Abstract
Community-acquired pneumonia (CAP) is the leading cause of infectious death in the world. Immune dysregulation during acute lung infection plays a role in lung injury and the systemic inflammatory response. Cytokines seem to be major players in severe lung infection cases. Here, we present a review of published papers in the last 3 years regarding this topic. The cytokine response during pneumonia is different in bacterial vs viral infections; some of these cytokines correlate with clinical severity scales such as CURB65 or SOFA. Treatment focused in the cytokine environment is an interesting area that could impact the prognosis of CAP. Some of the agents that have been studied as co-adjuvant therapy are corticosteroids, macrolides, and linezolid, but anyone of those have shown a clear or proven efficacy or have been recommended as a part of the standard of care for CAP. More studies designed to define the role of immunomodulatory agents, such as co-adjuvant therapy in pneumonia, are needed.
Similar content being viewed by others
Introduction
Pneumonia is an infection of the lung tissue, and it is considered the forgotten killer. The World Health Organization (WHO) places lower respiratory infections as the third most common cause of death on the planet. It is responsible for approximately 3.5 million deaths yearly. In the USA, pneumonia and influenza caused approximately 50,000 deaths in 2010 and were the ninth leading cause of death [1, 2••].
A total of 20 to 60 % of community-acquired pneumonia (CAP) patients require hospitalization, which is mainly due to severe disease. Of those, 10 to 22 % have severe CAP requiring critical care, 44 to 83 % may undergo mechanical ventilation, and up to 50 % suffer from concomitant septic shock. In spite of the availability of affective antibiotics and guidelines for its rationale use in CAP, the high mortality related to this common infection (11–56 %) has not changed over the last 50 years [3•].
Innate and acquired pulmonary defense mechanisms are sufficient to keep the lower respiratory tract sterile despite constant exposure to particulate material and microorganisms via micro aspiration. The development of CAP indicates exposure to a particularly virulent microorganism, an overwhelming inoculum of microorganisms, a defect in host defense, or a combination of those factors [3•].
Once the lung is infected, the initial immune response is led by neutrophils that release several granule subtypes with the ability to kill microbes but also with the potential to cause tissue damage. Failure to control excessive inflammation and/or neutrophil activation results in exaggerated lung and systemic inflammation that leads to severe disease [4••]. The immune response in CAP may vary as different microbes may trigger different inflammatory responses depending on their intrinsic properties [5••]. The clinical outcome in CAP is not only determined by the causal agent, interaction between the immune response and pathogen clearance is extremely important. The balance of this interaction is regulated through complex interactions between immune cells and pro- and anti-inflammatory cytokines [6••].
The role of cytokines in CAP is not fully understood, but it is currently being studied to discover how they affect CAP outcomes. It is well known that the cytokine storm is present in severe CAP. In addition, it is appealing that adjuvant therapy targeted to regulate the immune response could be useful in these cases. Glucocorticoids and other treatments have been used as immunomodulators, but to date, none of these treatments have been licensed for this purpose [7•]. Thus, the focus of this review is on recent findings (from 2012 to date) regarding the systemic and local cytokine environment and their possible implications during CAP in the severity of lung infection. In addition, co-adjuvant therapies will be analyzed in this review that may represent an area of opportunity for therapeutic re-direction of CAP.
CAP Overview
Etiology
There is a wide variation in the reported etiology of CAP. This may depend in part on the diagnostic techniques used and the population studied [8••]. In the pre-antibiotic era, Streptococcus pneumoniae caused 95 % of pneumonia cases for which an etiology was found. Because new diagnostic tools are now available, the identification of other microorganisms is currently easier and has changed previous thinking regarding the causes of CAP. Most of the information we have obtained is from moderate to severe CAP patients who needed hospital care. Numerous viruses and bacteria are responsible for CAP. Infection can be caused by a single microorganism, but co-infection is also possible. Recent studies showed that respiratory viruses are the main cause. Among bacteria, S. pneumoniae remained the most commonly identified cause of CAP, but it represented only a minority of the patients [9••]. A meta-analysis also found respiratory viruses, particularly influenza and respiratory syncytial virus (RSV), as a common cause of CAP in 15 to 54 % of hospitalized adults [10••]. Co-infection with viruses and bacteria was found in 4 to 30 % in that report (Table 1).
