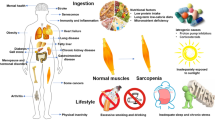
Abstract
Cachexia is a metabolic syndrome driven by inflammation and characterized by loss of muscle with or without loss of fat mass. In cancer cachexia, the tumor burden and host response induce increased inflammation, decreased anabolic tone, and suppressed appetite leading to the clinical presentation of reduced body weight and quality of life (QOL). There is no approved treatment for cancer cachexia, and commonly used nutritional and anti-inflammatory strategies alone have proven ineffective for management of symptoms. Several other pharmacological agents are currently in development and have shown promise as a clinical strategy in early-phase trials. Recently, it has been proposed that multimodal strategies, with an anabolic focus, initiated early in the disease/treatment progression may provide the most therapeutic potential for symptom management. Here we review the data from recent clinical trials in cancer cachexia including pharmacological, exercise, and nutritional interventions.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Coussens LM, Werb Z. Inflammation and cancer. Nature. 2002;420(6917):860–7.
Mantovani A, et al. Cancer-related inflammation. Nature. 2008;454(7203):436–44.
Laviano A, Koverech A, Mari A. Cachexia: clinical features when inflammation drives malnutrition. Proc Nutr Soc. 2015;74(4):348–54.
Fearon K, et al. Definition and classification of cancer cachexia: an international consensus. Lancet Oncol. 2011;12(5):489–95.
Homsi J, Luong D. Symptoms and survival in patients with advanced disease. J Palliat Med. 2007;10(4):904–9.
Utech AE, et al. Predicting survival in cancer patients: the role of cachexia and hormonal, nutritional and inflammatory markers. J Cachexia Sarcopenia Muscle. 2012;3(4):245–51.
• Mondello P, et al. Cancer cachexia syndrome: pathogenesis, diagnosis, and new therapeutic options. Nutr Cancer. 2015;67(1):12–26. This is a comprehensive review of the pathophysiology of cancer cachexia and the diagnosis of cancer anorexia-cachexia syndrome. This review also provides a resourceful synopsis of common treatment strategies and potential future biologic therapeutic options
Ali S, Chen JA, Garcia JM. Clinical development of ghrelin axis-derived molecules for cancer cachexia treatment. Curr Opin Support Palliat Care. 2013;7(4):368–75.
Batista Jr ML, et al. Adipose tissue inflammation and cancer cachexia: possible role of nuclear transcription factors. Cytokine. 2012;57(1):9–16.
Plata-Salaman CR. Brain cytokine production and action in anorexia and cachexia. Cytokine. 2001;15(1):1–3.
Del Fabbro E, et al. Associations among hypogonadism, C-reactive protein, symptom burden, and survival in male cancer patients with cachexia: a preliminary report. J Pain Symptom Manag. 2010;39(6):1016–24.
Garcia JM, et al. Hypogonadism in male patients with cancer. Cancer. 2006;106(12):2583–91.
Quinten C, et al. Baseline quality of life as a prognostic indicator of survival: a meta-analysis of individual patient data from EORTC clinical trials. Lancet Oncol. 2009;10(9):865–71.
Laviano A, Meguid MM. Nutritional issues in cancer management. Nutrition. 1996;12(5):358–71.
August DA, et al. A.S.P.E.N. clinical guidelines: nutrition support therapy during adult anticancer treatment and in hematopoietic cell transplantation. JPEN J Parenter Enteral Nutr. 2009;33(5):472–500.
Baldwin C, et al. Oral nutritional interventions in malnourished patients with cancer: a systematic review and meta-analysis. J Natl Cancer Inst. 2012;104(5):371–85.
Fearon KC, et al. Double-blind, placebo-controlled, randomized study of eicosapentaenoic acid diester in patients with cancer cachexia. J Clin Oncol. 2006;24(21):3401–7.
Cannabis In Cachexia Study, Strasser F, Luftner D, Possinger K, Ernst G, Ruhstaller T, Meissner W, Ko YD, Schnelle M, Reif M, Cerny T, et al. Comparison of orally administered cannabis extract and delta-9-tetrahydrocannabinol in treating patients with cancer-related anorexia-cachexia syndrome: a multicenter, phase III, randomized, double-blind, placebo-controlled clinical trial from the Cannabis-In-Cachexia-Study-Group. J Clin Oncol. 2006;24(21):3394–400.
