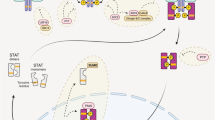
Abstract
Neurofibromatosis type 1 (NF1), also known as von Recklinghausen disease, is a common autosomal dominant genetic disorder affecting approximately 1 in 3000 individuals worldwide. NF1 results from heritable or spontaneous mutations of the NF1 tumor suppressor gene. NF1 encodes the protein neurofibromin, which functions to negatively regulate Ras-activity. Approximately 50 % of NF1 patients develop osteopenia or osteoporosis, resulting in significantly increased rates of long-bone fracture and morbidity. While defective osteoblast bone anabolism has been implicated as a central factor in the pathogenesis of NF1 associated skeletal deficits, recent data suggest that NF1 (Nf1) haploinsufficiency within the hematopoietic compartment, particularly in osteoclasts and myeloid progenitors, plays a pivotal role in engendering NF1 osseous manifestations. In this chapter, we review the latest data from clinical studies and murine models delineating a critical role for hematopoietic compartment, myeloid progenitors of NF1 (Nf1) haploinsufficient and their progeny-osteoclasts, in the pathogenesis of NF1 associated osteopenia/osteoporosis and discuss putative targets for future therapeutics.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance, •• Of major importance
Friedman J et al. Neurofibromatosis: phenotype, natural history, and pathogenesis. 3rd ed. Baltimore: The Johns Hopkins University Press; 1999.
Martin GA et al. The GAP-related domain of the neurofibromatosis type 1 gene product interacts with ras p21. Cell. 1990;63(4):843–9.
Ballester R et al. The NF1 locus encodes a protein functionally related to mammalian GAP and yeast IRA proteins. Cell. 1990;63(4):851–9.
Riccardi VM. Von Recklinghausen neurofibromatosis. N Engl J Med. 1981;305(27):1617–27.
Friedman JM, Birch PH. Type 1 neurofibromatosis: a descriptive analysis of the disorder in 1,728 patients. Am J Med Genet. 1997;70(2):138–43.
Friedman JM. Epidemiology of neurofibromatosis type 1. Am J Med Genet. 1999;89(1):1–6.
Ferner RE, Gutmann DH. International consensus statement on malignant peripheral nerve sheath tumors in neurofibromatosis. Cancer Res. 2002;62(5):1573–7.
Listernick R et al. Optic gliomas in children with neurofibromatosis type 1. J Pediatr. 1989;114(5):788–92.
Listernick R et al. Natural history of optic pathway tumors in children with neurofibromatosis type 1: a longitudinal study. J Pediatr. 1994;125(1):63–6.
Brodeur GM. The NF1 gene in myelopoiesis and childhood myelodysplastic syndromes. N Engl J Med. 1994;330(9):637–9.
Side L et al. Homozygous inactivation of the NF1 gene in bone marrow cells from children with neurofibromatosis type 1 and malignant myeloid disorders. N Engl J Med. 1997;336(24):1713–20.
Emanuel PD et al. The role of monocyte-derived hemopoietic growth factors in the regulation of myeloproliferation in juvenile chronic myelogenous leukemia. Exp Hematol. 1991;19(10):1017–24.
Hyman SL, Shores A, North KN. The nature and frequency of cognitive deficits in children with neurofibromatosis type 1. Neurology. 2005;65(7):1037–44.
Lin AE et al. Cardiovascular malformations and other cardiovascular abnormalities in neurofibromatosis 1. Am J Med Genet. 2000;95(2):108–17.
Fossali E et al. Renovascular disease and hypertension in children with neurofibromatosis. Pediatr Nephrol. 2000;14(8-9):806–10.
Rasmussen SA, Yang Q, Friedman JM. Mortality in neurofibromatosis 1: an analysis using U.S. death certificates. Am J Hum Genet. 2001;68(5):1110–8.
Friedman JM et al. Cardiovascular disease in neurofibromatosis 1: report of the NF1 Cardiovascular Task Force. Genet Med. 2002;4(3):105–11.
Lama G et al. Blood pressure and cardiovascular involvement in children with neurofibromatosis type1. Pediatr Nephrol. 2004;19(4):413–8.
Rea D et al. Cerebral arteriopathy in children with neurofibromatosis type 1. Pediatrics. 2009;124(3):e476–83.
Lammert M et al. Decreased bone mineral density in patients with neurofibromatosis 1. Osteoporos Int. 2005;16(9):1161–6.
Kuorilehto T et al. Decreased bone mineral density and content in neurofibromatosis type 1: lowest local values are located in the load-carrying parts of the body. Osteoporos Int. 2005;16(8):928–36.