Diagnostics
Markers of infection (fever or chills and leukocytosis), respiratory signs and/or symptoms (cough, sputum production, shortness of breath, chest pain, and abnormal pulmonary examination), and a new or changed infiltrate on a chest X-ray are consistent with CAP. The diagnosis of CAP can be more challenging in some specific groups, such as those who are elderly or those with comorbidities or immunosuppression states, in which the characteristic presentation of CAP is not present. Because empirical therapy is mainly based on the severity of the disease, extensive diagnostic testing is not a standard of care and the responsible agent is not identified in most patients. Extensive diagnostic testing seems to be more useful in those patients who present with severe CAP, and in which the results of the tests may lead to individualized therapy [2••, 8••, 11].
During the diagnosis of CAP, the clinician must determine the severity of the infection to decide the adequate antibiotic to use and if the patient needs to be either in a general ward or the ICU in the hospital. Several clinical scales of severity are available: CURB 65, APACHE II, SOFA, PSI, PIRO, and SMART COP. CURB 65 is based on five simple parameters: confusion, urea, respiratory rate, blood pressure, and age. CURB 65 stratifies patients according to the risk of death, which leads to further management: higher risk requires more aggressive interventions. APACHE II and SOFA are mainly used in critically ill patients. The other scales mentioned are not particularly useful in CAP patient evaluation. None of them include cytokine levels or another immune biomarker as a component of the scale [12].
Treatment
Antibiotic(s) selection is based mainly on the epidemiologic setting of the patient and in the severity of CAP. It is well known that starting early therapy is related with a better prognosis. A delay of more than 4 h between the initial presentation and the first antibiotic dose is associated with increased in-hospital mortality [13••]. That window is even shorter (1 h) in patients with septic shock due to CAP [14]. The length of therapy varies from 5 to 7 days in most cases [2••, 8••, 15].
Outpatients with CAP without comorbidities can be treated with only a macrolide. In cases of patients with coexistent illness and in in-patients, a macrolide plus a beta-lactam must be used or monotherapy with a respiratory fluoroquinolone, such as levofloxacin or moxifloxacin. These regimens cure approximately 90 % of the patients with mild or moderate CAP [16, 17].
Severe CAP requiring ICU management can be due to more aggressive bacteria such as Pseudomonas aeruginosa or Staphylococcus aureus, and the above recommendations do not cover these bacteria so other specific antibiotics must be considered. Adjuvant therapy in severe CAP has included systemic steroids, statins, and immunomodulatory agents, and their use deserves further research before being recommended by the IDSA/ATS guidelines for CAP [13••]. Among the recommended antibiotics, macrolides may play a role as immune modulators.
Cytokines in CAP
Cytokines are important soluble mediators of the innate and acquired immune response. Cell differentiation, chemotaxis, activators, or regulators of inflammatory and anti-inflammatory processes are among some of the main functions of cytokines in response to a pathogen. CAP is characterized by a systemic and local inflammatory response, where the number of identified cytokines has increased considerably over the years. Studies remain focused on well-known cytokines of the innate immune response, including interleukin (IL)-4, IL-6, IL-10, IL-8, IL-1β, tumor necrosis factor (TNF)-α, and transforming growth factor (TGF)-β. IL-17A and IL-22, which belong to the T-helper (Th) 17 subset [4••, 18]. Here we will focus in cytokines pattern found in bacterial and viral CAP, as well as, their possible role as severity biomarkers.
Bacterial CAP and Cytokines
A prospective study of 658 patients admitted with CAP showed higher levels of C reactive protein (CRP), procalcitonin (PCT), TNF-α, and IL-6 in those with bacteremia compared to CAP of an unknown cause but was not considered secondary to bacteria. Different cytokine profiles and biomarkers were found depending on the cause: atypical bacteria (lower PCT and IL-6), Enterobacteriaceae (higher IL-8), S. pneumoniae (high PCT), and Legionella pneumophila (higher CRP and TNF-α) [6••]. A pediatric population study demonstrated that IL-8 was associated with the severity of lung necrosis in CAP due to S. pneumoniae [19••].
