Abstract
Purpose of Review
Enhanced recovery pathways are a multimodal, multidisciplinary approach to patient care that aims to reduce the surgical stress response and maintain organ function resulting in faster recovery and improved outcomes.
Recent Findings
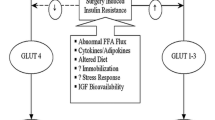
A PubMed literature search was performed for articles that included the terms of metabolic surgical stress response considerations to improve postoperative recovery. The surgical stress response occurs due to direct and indirect injuries during surgery. Direct surgical injury can result from the dissection, retraction, resection, and/or manipulation of tissues, while indirect injury is secondary to events including hypotension, blood loss, and microvascular changes. Greater degrees of tissue injury will lead to higher levels of inflammatory mediator and cytokine release, which ultimately drives immunologic, metabolic, and hormonal processes in the body resulting in the stress response. These processes lead to altered glucose metabolism, protein catabolism, and hormonal dysregulation among other things, all which can impede recovery and increase morbidity. Fluid therapy has a direct effect on intravascular volume and cardiac output with a resultant effect on oxygen and nutrient delivery, so a balance must be maintained without excessively loading the patient with water and salt. All in all, attenuation of the surgical stress response and maintaining organ and thus whole-body homeostasis through enhanced recovery protocols can speed recovery and reduce complications.
Summary
The present investigation summarizes the clinical application of enhanced recovery pathways, and we will highlight the key elements that characterize the metabolic surgical stress response and improved postoperative recovery.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
•• Ljungqvist O, Scott M, Fearon KC. Enhanced recovery after surgery. JAMA Surg. 2017;152:292–8. https://doi.org/10.1001/jamasurg.2016.4952 .Excellent review of literature related to enhanced recovery after surgery.
•• Scott MJ, Urman RD. Concepts in physiology and pathophysiology of enhanced recovery after surgery. Int Anesthesiol Clin. 2017;55:38–50. https://doi.org/10.1097/AIA.0000000000000166 Excellent review of concepts in physiology and pathophysiology related to enhanced recovery after surgery.
Horres CR, Adam MA, Sun Z, Thacker JK, Moon RE, Miller TE, et al. Enhanced recovery protocols for colorectal surgery and postoperative renal function: a retrospective review. Perioper Med (London, England). 2017;6:13. https://doi.org/10.1186/s13741-017-0069-0.
Steinthorsdottir KJ, Kehlet H, Aasvang EK. Surgical stress response and the potential role of preoperative glucocorticoids on post-anesthesia care unit recovery. Minerva Anestesiol. 2017;83:1324–31. https://doi.org/10.23736/S0375-9393.17.11878-X.
Notaras AP, Demetriou E, Galvin J, Ben-Menachem E. A cross-sectional study of preoperative medication adherence and early postoperative recovery. J Clin Anesth. 2016;35:129–35. https://doi.org/10.1016/j.jclinane.2016.07.007.
Rasmussen LS, Johnson T, Kuipers HM, Kristensen D, Siersma VD, Vila P, et al. Does anaesthesia cause postoperative cognitive dysfunction? A randomised study of regional versus general anaesthesia in 438 elderly patients. Acta Anaesthesiol Scand. 2003;47:260–6.
Nygren J, Thacker J, Carli F, Fearon KCH, Norderval S, Lobo DN, et al. Guidelines for perioperative care in elective rectal/pelvic surgery: enhanced recovery after surgery (ERAS®) society recommendations. World J Surg. 2013;37:285–305. https://doi.org/10.1007/s00268-012-1787-6.
Hausel J, Nygren J, Lagerkranser M, Hellström PM, Hammarqvist F, Almström C, et al. A carbohydrate-rich drink reduces preoperative discomfort in elective surgery patients. Anesth Analg. 2001;93:1344–50. https://doi.org/10.1097/00132586-200304000-00042.
