. N. (2003). Immunosuppressive and anti-inflammatory mechanisms of triptolide, the principal active diterpenoid from the Chinese medicinal herb Tripterygium wilfordii Hook. f. Drugs in R & D, 4, 1–18.
Chen, Z. H., Qin, W. S., Zeng, C. H., Zheng, C. X., Hong, Y. M., Lu, Y. Z., et al. (2010). Triptolide reduces proteinuria in experimental membranous nephropathy and protects against C5b-9-induced podocyte injury in vitro. Kidney International, 77, 974–988.
Han, R., Rostami-Yazdi, M., Gerdes, S., & Mrowietz, U. (2012). Triptolide in the treatment of psoriasis and other immune-mediated inflammatory diseases. British Journal of Clinical Pharmacology, 74, 424–436.
Zheng, C. X., Chen, Z. H., Zeng, C. H., Qin, W. S., Li, L. S., & Liu, Z. H. (2008). Triptolide protects podocytes from puromycin aminonucleoside induced injury in vivo and in vitro. Kidney International, 74, 596–612.
Gao, Q., Shen, W., Qin, W., Zheng, C., Zhang, M., Zeng, C., et al. (2010). Treatment of db/db diabetic mice with triptolide: A novel therapy for diabetic nephropathy. Nephrology Dialysis Transplantation, 25(11), 3539–47.
Yang, Q., Sun, M., Chen, Y., Lu, Y., & Wang, J. (2017). Triptolide protects podocytes from TGF-β-induced injury by preventing miR-30 downregulation. American Journal of Translational Research, 9, 5150.
White, K. E., & Bilous, R. W. (2004). Estimation of podocyte number: A comparison of methods. Kidney International, 66, 663–667.
Pei, W. Y., Yang, C. H., & Zhang, X. L. (2016). Effects and mechanism of Tripterygium wilfordii on chronic glomerulo nephritis. Genetics and Molecular Research: GMR, 15.
Liu, Y. (2010). New insights into epithelial-mesenchymal transition in kidney fibrosis. Journal of the American Society of Nephrology Jasn, 21, 212.
Kang, Y. S., Li, Y., Dai, C., Kiss, L. P., Wu, C., & Liu, Y. (2010). Inhibition of integrin-linked kinase blocks podocyte epithelial–mesenchymal transition and ameliorates proteinuria. Kidney International, 78, 363–373.
Jim, B., Ghanta, M., Qipo, A., Fan, Y., Chuang, P. Y., Cohen, H. W., et al. (2012). Dysregulated nephrin in diabetic nephropathy of type 2 diabetes: A cross sectional study. PLoS One, 7, e36041.
Gerke, P. (2005). NEPH2 is located at the glomerular slit diaphragm, interacts with nephrin and is cleaved from podocytes by metalloproteinases. Journal of the American Society of Nephrology Jasn, 16, 1693.
Koop, K. (2003). Expression of podocyte-associated molecules in acquired human kidney diseases. Journal of the American Society of Nephrology, 14, 2063–2071.
Zeisberg, M., & Neilson, E. G. (2009). Biomarkers for epithelial-mesenchymal transitions. The Journal of Clinical Investigation, 119, 1429–1437.
Jinde, K., Nikolic-Paterson, D. J., Huang, X. R., Sakai, H., Kurokawa, K., Atkins, R. C., et al. (2001). Tubular phenotypic change in progressive tubulointerstitial fibrosis in human glomerulonephritis. American Journal of Kidney Diseases, 38, 761–769.
Loeffler, I., & Wolf, G. (2014). Transforming growth factor-β and the progression of renal disease. Nephrology Dialysis Transplantation Official Publication of the European Dialysis & Transplant Association European Renal Association, 29(Suppl 1), i37–i45.
Funding
This study was financially supported by the Huzhou Municipal Science and Technology Bureau Public Welfare Application Research Project (grant No. 2017GY23, 2020GY17); Zhejiang Medical and Health Science and Technology Project (grant No. 2021KY1093); Zhejiang Science and Technology Planning Project (grant No. 2017C37106); Zhejiang Medical and Health Science and Technology Project (Grant No. 2016KYA172); and Zhejiang Medical and Health Science and Technology Project (Grant No. 2017KY639).
Author information
Authors and Affiliations
Contributions
Authors contributed equally.
Corresponding author
Ethics declarations
Ethical Approval
Not applicable.
Consent to Participate
All authors have their consent to participate.
Consent to Publish
All authors have their consent to publish their work.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ren, L., Wan, R., Chen, Z. et al. Triptolide Alleviates Podocyte Epithelial-Mesenchymal Transition via Kindlin-2 and EMT-Related TGF-β/Smad Signaling Pathway in Diabetic Kidney Disease. Appl Biochem Biotechnol 194, 1000–1012 (2022). https://doi.org/10.1007/s12010-021-03661-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-021-03661-2