In a Brazilian population with CAP, the IL-6 levels showed a good association with severity scores, such as APACHE II and CURB-65; however, other studies do not support this finding. IL-6 and TNF-α levels were associated with early death [20].
In a case–control study with of 253 patients, 83 patients had treatment failure which correlated with IL-6 levels [21]. An IL-6-174G/C gene polymorphism was associated with severe sepsis, acute respiratory failure, and ICU and hospital mortality in Egyptian children with CAP; however, IL-6 was found in similar concentrations in the different IL-6-174G/C genotypes. These findings may indicate that CAP susceptibility is a multifactorial disease and other molecules or genes may be involved [22••].
Paats et al. (2013) reported that patients with CAP showed high levels of Th17 subset expressing the signature cytokines interleukin IL-17A and IL-22 cells in BAL and peripheral blood, which suggests a protective role [23•]. IL-17 and IL-22 have previously been associated with neutrophilic inflammation, production of defensins, and are important for clearance of fungi and extracellular bacteria [24, 25].
S. pneumoniae was the common causative microorganism in a study that included 46 CAP patients ≤50 years old (median 37 years) and 41 CAP patients ≥80 years old (median 84 years). The serum concentrations of pro-inflammatory (IL-6 and IL-8) and anti-inflammatory cytokines (IL-10 and IL-1 receptor antagonist) did not differ between the age groups, although IL-8 levels at admission tended to be higher in elderly patients. Cytokine levels were positively correlated with PSI in young but not in elderly patients. These results suggest that elderly patients show a reduction in their systemic inflammatory response on admission for CAP [26•, 27].
Stromal-Cell-Derived Factor-1 (SDF-1) is a chemotactic cytokine of the CXC chemokines family. SDF-1 regulates the movement of neutrophils, monocytes, T lymphocytes, and basophils, and SDF-1 also induces cell migration, cell adhesion, neutrophil activation, and inflammation [28, 29]. SDF-1 has a positive correlation with CURB-65, APACHE II, and especially the PSI score in patients with CAP. Therefore, its use as a biological marker for the early diagnosis of CAP should be explored in futures studies [30•].
Matrix metalloproteinases (MMPs) are a large family of zinc-dependent endopeptidases and are also another important biomarker. In addition, MMPs are known as the major enzymes responsible for the proteolytic degradation of proteinaceous components of the extracellular matrix (ECM). MMPs also interact with multiple cytokines, participate in the pathology of infection and inflammation, and MMPs are key factors in the pathogenesis of various types of pneumonia [31].
In summary, the cytokine response plays an important role in the clinical evolution and severity of bacterial CAP. The most important cytokines, IL-6 and TNF-α, have previously been correlated with worse outcomes. IL-6 levels have shown a good association with the prediction scores such as APACHE II and CURB-65. High levels of IL-6 and TNF-α were associated with the development of acute kidney injury, the necessity for invasive mechanical ventilation, early death, and mortality. Another biomarker, SDF-1, has a positive correlation with clinical scores and pathogenesis but is less important in clinical evolution. The inflammatory-aging phenomenon observed in older patients must be considered when analyzing the present data.
Viral CAP and Cytokines
The severity of viral CAP is different, not only because of the specific virus but also because of different age groups. Influenza A virus (IAV) infection is the most common cause of pneumonia-related death in the developed world and mortality attributable to IAV infection can be much higher during pandemics. Currently available agents to treat IAV infection, including oseltamivir and zanamivir, inhibit viral NA. Therefore, they are most effective at limiting viral replication in the early stages of infection and in immunocompromised patients and are less effective once an antiviral innate immune response is established. The presence of viral RNA in the cytosol activates three major intracellular immune pathways that initiate the innate immune response toward the virus: retinoic acid inducible gene-1 (RIG-1) proteins, Toll-like receptors (TLRs; primarily TLR3 and TLR7), and inflammasome. Binding of viral RNA to helicase domains on RIG-1 triggers its interaction with mitochondria-associated antiviral signaling protein (MAVS), which induces the generation of type I and III interferons (IFN-α/β and IFN-λ) and activates the pro-inflammatory transcription factor nuclear factor (NF)-κB. Replication of the influenza virus replication critically depends on cellular NF-κB activity and NF-κB is a crucial pro-inflammatory signaling module, suggesting that strategies to inhibit the NF-κB pathway may be suitable for intervention. Finally, GM-CSF is an important mediator of antiviral host defense and a factor promoting repair of the alveolar epithelium [32••].
