Abstract
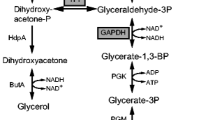
Ethanol was found as the major by-product in lactate fermentation by Rhizopus oryzae. Several methods have been conducted in order to limit ethanol formation, thus increasing the lactate yield. The direct way to suppress ethanol production can be done by inhibition of the responsible enzymes in the related pathway. Pyruvate decarboxylase (PDC) and alcohol dehydrogenase (ADH) are responsible for ethanol production in R. oryzae. Shunting the ethanol production pathway by targeting at PDC was attempted in this study. Three compounds including 4-methylpyrazole, glyoxylic acid, and 3-hydroxypyruvate with the in vitro reversible inhibitory effect on PDC were selected from the literature and were used to regulate the living cell of R. oryzae during the fermentation. The results show that 0.1 mM 4-methylpyrazole of which the structure resembled a thiazolium ring in thiamine diphosphate, PDC cofactor, and 1.0 μm 3-hydroxypyruvate, pyruvate analog, effectively hampered ethanol production. Further observation on the enzyme expression indicated that these two regulators not only targeted PDC but also caused changes in ADH and lactate dehydrogenase (LDH) activities. This was perhaps due to the living cell of R. oryzae that responded to the presence of the regulators to balance the pyruvate flux and subsequently maintain its metabolic activities.






Similar content being viewed by others
References
Skory, D. C. (2000). Applied and Environmental Microbiology, 66, 2343–2348.
Skory, D. C. (2003). Current Microbiology, 47, 59–64.
Papagianni, M. (2004). Biotechnology Advances, 22, 189–259.
Thitiprasert, S., Sooksai, S., & Thongchul, N. (2011). Applied Biochemistry and Biotechnology, 164, 1305–1322.
Thitiprasert, S., Sooksai, S., & Thongchul, N. (2014). Applied Biochemistry and Biotechnology, 172, 1673–1686.
Acar, S., Yucel, M., & Hamamci, H. (2007). Enzyme and Microbial Technology, 40, 675–682.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Journal of Biological Chemistry, 193, 265–275.
Pritchard, G. G. (1971). Biochimica et Biophysica Acta - Enzymology, 250, 25–34.
Chotisubha-anandha, N., Thitiprasert, S., Tolieng, V., & Thongchul, N. (2011). Bioprocess and Biosystems Engineering, 34, 163–172.
Park, E. Y., Kosakai, Y., & Okabe, M. (1998). Biotechnology Progress, 14, 699–704.
Tay, A., & Yang, S. T. (2002). Biotechnology and Bioengineering, 80, 1–12.
Kutter, S., Spinka, M., Koch, M. H. J., & Konig, S. (2007). Protein Journal, 26, 585–591.
Kutter, S., Wille, G., Relle, S., Weiss, M. S., Hübner, G., & Konig, S. (2006). FEBS Journal, 273, 4199–4209.
Furey, W., Arjunan, P., Chen, L., Sax, M., Guo, F., & Jordan, F. (1998). Biochimica et Biophysica Acta - Protein Structure and Molecular Enzymology, 1385, 253–270.
Acknowledgments
This work has been financially supported by the Ratchadaphiseksomphot Endowment Fund (RES560530182-AM). The authors also thank the Graduate School of Chulalongkorn University for the Partial Support from Ratchadaphiseksomphot Endowment Fund via the 90th anniversary of Chulalongkorn University Fund, National Research University Project Office of Higher Education Commission (WCU-044-EN-57), and the National Research Council of Thailand via the annual government statement of expenditure.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thitiprasert, S., Songserm, P., Boonkong, W. et al. Manipulating Pyruvate Decarboxylase by Addition of Enzyme Regulators during Fermentation of Rhizopus oryzae to Enhance Lactic Acid Production. Appl Biochem Biotechnol 174, 1795–1809 (2014). https://doi.org/10.1007/s12010-014-1155-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-1155-0