Henriksen MG, Hessov I, Dela F, Hansen HV, Haraldsted V, Rodt SA. Effects of preoperative oral carbohydrates and peptides on postoperative endocrine response, mobilization, nutrition and muscle function in abdominal surgery. Acta Anaesthesiol Scand. 2003;47:191–9.
Noblett SE, Watson DS, Huong H, Davison B, Hainsworth PJ, Horgan AF. Pre-operative oral carbohydrate loading in colorectal surgery: a randomized controlled trial. Color Dis. 2006;8:563–9. https://doi.org/10.1111/j.1463-1318.2006.00965.x.
Jankowski CJ. Preparing the patient for enhanced recovery after surgery. Int Anesthesiol Clin. 2017;55:12–20. https://doi.org/10.1097/AIA.0000000000000157.
Scott MJ, Miller TE. Pathophysiology of major surgery and the role of enhanced recovery pathways and the anesthesiologist to improve outcomes. Anesthesiol Clin. 2015;33:79–91. https://doi.org/10.1016/j.anclin.2014.11.006.
Feldheiser A, Aziz O, Baldini G, Cox BPBW, Fearon KCH, Feldman LS, et al. Enhanced recovery after surgery (ERAS) for gastrointestinal surgery, part 2: consensus statement for anaesthesia practice. Acta Anaesthesiol Scand. 2016;60:289–334. https://doi.org/10.1111/aas.12651.
Rosenberg TPGJ, Rosenberg J. Vertical compared with transverse incisions in abdominal surgery. Eur J Surg. 2001;167:260–7. https://doi.org/10.1080/110241501300091408.
Seiler CM, Deckert A, Diener MK, Knaebel H-P, Weigand MA, Victor N, et al. Midline versus transverse incision in major abdominal surgery. Ann Surg. 2009;249:913–20. https://doi.org/10.1097/SLA.0b013e3181a77c92.
Stephan RN, Kupper TS, Geha AS, Baue AE, Chaudry IH. Hemorrhage without tissue trauma produces immunosuppression and enhances susceptibility to sepsis. Arch Surg. 1987;122:62–8.
Page AJ, Ejaz A, Spolverato G, Zavadsky T, Grant MC, Galante DJ, et al. Enhanced recovery after surgery protocols for open hepatectomy—physiology, immunomodulation, and implementation. J Gastrointest Surg. 2015;19:387–99. https://doi.org/10.1007/s11605-014-2712-0.
Dowson HM, Bong JJ, Lovell DP, Worthington TR, Karanjia ND, Rockall TA. Reduced adhesion formation following laparoscopicversus open colorectal surgery. Br J Surg. 2008;95:909–14. https://doi.org/10.1002/bjs.6211.
Schwenk W, Haase O, Neudecker JJ, Müller JM. Short term benefits for laparoscopic colorectal resection. Cochrane Database Syst Rev 2005;CD003145. https://doi.org/10.1002/14651858.CD003145.pub2.
Baigrie RJ, Lamont PM, Kwiatkowski D, Dallman MJ, Morris PJ. Systemic cytokine response after major surgery. Br J Surg. 1992;79:757–60.
Watt DG, Horgan PG, McMillan DC. Routine clinical markers of the magnitude of the systemic inflammatory response after elective operation: a systematic review. Surgery. 2015;157:362–80. https://doi.org/10.1016/j.surg.2014.09.009.
Kim TK, Yoon JR. Comparison of the neuroendocrine and inflammatory responses after laparoscopic and abdominal hysterectomy. Korean J Anesthesiol. 2010;59:265–9. https://doi.org/10.4097/kjae.2010.59.4.265.
Jakeways MS, Chadwick SJ, Carli F. A prospective comparison of laparoscopic versus open cholecystectomy. Ann R Coll Surg Engl. 1993;75:142.
