Abstract
Background
To determine the importance of MRI abnormalities in metal-on-metal (MoM) bearings, it is important to understand the baseline features of this diagnostic tool in conventional metal-on-polyethylene (MoP) bearings.
Questions/purposes
What are the frequency, size, and types of MRI-documented adverse local tissue reactions in asymptomatic patients with MoP bearings?
Methods
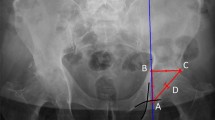
We recruited 50 patients 5 years after a MoP total hip arthroplasty from a pool of patients in our joint registry who had a Harris hip score of > 90. To be included, patients had to be without pain and have adequate radiographs. Our data set included 50 asymptomatic patients with MoP bearings who underwent a metal artifact reduction sequence MRI.
Results
MRI abnormalities were seen in 14 of 50 (28%) asymptomatic patients who were studied. Thirteen of the 14 abnormalities were cystic thin-walled lesions with a mean of 18 cm3 (range, 1–79 cm3).
Conclusions
MRI abnormalities were noted in nearly one-third of asymptomatic patients with MoP bearings. Decisions concerning revision of MoM bearings should not be based on isolated MRI findings because MRI abnormalities are commonly seen regardless of bearing type. A number of factors should determine the need for intervention including pain, mechanical symptoms, abductor weakness, component type, component position, and ion levels as well as MRI findings.
Level of Evidence
Level IV, therapeutic study. See Guidelines for Authors for a complete description of levels of evidence.

Similar content being viewed by others
References
Almousa SA, Greidanus NV, Masri BA, Duncan CP, Garbuz DS. The natural history of inflammatory pseudotumors in asymptomatic patients after metal-on-metal hip arthroplasty. Clin Orthop Relat Res. 2013;471:3814–3821.
Bisseling P, Tan T, Lu Z, Campbell PA, Susante JL. The absence of a metal-on-metal bearing does not preclude the formation of a destructive pseudotumor in the hip—a case report. Acta Orthop. 2013;84:437–441.
Cooper HJ, Ranawat AS, Potter HG, Foo LF, Koob TW, Ranawat CS. Early reactive synovitis and osteolysis after total hip arthroplasty. Clin Orthop Relat Res. 2010;468:3278–3285.
Fehring TK, Odum S, Sproul R, Weathersbee J. High frequency of adverse local tissue reactions in asymptomatic patients with metal-on-metal THA. Clin Orthop Relat Res. 2014;472:517–522.
Hart AJ, Satchithananda K, Liddle AD, Sabah SA, McRobbie D, Henckel J, Cobb JP, Skinner JA, Mitchell AW. Pseudotumors in association with well functioning metal-on-metal hip prostheses. J Bone Joint Surg Am. 2012;94:317–325.
Hsu AR, Gross CE, Levine BR. Pseudotumor from modular neck corrosion after ceramic-on-polyethylene total hip arthroplasty. Am J Orthop (Belle Mead NJ). 2012;41:422–426.
Lin KH, Lo NN. Failure of polyethylene in total hip arthroplasty presenting as a pelvic mass. J Arthroplasty. 2009;24:1144.e13–1144.e15.
Mak KH, Wong TK, Poddar NC. Wear debris from total hip arthroplasty presenting as an intrapelvic mass. J Arthroplasty. 2001;16:674–676.
Mao X, Tay GH, Godbolt DB, Crawford RW. Pseudotumor in a well-fixed metal-on-polyethylene uncemented hip arthroplasty. J Arthroplasty. 2012;27:493.e13–493.e17.
Messana J, Adelani M, Goodman SB. Case report: pseudotumor associated with corrosion of a femoral component with a modular neck and a ceramic-on-polyethylene bearing. J Long Term Eff Med Implants. 2014;24:1–5.
Potter HG, Nestor BJ, Sofka CM, Ho ST, Peters LE, Salvati EA. Magnetic resonance imaging after total hip arthroplasty: evaluation of periprosthetic soft tissue. J Bone Joint Surg Am. 2004;86:1947–1954.
Santavirta S, Hoikka V, Eskola A, Konttinen YT, Paavilainen T, Tallroth K. Aggressive granulomatous lesions in cementless total hip arthroplasty. J Bone Joint Surg Br. 1990;72:980–984.
Scully WF, Teeny SM. Pseudotumor associated with metal-on-polyethylene total hip arthroplasty. Orthopedics. 2013;36:e666–670.
Shilt JS, Rozencwaig R, Wilson MR. Pelvic mass secondary to polyethylene and titanium alloy wear debris resulting in recurrent deep vein thrombosis. J Arthroplasty. 1997;12:946–949.
Svensson O, Mathiesen EB, Reinholt FP, Blomgren G. Formation of a fulminant soft-tissue pseudotumor after uncemented hip arthroplasty. A case report. J Bone Joint Surg Am. 1988;70:1238–1242.
Tallroth K, Eskola A, Santavirta S, Konttinen YT, Lindholm TS. Aggressive granulomatous lesions after hip arthroplasty. J Bone Joint Surg Br. 1989;71:571–575.
Walsh AJ, Nikolaou VS, Antoniou J. Inflammatory pseudotumor complicating metal-on-highly cross-linked polyethylene total hip arthroplasty. J Arthroplasty. 2012;27:324.e5–324.e8.
Wang JW, Lin CC. Pelvic mass caused by polyethylene wear after uncemented total hip arthroplasty. J Arthroplasty. 1996;11:626–628.
White LM, Kim JK, Mehta M, Merchant N, Schweitzer ME, Morrison WB, Hutchison CR, Gross AE. Complications of total hip arthroplasty: MR imaging-initial experience. Radiology. 2000;215:254–262.
Williams DH, Greidanus NV, Masri BA, Duncan CP, Garbuz DS. Prevalence of pseudotumor in asymptomatic patients after metal-on-metal hip arthroplasty. J Bone Joint Surg Am. 2011;93:2164–2171.
Acknowledgments
We thank OrthoCarolina Research Institute, Inc. Special thanks to Rebecca Haug for her hard work and dedication to the study and the study patients.
Author information
Authors and Affiliations
Corresponding author
Additional information
The institution of one or more authors (TKF) has received, during the study period, funding from Carolinas Healthcare Foundation, Winkler Orthopaedic Fellowship Fund. One of the authors certifies that he (TKF), or a member of his or her immediate family, has or may receive payments or benefits, during the study period, an amount of USD 100,001 to USD 1,000,000 from DePuy Orthopaedics (Warsaw, IN, USA).
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research ® editors and board members are on file with the publication and can be viewed on request.
Clinical Orthopaedics and Related Research ® neither advocates nor endorses the use of any treatment, drug, or device. Readers are encouraged to always seek additional information, including FDA-approval status, of any drug or device prior to clinical use.
Each author certifies that his or her institution approved the human protocol for this investigation, that the study was approved by an institutional review board, that all investigations were conducted in conformity with ethical principles of research, and that informed consent for participation in the study was obtained.
This work was performed at OrthoCarolina Hip and Knee Center, Charlotte, NC, USA.
About this article
Cite this article
Fehring, T.K., Fehring, K. & Odum, S.M. Metal Artifact Reduction Sequence MRI Abnormalities Occur in Metal-on-polyethylene Hips. Clin Orthop Relat Res 473, 574–580 (2015). https://doi.org/10.1007/s11999-014-3873-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11999-014-3873-6