Jatoi A, et al. Dronabinol versus megestrol acetate versus combination therapy for cancer-associated anorexia: a North Central Cancer Treatment Group study. J Clin Oncol. 2002;20(2):567–73.
Tchekmedyian NS, et al. Megestrol acetate in cancer anorexia and weight loss. Cancer. 1992;69(5):1268–74.
Takaya K, et al. Ghrelin strongly stimulates growth hormone release in humans. J Clin Endocrinol Metab. 2000;85(12):4908–11.
•• Garcia JM, et al. Anamorelin for patients with cancer cachexia: an integrated analysis of two phase 2, randomised, placebo-controlled, double-blind trials. Lancet Oncol. 2015;16(1):108–16. The improvement in appendicular lean mass, maintenance of handgrip strength, and trend for improved quality of life scores compared to placebo over 12 weeks provides suggestive evidence forthe long-term therapeutic potential of anamorelin for cachexia symptom management
• Garcia JM, Friend J, Allen S. Therapeutic potential of anamorelin, a novel, oral ghrelin mimetic, in patients with cancer-related cachexia: a multicenter, randomized, double-blind, crossover, pilot study. Support Care Cancer. 2013;21(1):129–37. This is the first study to evaluate the acute effects of anamorelin on cancer cachexia. Anamorelin was safe and increased body weight and appetite score compared to placebo afterthree days
•• Temel JS, et al. Anamorelin in patients with non-small-cell lung cancer and cachexia (ROMANA 1 and ROMANA 2): results from two randomised, double-blind, phase 3 trials. Lancet Oncol. 2016;17(4):519–31. Two large, international trials were combined to report the 12-week efficacy of anamorelin for cancer cachexia symptom management. Improvements in lean mass and quality of life scores were also reported here, further promoting the therapeutic potential of anamorelin
Penet MF, Bhujwalla ZM. Cancer cachexia, recent advances, and future directions. Cancer J. 2015;21(2):117–22.
Hwang CL, et al. Effects of exercise training on exercise capacity in patients with non-small cell lung cancer receiving targeted therapy. Support Care Cancer. 2012;20(12):3169–77.
Baracos VE, et al. Body composition in patients with non-small cell lung cancer: a contemporary view of cancer cachexia with the use of computed tomography image analysis. Am J Clin Nutr. 2010;91(4):1133S–7S.
Tisdale MJ. Mechanisms of cancer cachexia. Physiol Rev. 2009;89(2):381–410.
Capozzi LC, et al. Patient-reported outcomes, body composition, and nutrition status in patients with head and neck cancer: results from an exploratory randomized controlled exercise trial. Cancer. 2016;122(8):1185–200.
Kraft M, et al. L-Carnitine-supplementation in advanced pancreatic cancer (CARPAN)—a randomized multicentre trial. Nutr J. 2012;11:52.
Kapoor N, et al. A prospective randomized controlled trial to study the impact of a nutrition-sensitive intervention on adult women with cancer cachexia undergoing palliative care in India. Integr Cancer Ther. 2016.
Uster A, et al. Influence of a nutritional intervention on dietary intake and quality of life in cancer patients: a randomized controlled trial. Nutrition. 2013;29(11–12):1342–9.
Bohl CE, et al. Structural basis for accommodation of nonsteroidal ligands in the androgen receptor. J Biol Chem. 2005;280(45):37747–54.
Narayanan R, et al. Steroidal androgens and nonsteroidal, tissue-selective androgen receptor modulator, S-22, regulate androgen receptor function through distinct genomic and nongenomic signaling pathways. Mol Endocrinol. 2008;22(11):2448–65.
• Dobs AS, et al. Effects of enobosarm on muscle wasting and physical function in patients with cancer: a double-blind, randomised controlled phase 2 trial. Lancet Oncol. 2013;14(4):335–45. This is the first study to administer SARM treatment for the management of cancer cachexia. Both enobosarm doses were safe and were similarly efficacious at improving lean mass and stair climb power over roughly 16 weeks of treatment
Dobs AS, et al. SUN-261: prevalence and impact of hypogonadism in cancer patients with muscle wasting in a phase IIb enobosarm trial. in The Endocrine Society’s 94th Annual Meeting and Expo. 2012.