Podgoreanu MV, Michelotti GA, Sato Y, Smith MP, Lin S, Morris RW, et al. Differential cardiac gene expression during cardiopulmonary bypass: ischemia-independent upregulation of proinflammatory genes. J Thorac Cardiovasc Surg. 2005;130:330–9. https://doi.org/10.1016/j.jtcvs.2004.11.052.
•• Carli F. Physiologic considerations of enhanced recovery after surgery (ERAS) programs: implications of the stress response. Can J Anesth Can Anesth. 2015;62:110–9. https://doi.org/10.1007/s12630-014-0264-0 Excellent review of concepts in stress pathophysiology related to enhanced recovery after surgery.
Chowdhury AH, Lobo DN. Fluids and gastrointestinal function. Curr Opin Clin Nutr Metab Care. 2011;14:469–76. https://doi.org/10.1097/MCO.0b013e328348c084.
Eberhart LH, Graf J, Morin AM, Stief T, Kalder M, Lattermann R, et al. Randomised controlled trial of the effect of oral premedication with dexamethasone on hyperglycaemic response to abdominal hysterectomy. Eur J Anaesthesiol. 2011;28:195–201. https://doi.org/10.1097/EJA.0b013e32834296b9.
Gandhi GY, Nuttall GA, Abel MD, Mullany CJ, Schaff HV, Williams BA, et al. Intraoperative hyperglycemia and perioperative outcomes in cardiac surgery patients. Mayo Clin Proc. 2005;80:862–6. https://doi.org/10.4065/80.7.862.
Krinsley JS. Association between hyperglycemia and increased hospital mortality in a heterogeneous population of critically ill patients. Mayo Clin Proc. 2003;78:1471–8. https://doi.org/10.4065/78.12.1471.
Umpierrez GE, Isaacs SD, Bazargan N, You X, Thaler LM, Kitabchi AE. Hyperglycemia: an independent marker of in-hospital mortality in patients with undiagnosed diabetes. J Clin Endocrinol Metab. 2002;87:978–82. https://doi.org/10.1210/jcem.87.3.8341.
Deane AM, Horowitz M. Dysglycaemia in the critically ill - significance and management. Diabetes Obes Metab. 2013;15:792–801. https://doi.org/10.1111/dom.12078.
Sato H, Carvalho G, Sato T, Lattermann R, Matsukawa T, Schricker T. The association of preoperative glycemic control, intraoperative insulin sensitivity, and outcomes after cardiac surgery. J Clin Endocrinol Metab. 2010;95:4338–44. https://doi.org/10.1210/jc.2010-0135.
Kinney JM, Elwyn DH. Protein metabolism and injury. Annu Rev Nutr. 1983;3:433–66. https://doi.org/10.1146/annurev.nu.03.070183.002245.
Schricker T, Gougeon R, Eberhart L, Wykes L, Mazza L, Carvalho G, et al. Type 2 diabetes mellitus and the catabolic response to surgery. Anesthesiology. 2005;102:320–6.
Schricker T, Lattermann R. Perioperative catabolism. Can J Anesth. 2015;62:182–93. https://doi.org/10.1007/s12630-014-0274-y.
Thorell A, Loftenius A, Andersson B, Ljungqvist O. Postoperative insulin resistance and circulating concentrations of stress hormones and cytokines. Clin Nutr. 1996;15:75–9.
Thorell A, Nygren J, Ljungqvist O. Insulin resistance: a marker of surgical stress. Curr Opin Clin Nutr Metab Care. 1999;2:69–78.
Downs JH, Haffejee A. Nutritional assessment in the critically ill. Curr Opin Clin Nutr Metab Care. 1998;1:275–9.
Gustafsson UO, Scott MJ, Schwenk W, Demartines N, Roulin D, Francis N, et al. Guidelines for perioperative care in elective colonic surgery: enhanced recovery after surgery (ERAS®) society recommendations. World J Surg. 2013;37:259–84. https://doi.org/10.1007/s00268-012-1772-0.
Crowe PJ, Dennison A, Royle GT. The effect of pre-operative glucose loading on postoperative nitrogen metabolism. Br J Surg. 1984;71:635–7.
