Abstract
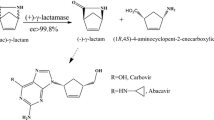
A thermostable formamidase from the aerobic hyperthermophilic archaeon Aeropyrum pernix was revealed a novel type II (+)-γ-lactamase. This type II (+)-γ-lactamase is only composed of 377 amino acid residues, in contrast to another thermostable (+)-γ-lactamase from Sulfolobus solfataricus with 504 amino acid residues (type I). It is interesting that there are low identities between these two (+)-γ-lactamases, and herein, we further proved that at least two types of (+)-γ-lactamases exist in nature due to enzyme promiscuity. The gene of this thermostable (+)-γ-lactamase was cloned, functionally expressed in Escherichia coli BL21, and purified by a simple yet effective heat treatment method. It showed incredible thermostability, retaining 100 % of its activity after 12 h at 100 °C. The optimum temperature for this enzyme was supposed to be more than 100 °C, and the optimum pH for this enzyme was about 9.0. The lactamase maintained its activity in the presence of most metal ions, except for Cu2+. This thermo- and alkaline-tolerant (+)-γ-lactamase presents promising properties for the industrial application. Specifically, it could be used for the production of chirally pure (−)-γ-lactam for the synthesis of well-known carbocyclic nucleosides like abacavir and peramivir. The optical purity of the chiral product reached over 97 % enantiomeric excess.







Similar content being viewed by others
References
Arnold, K., Bordoli, L., Kopp, J., & Schwede, T. (2006). Bioinformatics, 22, 195–201.
Cilia, E., Fabbri, A., Uriani, M., Scialdone, G. G., & Ammendola, S. (2005). FEBS Journal, 272, 4716–4724.
Connelly, S., Line, K., Isupov, M. N., & Littlechild, J. A. (2005). Organic & Biomolecular Chemistry, 3, 3260–3262.
d’Abusco, A., Ammendola, S., Scandurra, R., & Politi, L. (2001). Extremophiles, 5, 183–192.
Demirjian, D. C., Morı́s-Varas, F., & Cassidy, C. S. (2001). Current Opinion in Chemical Biology, 5, 144–151.
Egorova, K., & Antranikian, G. (2005). Current Opinion in Microbiology, 8, 649–655.
Gao, R., Feng, Y., Ishikawa, K., Ishida, H., Ando, S., Kosugi, Y., & Cao, S. (2003). Journal of Molecular Catalysis B: Enzymatic, 24–25, 1–8.
Hickey, A. M., Ngamsom, B., Wiles, C., Greenway, G. M., Watts, P., & Littlechild, J. A. (2009). Biotechnology Journal, 4, 510–516.
Hirakawa, H., Kamiya, N., Kawarabayashi, Y., & Nagamune, T. (2004). Journal of Bioscience and Bioengineering, 97, 202–206.
Ohtaki, A., Murata, K., Sato, Y., Noguchi, K., Miyatake, H., Dohmae, N., Yamada, K., Yohda, M., & Odaka, M. (2010). Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics, 1804, 184–192.
Patel, R. N. (2008). Coordination Chemistry Reviews, 252, 659–701.
Qin, X., Wang, J., & Zheng, G. (2010). Applied Biochemistry and Biotechnology, 162, 2345–2354.
Rasor, J. P., & Voss, E. (2001). Applied Catalysis A: General, 221, 145–158.
Sako, Y., Nomura, N., Uchida, A., Ishida, Y., Morii, H., Koga, Y., Hoaki, T., & Maruyama, T. (1996). International Journal of Systematic Bacteriology, 46, 1070–1077.
Staszewski S, K. P. M. J. & et al. (2001). JAMA: The Journal of the American Medical Association, 285, 1155–1163.
Taylor, S. J. C., Brown, R. C., Keene, P. A., & Taylor, I. N. (1999). Bioorganic & Medicinal Chemistry, 7, 2163–2168.
Taylor, S. J. C., McCague, R., Wisdom, R., Lee, C., Dickson, K., Ruecroft, G., O’Brien, F., Littlechild, J., Bevan, J., Roberts, S. M., & Evans, C. T. (1993). Tetrahedron: Asymmetry, 4, 1117–1128.
Toogood, H., Hollingsworth, E., Brown, R., Taylor, I., Taylor, S., McCague, R., & Littlechild, J. (2002). Extremophiles, 6, 111–122.
Toogood, H. S., Brown, R. C., Line, K., Keene, P. A., Taylor, S. J. C., McCague, R., & Littlechild, J. A. (2004). Tetrahedron, 60, 711–716.
Toogood, H. S., Taylor, I. N., Brown, R. C., Taylor, S. J. C., McCague, R., & Littlechild, J. A. (2002). Biocatalysis and Biotransformation, 20, 241–249.
Torres, L. L., Schlie, Schmidt, M., Silva-Martin, N., Hermoso, J. A., Berenguer, J., Bornscheuer, U. T. & Hidalgo, A. (2012). Organic & Biomolecular Chemistry, 10, 3388–3392.
Wang, J., Zhang, X., Min, C., Wu, S., & Zheng, G. (2011). Process Biochemistry, 46, 81–87.
Willies, S., Isupov, M., & Littlechild, J. (2010). Environmental Technology, 31, 1159–1167.
Yu, Y., Garg, S., Yu, P. A., Kim, H.-J., Patel, A., Merlin, T., Redd, S., & Uyeki, T. M. (2012). Clinical Infectious Diseases, 55, 8–15.
Zhu, S., Gong, C., Ren, L., Li, X., Song, D., & Zheng, G. (2013). Applied Microbiology and Biotechnology, 97, 837–845.
Zhu, S., Gong, C., Song, D., Gao, S., & Zheng, G. (2012). Applied and Environmental Microbiology, 78, 7492–7495.
Zhu, S., Song, D., Gong, C., Tang, P., Li, X., Wang, J., & Zheng, G. (2013). Applied Microbiology and Biotechnology, 97, 6769–6778.
Author information
Authors and Affiliations
Corresponding author
Additional information
Lu Ren and Shaozhou Zhu contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 796 kb)
Rights and permissions
About this article
Cite this article
Ren, L., Zhu, S., Shi, Y. et al. Enantioselective Resolution of γ-Lactam by a Novel Thermostable Type II (+)-γ-Lactamase from the Hyperthermophilic Archaeon Aeropyrum pernix . Appl Biochem Biotechnol 176, 170–184 (2015). https://doi.org/10.1007/s12010-015-1565-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1565-7