IL-6 and IL-8 are important mediators in influenza pneumonia. A recent study demonstrated that IL-8 is associated with disease progression among hospitalized patients with influenza A (H1N1) pdm09. IL-6, which has been identified as one of the critical mediators of damage due to the influenza A (H1N1)pdm09 virus, was also significantly elevated in non-(H1N1)pdm09 pneumonia patients [33]. Patients infected with influenza A H1N1pdm09 showed a strong Th17 response (IL-6, IL-8, and IL-17). It has been demonstrated that animal models develop severe pneumonia with the experimental infection of influenza A H1N1pdm09; however, IL-17-deficient mice ameliorated acute lung injury, and those mice treated with monoclonal antibodies against IL-17 were protected against acute lung injury [34•].
Our research group reported that high levels of inflammatory cytokines (IL-17, TNF-α, IFN-γ, and IL-4) were found in human lung tissue from fatal cases of influenza A (H1N1pdm09 and seasonal) but not IL-10. These findings may indicate that an inflammatory cytokine storm is involved in the immunopathogenesis of influenza A pneumonia, and regulatory mechanisms were not enough to control the inflammatory response [35•].
Finally, TGF-β is an immunoregulatory cytokine that plays an important role during inflammatory processes [36]. Rendon-Ramirez et al. found a negative correlation between SOFA score and TGF-β levels in patients with influenza A (H1N1)pdm09 [37•].
RSV infection is most common in young children in whom an early immune response cytokine, such as IL-8 (an important chemokine involved in neutrophil recruitment), is produced. In adaptative immunity, Th1/Th2 and Th17 responses have been detected in patients infected with RSV. Th17 cytokines induce chemokine, which is implicated in neutrophil recruitment and other effects such as IL-13 induction, mucus over-production, and inhibition of cytotoxicity mediated by T CD8 lymphocytes [38, 39]. Children with RSV-associated pneumonia presented low levels of IL-2, which probably indicated poor T cell stimulation. In fact, high levels of IFN-γ, IL-4, and IL-10 were found in these patients. This cytokine pattern is characteristic of the Th1/Th2/Treg response coexisting to control infection and/or avoid tissue injury [40].
In summary, there is a preponderance of cytokines in influenza pneumonia, and the most relevant cytokines is TGF-β. Influenza virus replication critically depends on cellular NF-κB activity (GM-CSF), an important mediator of the antiviral host defense and a factor promoting repair of the alveolar epithelium. IL-6 and IL-8 are mediators in the disease progression in influenza A, but what levels are exclusively from influenza must be questioned. Individuals infected with RSV show high levels of cytokines related with Th1/Th2/Th17/Treg lymphocyte subpopulations. Little is known about other respiratory viruses, and this is an opportunity to explore specific cytokine patterns.
Non-cytokine Biomarkers in CAP
There are several nonspecific non-cytokines biomarkers that we can use together with the severity pneumoniae scores: CRP, B-type natriuretic and peptide (BNP), D-dimers and ACE activity, and Pro-calcitonin. CRP on day 3 of antibiotic therapy was a marker of good prognosis if it showed a decrease. CRP <100 mg/L was independently associated with a lower risk of mortality. A CRP level that fails to fall by 50 % was independently associated with mortality. The AUROC for CRP was smaller than that of PSI and CURB-65. A BNP increase was independently associated with mortality. The AUROC for BNP was comparable with the AUROC of PSI. Adding BNP to PSI significantly improved the prognostic accuracy of PSI alone. D-Dimers and ACE activity had controversial results with no mortality impact [41•].
Pro-calcitonin is another important biomarker. The semi-quantitative procalcitonin serum level at hospital admission was less predictive of mortality from CAP compared with the blood urea nitrogen/serum albumin B/A ratio. Patients with serum procalcitonin levels >10.0 ng/mL required intensive care more than those with lower levels. Meanwhile, the PCT kinetics over the first 72 h is a useful tool for predicting the 30-day mortality in patients with severe sepsis and septic shock admitted to an intermediate care unit [42, 43]. Causal microorganisms play a key role in the host response and may trigger different inflammatory responses depending on their intrinsic properties such as the presence of a capsule, lipopolysaccharides in the cell wall, virulence factors, and spread of infection [5••] .