Ljungqvist O, Thorell A, Gutniak M, Häggmark T, Efendic S. Glucose infusion instead of preoperative fasting reduces postoperative insulin resistance. J Am Coll Surg. 1994;178:329–36.
Yagci G, Can MF, Ozturk E, Dag B, Ozgurtas T, Cosar A, et al. Effects of preoperative carbohydrate loading on glucose metabolism and gastric contents in patients undergoing moderate surgery: a randomized, controlled trial. Nutrition. 2008;24:212–6. https://doi.org/10.1016/j.nut.2007.11.003.
Hatzakorzian R, Bui H, Carvalho G, Pi Shan WL, Sidhu S, Schricker T. Fasting blood glucose levels in patients presenting for elective surgery. Nutrition. 2011;27:298–301. https://doi.org/10.1016/j.nut.2010.02.003.
Horber FF, Krayer S, Miles J, Cryer P, Rehder K, Haymond MW. Isoflurane and whole body leucine, glucose, and fatty acid metabolism in dogs. Anesthesiology. 1990;73:82–92.
Herndon DN, Hart DW, Wolf SE, Chinkes DL, Wolfe RR. Reversal of catabolism by beta-blockade after severe burns. N Engl J Med. 2001;345:1223–9. https://doi.org/10.1056/NEJMoa010342.
Schricker T, Meterissian S, Wykes L, Eberhart L, Lattermann R, Carli F. Postoperative protein sparing with epidural analgesia and hypocaloric dextrose. Ann Surg. 2004;240:916–21.
NICE-SUGAR Study Investigators, Finfer S, Liu B, Chittock DR, Norton R, Myburgh JA, et al. Hypoglycemia and risk of death in critically ill patients. N Engl J Med. 2012;367:1108–18. https://doi.org/10.1056/NEJMoa1204942.
Van den Berghe G, Wouters P, Weekers F, Verwaest C, Bruyninckx F, Schetz M, et al. Intensive insulin therapy in critically ill patients. N Engl J Med. 2001;345:1359–67. https://doi.org/10.1056/NEJMoa011300.
Askanazi J, Rosenbaum SH, Hyman AI, Silverberg PA, Milic-Emili J, Kinney JM. Respiratory changes induced by the large glucose loads of total parenteral nutrition. JAMA. 1980;243:1444–7.
Boelens PG, Heesakkers FFBM, Luyer MDP, van Barneveld KWY, de Hingh IHJT, Nieuwenhuijzen GAP, et al. Reduction of postoperative ileus by early enteral nutrition in patients undergoing major rectal surgery. Ann Surg. 2014;259:649–55. https://doi.org/10.1097/SLA.0000000000000288.
Levy BF, Scott MJP, Fawcett WJ, Rockall TA. 23-hour-stay laparoscopic colectomy. Dis Colon Rectum. 2009;52:1239–43. https://doi.org/10.1007/DCR.0b013e3181a0b32d.
Brandstrup B, Svendsen PE, Rasmussen M, Belhage B, Rodt SÅ, Hansen B, et al. Which goal for fluid therapy during colorectal surgery is followed by the best outcome: near-maximal stroke volume or zero fluid balance? Br J Anaesth. 2012;109:191–9. https://doi.org/10.1093/bja/aes163.
Bauer AJ, Boeckxstaens GE. Mechanisms of postoperative ileus. Neurogastroenterol Motil. 2004;16:54–60. https://doi.org/10.1111/j.1743-3150.2004.00558.x.
Panchal SJ, Müller-Schwefe P, Wurzelmann JI. Opioid-induced bowel dysfunction: prevalence, pathophysiology and burden. Int J Clin Pract. 2007;61:1181–7. https://doi.org/10.1111/j.1742-1241.2007.01415.x.
Holte K, Kehlet H. Epidural anaesthesia and analgesia - effects on surgical stress responses and implications for postoperative nutrition. Clin Nutr. 2002;21:199–206.