Crawford J, Dalton JT, Hancock ML, Johnston MA, Steiner M., Results from two phase 3 randomized trials of enobosarm, selective androgen receptor modulator (SARM), for the prevention and treatment of muscle wasting in NSCLC, in European Cancer Congress, E.S.f.M. Oncology, Editor 2013: Amsterdam, Netherlands.
Lundholm K, et al. Effects by daily long term provision of ghrelin to unselected weight-losing cancer patients: a randomized double-blind study. Cancer. 2010;116(8):2044–52.
Hiura Y, et al. Effects of ghrelin administration during chemotherapy with advanced esophageal cancer patients: a prospective, randomized, placebo-controlled phase 2 study. Cancer. 2012;118(19):4785–94.
Williams GR, Muss HB, Shachar SS. Cachexia in patients with cancer. Lancet Oncol. 2016;17(6):e220.
Greig CA, et al. Phase I/II trial of formoterol fumarate combined with megestrol acetate in cachectic patients with advanced malignancy. Support Care Cancer. 2014;22(5):1269–75.
Stewart Coats AJ, et al. Espindolol for the treatment and prevention of cachexia in patients with stage III/IV non-small cell lung cancer or colorectal cancer: a randomized, double-blind, placebo-controlled, international multicentre phase II study (the ACT-ONE trial). J Cachexia Sarcopenia Muscle. 2016;7(3):355–65.
Chasen M, Hirschman SZ, Bhargava R. Phase II study of the novel peptide-nucleic acid OHR118 in the management of cancer-related anorexia/cachexia. J Am Med Dir Assoc. 2011;12(1):62–7.
Yennurajalingam S, et al. The role of thalidomide and placebo for the treatment of cancer-related anorexia-cachexia symptoms: results of a double-blind placebo-controlled randomized study. J Palliat Med. 2012;15(10):1059–64.
Wilkes EA, et al. Poor tolerability of thalidomide in end-stage oesophageal cancer. Eur J Cancer Care (Engl). 2011;20(5):593–600.
Mantovani G, et al. Randomized phase III clinical trial of five different arms of treatment in 332 patients with cancer cachexia. Oncologist. 2010;15(2):200–11.
Maccio A, Madeddu C, Mantovani G. Current pharmacotherapy options for cancer anorexia and cachexia. Expert Opin Pharmacother. 2012;13(17):2453–72.
Madeddu C, et al. Randomized phase III clinical trial of a combined treatment with carnitine + celecoxib +/− megestrol acetate for patients with cancer-related anorexia/cachexia syndrome. Clin Nutr. 2012;31(2):176–82.
Sanchez-Lara K, et al. Effects of an oral nutritional supplement containing eicosapentaenoic acid on nutritional and clinical outcomes in patients with advanced non-small cell lung cancer: randomised trial. Clin Nutr. 2014;33(6):1017–23.
Rigas JR, et al. And the ALD518 study group. Efect of ALD518, a humanized anti-IL-6 antibody, on lean body mass loss and symptoms in patients with advanced non-small cell lung cancer (NSCLC): results of a phase II randomized, double-blind safety and efficacy trial. in American Society of Clinical Oncology. 2010. J Clin Oncol. (Meeting Abstracts).
Jatoi A, et al. A placebo-controlled, double-blind trial of infliximab for cancer-associated weight loss in elderly and/or poor performance non-small cell lung cancer patients (N01C9). Lung Cancer. 2010;68(2):234–9.
Hong DS, et al. MABp1, a first-in-class true human antibody targeting interleukin-1alpha in refractory cancers: an open-label, phase 1 dose-escalation and expansion study. Lancet Oncol. 2014;15(6):656–66.
Hong DS, et al. Xilonix, a novel true human antibody targeting the inflammatory cytokine interleukin-1 alpha, in non-small cell lung cancer. Investig New Drugs. 2015;33(3):621–31.
Balstad TR, Kaasa S, Solheim TS. Multimodal nutrition/anabolic therapy for wasting conditions. Curr Opin Clin Nutr Metab Care. 2014;17(3):226–35.
Argiles JM, et al. Are there any benefits of exercise training in cancer cachexia? J Cachexia Sarcopenia Muscle. 2012;3(2):73–6.