Soluble receptor for advanced glycation end products (sRAGE) is the soluble form of RAGE and it is elevated in the inflammatory condition, acute lung injury, and acute respiratory distress syndrome (ARDS) [44]. An association between elevated sRAGE and fatal outcomes suggests that it may have an independent causal effect in CAP. SOFA scores were the only clinical factor with the ability to identify surviving and ARDS patients, and logistic regression modeling demonstrated that SOFA and sRAGE were the only variables that modified the probability of a fatal outcome [45•].
Little is known regarding the participation of immunoglobulin isotypes in the pathogenesis of the severe disease caused by this virus. A study evaluated the association between plasma levels of IgG1, IgG2, IgG3, IgG4, IgA, IgM, and IgE, and the outcome in patients with severe pandemic influenza. It found that a high IgM level at admission to the ICU was a good prognostic factor in patients with severe disease caused by influenza A(H1N1)pdm09. This finding indicated the central role of IgM in preventing uncontrolled inflammatory response and mortality. The early assessment of IgM could contribute to directing clinical decisions in these patients [46].
Adjuvant Anti-cytokine Therapy in CAP
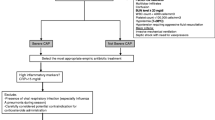
Research into adjuvant therapies for severe pneumonia have previously focused on immunosuppressive agents, such as glucocorticoids, and other treatments that include some antibiotics such as the macrolides [7•] (Fig. 1 and Table 2).
Corticosteroids
Systemic corticosteroids have anti-inflammatory effects that may attenuate the local and systemic inflammatory process observed in CAP. CAP treated with corticosteroids experienced a greater decline in levels of CRP and IL-6. The effects of steroids on the immune system are many and complex. Corticosteroids can switch off genes that encode pro-inflammatory cytokines (e.g., IL-6 and IL-8) and switch on genes that encode anti-inflammatory cytokines (e.g., IL-10). However, the use of steroids also exerts an influence on the immune function of different host defenses against bacteria when high dosages and prolonged treatment are used [47••]. Most of the corticosteroid studies in CAP have focused on clinical outcomes more than on the role of the cytokines. A meta-analysis did not find a benefit with the inclusion of corticosteroids in the treatment of CAP in adults. The studies were very heterogeneous and not restricted to severe CAP [48••].
A multicenter, randomized, double-blind, placebo-controlled trial compared methylprednisolone with a placebo in patients with both severe CAP and high inflammatory response (defined by a C-reactive protein level greater than 150 mg/L). Corticosteroids reduced the risk of treatment failure, but in-hospital mortality was not different [47••].
Another double-blind, randomized multicenter study compared 7 days of prednisone at 50 mg with a placebo in adult patients hospitalized with CAP. This study is the largest available and included 785 patients: the prednisone group included 392 individuals and the placebo group contained 393 individuals. The median time to clinical stability was shorter in the prednisone group than in the placebo group. Pneumonia-associated complications until day 30 did not differ between groups (11 (3 %) in the prednisone group and 22 (6 %) in the placebo group). This study concluded that prednisone, given for 7 days to patients hospitalized with CAP, shortens time to clinical stability without an increase in complications [49••]. Inhaled corticosteroids (ICS) treatment in patients with asthma, COPD, and CAP is associated with a reduced systemic inflammatory response. Patients who received ICS had reduced levels of TNF-α and IL-6, but there was no impact on long-term mortality [50].
Macrolides
Of the current antibiotics recommended for the treatment of CAP, macrolides such as clarithromycin and azithromycin seem to produce significant immunomodulatory effects. These effects include the suppression of specific transcription factors with a resultant decreased production of pro-inflammatory cytokines such as TNF-α, IL-1, IL-6, IL-8, and TGF-β; decreased neutrophil chemotaxis to the airways; and increased production of anti-inflammatory cytokines such as IL-10. In addition, macrolides have been shown to inhibit biofilm formation and decrease mucus hypersecretion, which leads to improved mucociliary clearance [51].