Marret E, Remy C, Bonnet F. Postoperative pain forum group. Meta-analysis of epidural analgesia versus parenteral opioid analgesia after colorectal surgery. Br J Surg. 2007;94:665–73. https://doi.org/10.1002/bjs.5825.
Nelson R, Tse B, Edwards S. Systematic review of prophylactic nasogastric decompression after abdominal operations. Br J Surg. 2005;92:673–80. https://doi.org/10.1002/bjs.5090.
Michell AR. Diuresis and diarrhea: is the gut a misunderstood nephron? Perspect Biol Med. 2000;43:399–405.
Zhuang C-L, Ye X-Z, Zhang C-J, Dong Q-T, Chen B-C, Yu Z. Early versus traditional postoperative oral feeding in patients undergoing elective colorectal surgery: a meta-analysis of randomized clinical trials. Dig Surg. 2013;30:225–32. https://doi.org/10.1159/000353136.
Tan EK, Cornish J, Darzi AW, Tekkis PP. Meta-analysis: alvimopan vs. placebo in the treatment of post-operative ileus. Aliment Pharmacol Ther. 2006;0:061017010431001–??? https://doi.org/10.1111/j.1365-2036.2006.03150.x.
McCarthy GC, Megalla SA, Habib AS. Impact of intravenous lidocaine infusion on postoperative analgesia and recovery from surgery. Drugs. 2010;70:1149–63. https://doi.org/10.2165/10898560-000000000-00000.
Sun Y, Li T, Wang N, Yun Y, Gan TJ. Perioperative systemic lidocaine for postoperative analgesia and recovery after abdominal surgery. Dis Colon Rectum. 2012;55:1183–94. https://doi.org/10.1097/DCR.0b013e318259bcd8.
Saczynski JS, Marcantonio ER, Quach L, Fong TG, Gross A, Inouye SK, et al. Cognitive trajectories after postoperative delirium. N Engl J Med. 2012;367:30–9. https://doi.org/10.1056/NEJMoa1112923.
Lepousé C, Lautner CA, Liu L, Gomis P, Leon A. Emergence delirium in adults in the post-anaesthesia care unit. Br J Anaesth. 2006;96:747–53. https://doi.org/10.1093/bja/ael094.
Degos V, Vacas S, Han Z, van Rooijen N, Gressens P, Su H, et al. Depletion of bone marrow–derived macrophages perturbs the innate immune response to surgery and reduces postoperative memory dysfunction. Anesthesiology. 2013;118:527–36. https://doi.org/10.1097/ALN.0b013e3182834d94.
Rasmussen LS, Steentoft A, Rasmussen H, Kristensen PA, Moller JT. Benzodiazepines and postoperative cognitive dysfunction in the elderly. ISPOCD group. International study of postoperative cognitive dysfunction. Br J Anaesth. 1999;83:585–9.
Disclaimer
The manuscript has been read and approved by all the authors, the requirements for authorship have been met, and each author believes that the manuscript represents honest work.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to the manuscript and are involved in institutional protocols and policies for enhanced recovery pathways.
Corresponding author
Ethics declarations
Conflict of Interest
Erik M. Helander, Michael P. Webb, Bethany Menard, Amit Prabhakar, John Helmstetter, Elyse M. Cornett, and Viet H. Nguyen declare no conflict of interest. Alan Kaye is on the speaker bureau for the Merck and Depomed, Inc. Richard D. Urman received research funding from the Medtronic.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Other Pain
Rights and permissions
About this article
Cite this article
Helander, E.M., Webb, M.P., Menard, B. et al. Metabolic and the Surgical Stress Response Considerations to Improve Postoperative Recovery. Curr Pain Headache Rep 23, 33 (2019). https://doi.org/10.1007/s11916-019-0770-4
Published:
DOI: https://doi.org/10.1007/s11916-019-0770-4