• Chevalier S, Winter A, et al. Curr Opin Clin Nutr Metab Care. 2014;17(3):213–8. This opinion article demomstrates the anabolic potential for patients with cancer even during conditions of weight loss, oncologic treatment , and inflammation. Specifically, high dose leucine administration with other essential amino acids provides the largest potential for increasing protein synthesis
Gagnon B, et al. A prospective evaluation of an interdisciplinary nutrition-rehabilitation program for patients with advanced cancer. Curr Oncol. 2013;20(6):310–8.
Fearon K, et al. Request for regulatory guidance for cancer cachexia intervention trials. J Cachexia Sarcopenia Muscle. 2015;6(4):272–4.
Muscaritoli M, et al. The “parallel pathway”: a novel nutritional and metabolic approach to cancer patients. Intern Emerg Med. 2011;6(2):105–12.
Gould DW, et al. Cancer cachexia prevention via physical exercise: molecular mechanisms. J Cachexia Sarcopenia Muscle. 2013;4(2):111–24.
Alves CR, et al. Aerobic exercise training as therapy for cardiac and cancer cachexia. Life Sci. 2015;125:9–14.
Sasso JP, et al. A framework for prescription in exercise-oncology research. J Cachexia Sarcopenia Muscle. 2015;6(2):115–24.
Fragala MS, Kenny AM, Kuchel GA. Muscle quality in aging: a multi-dimensional approach to muscle functioning with applications for treatment. Sports Med. 2015;45(5):641–58.
Leach HJ, et al. Exercise preferences, levels and quality of life in lung cancer survivors. Support Care Cancer. 2015;23(11):3239–47.
Yang F, et al. Dual targeting of MEK and PI3K pathways can act via tumor-intrinsic mechanisms to overcome resistance and tumor-extrinsic mechanisms to modulate immunity and limit cancer cachexia. in AACR 106th Annual Meeting. 2015. Philadelphia,PA: American Association for Cancer Research.
Tseng YC, et al. Preclinical investigation of the novel histone deacetylase inhibitor AR-42 in the treatment of cancer-induced cachexia. J Natl Cancer Inst. 2015;107(12):274.
Horie, K., Kanou, M., Takagi, K., Fujita, E., Hosoda, S., Sasaki, K., Watanabe, H., Hamada, M., Sugiyama, H., Hase, N., Azuma, Y., Kimura, T., and Yamana, K. Tei-SARM2: once-weekly oral SARM with robust muscle anabolism and minimal androgenic effects in reproductive tissues. in Endocrine Society’s 97th Annual Meeting and Expo. 2015. San Diego, CA: Endocrine Society.
Chen JA, et al. Ghrelin prevents tumour- and cisplatin-induced muscle wasting: characterization of multiple mechanisms involved. J Cachexia Sarcopenia Muscle. 2015;6(2):132–43.
Whirledge SD, et al. Ghrelin partially protects against cisplatin-induced male murine gonadal toxicity in a GHSR-1a-dependent manner. Biol Reprod. 2015;92(3):76.
Acknowledgements
This work was funded by the U.S. Dept. of Veterans Affairs (MERIT grants I01BX002807 and I01 CX000174, VA seed fund) and NIH Grants AG040583, AR067319, and CA185349 to JMG. We thank the University of Washington DERC (P30 DK017047) and NORC (P30 DK035816) for their help.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Lindsey J. Anderson declares that she has no conflict of interest.
Eliette D. Albrecht declares that she has no conflict of interest.
Jose M. Garcia has received research support from and served as a consultant for both Aeterna Zentaris and Helsinn.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Disclaimer
This is a work prepared by an officer or employee of the federal government as part of that person’s official duties. Under section 105 of the Copyright Act, such works are not entitled to domestic copyright protection under US law and are therefore in the public domain.
Additional information
This article is part of the Topical Collection on Gastrointestinal Cancers
An erratum to this article is available at http://dx.doi.org/10.1007/s11912-017-0595-4.
Rights and permissions
About this article
Cite this article
Anderson, L.J., Albrecht, E.D. & Garcia, J.M. Update on Management of Cancer-Related Cachexia. Curr Oncol Rep 19, 3 (2017). https://doi.org/10.1007/s11912-017-0562-0
Published:
DOI: https://doi.org/10.1007/s11912-017-0562-0