In spite of these effects, macrolide use did not show improved mortality when compared against fluoroquinolones in patients with CAP [17, 52]. A larger systematic review and meta-analysis that evaluated macrolide use and mortality due to CAP in 28 studies with almost 10,000 patients found a statistically significant lower risk of mortality compared with non-macrolide. The heterogeneity among the studies was substantial; however, the benefit may have been related to the non-antimicrobial immune modulatory properties of macrolides [53•].
Influenza pneumonia is associated with hypercytokinemia. A prospective, randomized, clinical trial with 107 enrolled patients using oseltamivir and azithromycin combination therapy for influenza was conducted. Although the difference in the inflammatory cytokine expression level was not statistically significant, combination therapy showed an early resolution of some symptoms [54].
Statins
Statins have pleiotropic effects that impact immunomodulatory, anti-inflammatory, anti-thrombotic, and direct microbicidal action. Statins are known to down-regulate cytokines and transcription factors that are abnormally increased during sepsis. Several in vitro and in vivo studies have demonstrated that statins reduce the levels of TNF-α, IL-1, IL-6, IL-8, NF-κB, and C-reactive protein. These effects inhibit the up-regulation of cytokines and potentially decrease inflammation and immune dysfunction. As a consequence, statins may decrease the severity of symptoms associated with sepsis [55]. A meta-analysis performed in 2013 included 28 studies and revealed an association between statins and the risk and mortality of CAP. However, the results constitute very low quality evidence, based on the GRADE framework, due to the observational study design, heterogeneity, and publication bias. Given the biological plausibility of these findings and the high burden and mortality of CAP, the meta-analysis results warranted additional and better-designed studies to define the utility of statins in CAP [56]. A recent multicenter, double blind, randomized clinical trial studied rosuvastatin versus a placebo in patients with sepsis-associated acute respiratory distress syndrome with 70 % of the cases due to pneumonia. Patients did not improve clinical outcomes with rosuvastatin but may have contributed to a higher degree of hepatic and renal dysfunction [57•].
Another randomized, double-blind, placebo-controlled trial was designed to determine if simvastatin, at 20 mg once daily for 4 days in hospitalized patients with CAP, improved clinical outcomes and reduced the concentration of inflammatory cytokines. The results suggest that the statin did not reduce either the time to clinical stability or the levels of inflammatory cytokines [41•].
Linezolid
Linezolid is an antibiotic of the oxazolidinone class. It has unique immunomodulatory effects in murine model of the post-influenza community that acquired MRSA pneumonia. Its use was associated with reduced levels of chemotactic chemokines and inflammatory cytokines KC, MIP-2, IFN-γ, TNF-α, and IL-1β in bronchoalveolar lavage and significantly lower levels of Panton-Valentine leukocidin toxin in the lungs. Linezolid also showed reduced lung injury when compared to vancomycin [58].
Conclusions
Hypercytokinemia and immune dysregulation have been described in patients with severe CAP. Multiple adjuvant therapies for pneumonia have been investigated, but none are currently licensed. Successful treatments must be targeted toward the immune response; promising biomarkers exist but have not yet reached common bedside practice. Corticosteroids and other agents that can regulate cytokine dysregulation may prevent treatment failure and improve the outcome of CAP. The use of steroids in severe CAP has promising findings; however, an integrative evaluation is necessary, especially in those cases with comorbidities. More studies designed to define the role of immunomodulatory agents as co-adjuvant therapies in CAP are needed.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Johnstone J, Mandell L. Guidelines and quality measures: do they improve outcomes of patients with community-acquired pneumonia? Infect Dis Clin North Am. 2013;27(1):71–86.
Wunderink RG, Waterer GW. Clinical practice. Community-acquired pneumonia. N Engl J Med. 2014;370(6):543–51. Major importance: It is an overview of CAP that help us to decide the antibiotic therapy, the extent of testing to determine the cause of pneumonia, and the appropriate location of treatment (home, inpatient floor, or ICU).