Dulai S et al. Decreased bone mineral density in neurofibromatosis type 1: results from a pediatric cohort. J Pediatr Orthop. 2007;27(4):472–5.
Yilmaz K et al. Bone mineral density in children with neurofibromatosis 1. Acta Paediatr. 2007;96(8):1220–2.
Stevenson DA et al. Bone mineral density in children and adolescents with neurofibromatosis type 1. J Pediatr. 2007;150(1):83–8.
Tucker T et al. Bone health and fracture rate in individuals with neurofibromatosis 1 (NF1). J Med Genet. 2009;46(4):259–65.
Crawford AH. Pitfalls of spinal deformities associated with neurofibromatosis in children. Clin Orthop Relat Res. 1989;245:29–42.
Crawford Jr AH, Bagamery N. Osseous manifestations of neurofibromatosis in childhood. J Pediatr Orthop. 1986;6(1):72–88.
Illes T et al. Decreased bone mineral density in neurofibromatosis-1 patients with spinal deformities. Osteoporos Int. 2001;12(10):823–7.
Clementi M et al. Neurofibromatosis type 1 growth charts. Am J Med Genet. 1999;87(4):317–23.
Szudek J, Birch P, Friedman JM. Growth in North American white children with neurofibromatosis 1 (NF1). J Med Genet. 2000;37(12):933–8.
Virdis R et al. Growth and pubertal disorders in neurofibromatosis type 1. J Pediatr Endocrinol Metab. 2003;16 Suppl 2:289–92.
Riccardi VM. Neurofibromatosis: phenotype, natural history, and pathogenesis. 2nd ed. 1992, Baltimore: Johns Hopkins University Press. ix, 498 p.
Alwan S, Tredwell SJ, Friedman JM. Is osseous dysplasia a primary feature of neurofibromatosis 1 (NF1)? Clin Genet. 2005;67(5):378–90.
Elefteriou F et al. Skeletal abnormalities in neurofibromatosis type 1: approaches to therapeutic options. Am J Med Genet A. 2009;149A(10):2327–38.
Young H, Hyman S, North K, Neurofibromatosis 1: clinical review and exceptions to the rules. J Child Neurol. 2002. 17(8):613-21. discussion 627-9, 646-51.
Friedman JM. Neurofibromatosis 1: clinical manifestations and diagnostic criteria. J Child Neurol. 2002. 17(8):548-54. discussion 571-2, 646-51.
Stevenson DA et al. Descriptive analysis of tibial pseudarthrosis in patients with neurofibromatosis 1. Am J Med Genet. 1999;84(5):413–9.
Stevenson DA et al. Evidence of increased bone resorption in neurofibromatosis type 1 using urinary pyridinium crosslink analysis. Pediatr Res. 2008;63(6):697–701.
Heerva E et al. A controlled register based study of 460 neurofibromatosis 1 (NF1) patients: Increased fracture risk in children and adults over 41 years. J Bone Miner Res. 2012. A large cohort clinical study shows that patients with NF1 have increased fracture risk depending on age due to decreased bone mass.
Lakkis MM, Epstein JA. Neurofibromin modulation of ras activity is required for normal endocardial-mesenchymal transformation in the developing heart. Development. 1998;125(22):4359–67.
Wu X et al. Neurofibromin plays a critical role in modulating osteoblast differentiation of mesenchymal stem/progenitor cells. Hum Mol Genet. 2006;15(19):2837–45.
Stevenson DA et al. Double inactivation of NF1 in tibial pseudarthrosis. Am J Hum Genet. 2006;79(1):143–8.
Wu X et al. The haploinsufficient hematopoietic microenvironment is critical to the pathological fracture repair in murine models of neurofibromatosis type 1. PLoS One. 2011;6(9):e24917.
Kolanczyk M et al. Multiple roles for neurofibromin in skeletal development and growth. Hum Mol Genet. 2007;16(8):874–86.
Wang W et al. Mice lacking Nf1 in osteochondroprogenitor cells display skeletal dysplasia similar to patients with neurofibromatosis type I. Hum Mol Genet. 2011;20(20):3910–24.
Li H et al. Ras dependent paracrine secretion of osteopontin by Nf1+/− osteoblasts promote osteoclast activation in a neurofibromatosis type I murine model. Pediatr Res. 2009;65(6):613–8.
Elefteriou F et al. ATF4 mediation of NF1 functions in osteoblast reveals a nutritional basis for congenital skeletal dysplasiae. Cell Metab. 2006;4(6):441–51.
Tang Y et al. TGF-beta1-induced migration of bone mesenchymal stem cells couples bone resorption with formation. Nat Med. 2009;15(7):757–65.