Sligl WI, Marrie TJ. Severe community-acquired pneumonia. Crit Care Clin. 2013;29(3):563–601. This article show us the epidemiology of severe CAP. Up to 22 % of patients with CAP require ICU admission, with 44 to 83 % requiring mechanical ventilation and up to 50 % presenting with concomitant septic shock.
Fernandez-Botran R, Uriarte SM, Arnold FW, et al. Contrasting inflammatory responses in severe and non-severe community-acquired pneumonia. Inflammation. 2014;37(4):1158–66. Major importance: this study compared systemic and local cytokine profiles in patients with severe versus non-severe CAP. Results indicate that patients with severe CAP fail to mount a local pro-inflammatory response but exhibit instead a more substantial systemic inflammatory response.
Menendez R, Sahuquillo-Arce JM, Reyes S, et al. Cytokine activation patterns and biomarkers are influenced by microorganisms in community-acquired pneumonia. Chest. 2012;141(6):1537–45. Major importance: There is a different inflammatory patterns elicited by different microorganisms. Recognizing these patterns may facilitate a broader understanding of host inflammatory response to microorganisms.
Paats MS, Bergen IM, Hanselaar WE, et al. Local and systemic cytokine profiles in nonsevere and severe community-acquired pneumonia. Eur Respir J. 2013;41(6):1378–85. Local and systemic cytokine profiles in nonsevere and severe community-acquired pneumonia.
Morton B, Pennington SH, Gordon SB. Immunomodulatory adjuvant therapy in severe community-acquired pneumonia. Expert Rev Respir Med. 2014;8(5):587–96. Immunomodulatory adjuvant therapy in severe community-acquired pneumonia.
Musher DM, Thorner AR. Community-acquired pneumonia. N Engl J Med. 2014;371(17):1619–28. This is a Major importance article; it is a review of CAP, since the epidemiology data to diagnostic methods and treatment.
Jain S, Self WH, Wunderink RG, et al. Community-acquired pneumonia requiring hospitalization among U.S. adults. N Engl J Med. 2015;373(5):415–27. This is a very important article. It show that the incidence of community-acquired pneumonia requiring hospitalization was highest among the oldest adults. No pathogen was detected in the majority of patients and respiratory viruses were detected more frequently than bacteria.
Qu JX, Gu L, Pu ZH, et al. Viral etiology of community-acquired pneumonia among adolescents and adults with mild or moderate severity and its relation to age and severity. BMC Infect Dis. 2015;15:89. Major importance, it is about recent epidemiological data of patients from Beijing, China, from 2010 to 2012.
Mandell LA. Community-acquired pneumonia: an overview. Postgrad Med. 2015;127(6):607–15.
Wiemken T, Kelley R, Ramirez J. Clinical scoring tools: which is best to predict clinical response and long-term outcomes? Infect Dis Clin North Am. 2013;27(1):33–48.
Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44 Suppl 2:S27–72. It is a very important article, these are the last CAP guidelines from the IDSA/ATS.
Dellinger RP, Levy MM, Rhodes A, et al. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med. 2013;41(2):580–637.
Nussenblatt V, Avdic E, Cosgrove S. What is the role of antimicrobial stewardship in improving outcomes of patients with CAP? Infect Dis Clin North Am. 2013;27(1):211–28.
Pakhale S, Mulpuru S, Verheij TJ, Kochen MM, Rohde GG, Bjerre LM. Antibiotics for community-acquired pneumonia in adult outpatients. Cochrane Database Syst Rev. 2014;10:CD002109.
Ruhe J, Mildvan D. Does empirical therapy with a fluoroquinolone or the combination of a beta-lactam plus a macrolide result in better outcomes for patients admitted to the general ward? Infect Dis Clin North Am. 2013;27(1):115–32.
Holloway TL, Rani M, Cap AP, Stewart RM, Schwacha MG. The association between the Th-17 immune response and pulmonary complications in a trauma ICU population. Cytokine. 2015;76(2):328–33.
Hsieh YC, Chi H, Chang KY, et al. Increase in fitness of Streptococcus pneumoniae is associated with the severity of necrotizing pneumonia. Pediatr Infect Dis J. 2015;34(5):499–505. Major Importance: a pediatric population study demonstrated that IL-8 was associated with the severity of lung necrosis in CAP due to S. pneumoniae.