Rhodes SD et al. Hyperactive transforming growth factor-beta1 signaling potentiates skeletal defects in a neurofibromatosis type 1 mouse model. J Bone Miner Res. 2013;28(12):2476–89.
Heerva E et al. Osteoclasts in neurofibromatosis type 1 display enhanced resorption capacity, aberrant morphology, and resistance to serum deprivation. Bone. 2010;47(3):583–90.
Stevenson DA et al. Multiple increased osteoclast functions in individuals with neurofibromatosis type 1. Am J Med Genet A. 2011;155A(5):1050–9.
Yu X et al. Neurofibromin and its inactivation of Ras are prerequisites for osteoblast functioning. Bone. 2005;36(5):793–802.
Yang FC et al. Hyperactivation of p21ras and PI3K cooperate to alter murine and human neurofibromatosis type 1-haploinsufficient osteoclast functions. J Clin Invest. 2006;116(11):2880–91.
Rhodes SD, et al. Hyperactive transforming growth factor-beta1 signaling potentiates skeletal defects in a neurofibromatosis type 1 mouse model. J Bone Miner Res. 2013;28(12):2476–89.
Rhodes SD et al. Nf1 Haploinsufficiency alters myeloid lineage commitment and function, leading to deranged skeletal homeostasis. J Bone Miner Res. 2015;30(10):1840–51. This study demonstrates a critical requirement for Nf1 haploinsufficiency at a more primitive/progenitor stage of myeloid development in perpetuating osteolytic activity.
Alanne MH, et al. Phenotypic characterization of transgenic mice harboring Nf1(+/−) or Nf1(−/−) osteoclasts in otherwise Nf1(+/+) background. J Cell Biochem. 2012;113(6):2136–46.
Chiu WS et al. Transgenic mice that express Cre recombinase in osteoclasts. Genesis. 2004;39(3):178–85.
Bollag G et al. Loss of NF1 results in activation of the Ras signaling pathway and leads to aberrant growth in haematopoietic cells. Nat Genet. 1996;12(2):144–8.
Largaespada DA et al. Nf1 deficiency causes Ras-mediated granulocyte/macrophage colony stimulating factor hypersensitivity and chronic myeloid leukaemia. Nat Genet. 1996;12(2):137–43.
Xu XL et al. Basic research and clinical applications of bisphosphonates in bone disease: what have we learned over the last 40 years? J Transl Med. 2013;11:303.
Heerva E et al. Osteoclasts derived from patients with neurofibromatosis 1 (NF1) display insensitivity to bisphosphonates in vitro. Bone. 2012;50(3):798–803.
Heerva E et al. Follow-up of six patients with neurofibromatosis 1-related osteoporosis treated with alendronate for 23 months. Calcif Tissue Int. 2014;94(6):608–12. This clinical study demonstrated a trend toward increased bone mineral density in five out of six patients with NF1-related osteoporosis following 23 months of alendronate therapy, although the effect size did not reach statistical significance.
Seitz S et al. High bone turnover and accumulation of osteoid in patients with neurofibromatosis 1. Osteoporos Int. 2010;21(1):119–27.
Brunetti-Pierri N et al. Generalized metabolic bone disease in neurofibromatosis type I. Mol Genet Metab. 2008;94(1):105–11.
He Y et al. c-Fms signaling mediates neurofibromatosis type-1 osteoclast gain-in-functions. PLoS One. 2012;7(11):e46900. This study reveals that the M-CSF/c-Fms signaling axis serves as a critical pathway underlying the aberrant functioning Nf1 of haploinsufficient osteoclasts and may provide a potential therapeutic target for treating NF1 associated osteoporosis and osteopenia.
Sharma R et al. Hyperactive Ras/MAPK signaling is critical for tibial nonunion fracture in neurofibromin-deficient mice. Hum Mol Genet. 2013;22(23):4818–28.
El-Hoss J et al. A combination of rhBMP-2 (recombinant human bone morphogenetic protein-2) and MEK (MAP kinase/ERK kinase) inhibitor PD0325901 increases bone formation in a murine model of neurofibromatosis type I pseudarthrosis. J Bone Joint Surg Am. 2014;96(14), e117.
de la Croix Ndong J et al. Asfotase-alpha improves bone growth, mineralization and strength in mouse models of neurofibromatosis type-1. Nat Med. 2014;20(8):904–10.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Steven D. Rhodes and Feng-Chun Yang declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Epidemiology and Pathophysiology
Rights and permissions
About this article
Cite this article
Rhodes, S.D., Yang, FC. Aberrant Myeloid Differentiation Contributes to the Development of Osteoporosis in Neurofibromatosis Type 1. Curr Osteoporos Rep 14, 10–15 (2016). https://doi.org/10.1007/s11914-016-0298-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11914-016-0298-z