Bacci MR, Leme RC, Zing NP, et al. IL-6 and TNF-alpha serum levels are associated with early death in community-acquired pneumonia patients. Braz J Med Biol Res. 2015;48(5):427–32. It is very important article because in a Brazilian population with CAP, the IL-6 levels showed a good association with severity scores, such as APACHE II and CURB-65.
Martin-Loeches I, Valles X, Menendez R, et al. Predicting treatment failure in patients with community acquired pneumonia: a case–control study. Respir Res. 2014;15:75.
Zidan HE, Elbehedy RM, Azab SF. IL6-174 G/C gene polymorphism and its relation to serum IL6 in Egyptian children with community-acquired pneumonia. Cytokine. 2014;67(2):60–4. The findings of this article may indicate that CAP susceptibility is a multifactorial disease and other molecules or genes may be involved.
Paats MS, Bergen IM, Hanselaar WE, et al. T helper 17 cells are involved in the local and systemic inflammatory response in community-acquired pneumonia. Thorax. 2013;68(5):468–74. Important article. Reported that patients with CAP showed high levels of Th17 subset, IL-17A and IL-22 cells in BAL and peripheral blood, which suggests a protective role.
Curtis MM, Way SS. Interleukin-17 in host defence against bacterial, mycobacterial and fungal pathogens. Immunology. 2009;126(2):177–85.
Stacey MA, Marsden M, Pham NT, et al. Neutrophils recruited by IL-22 in peripheral tissues function as TRAIL-dependent antiviral effectors against MCMV. Cell Host Microbe. 2014;15(4):471–83.
van Vught LA, Endeman H, Meijvis SC, et al. The effect of age on the systemic inflammatory response in patients with community-acquired pneumonia. Clin Microbiol Infect. 2014;20(11):1183–8. Important article. The results of this study suggest that elderly patients show a reduction in their systemic inflammatory response on admission for CAP.
Cevenini E, Monti D, Franceschi C. Inflamm-ageing. Curr Opin Clin Nutr Metab Care. 2013;16(1):14–20.
Petty JM, Sueblinvong V, Lenox CC, et al. Pulmonary stromal-derived factor-1 expression and effect on neutrophil recruitment during acute lung injury. J Immunol. 2007;178(12):8148–57.
Aggarwal NR, King LS, D’Alessio FR. Diverse macrophage populations mediate acute lung inflammation and resolution. Am J Physiol Lung Cell Mol Physiol. 2014;306(8):L709–25.
Tsai PK, Hsieh MJ, Wang HL, Chou MC, Yang SF, Yeh CB. Elevated plasma stromal-cell-derived factor-1 protein levels correlate with severity in patients with community-acquired pneumonia. Dis Markers. 2014;2014:829706. Important reference. It showed that stromal-cell-derived factor-1 (SDF-1) has a positive correlation with CURB-65, APACHE II, and especially the PSI score in patients with CAP.
Chiang TY, Tsao SM, Yeh CB, Yang SF. Matrix metalloproteinases in pneumonia. Clin Chim Acta. 2014;433:272–7.
Herold S, Becker C, Ridge KM, Budinger GR. Influenza virus-induced lung injury: pathogenesis and implications for treatment. Eur Respir J. 2015;45(5):1463–78. Major importance: this article is an overview of influenza virus and potential molecular treatment.
Davey Jr RT, Lynfield R, Dwyer DE, et al. The association between serum biomarkers and disease outcome in influenza A(H1N1)pdm09 virus infection: results of two international observational cohort studies. PLoS One. 2013;8(2):e57121.
Li C, Yang P, Sun Y, et al. IL-17 response mediates acute lung injury induced by the 2009 pandemic influenza A (H1N1) virus. Cell Res. 2012;22(3):528–38. Important article: it showed the correlation between IL-17 and acute lung injury.
Rodriguez-Ramirez HG, Salinas-Carmona MC, Barboza-Quintana O, et al. CD206+ cell number differentiates influenza A (H1N1)pdm09 from seasonal influenza A virus in fatal cases. Mediators Inflamm. 2014;2014:921054. Important article: this study showed that the inflammatory cytokine storm is involved in the immunopathogenesis of influenza A pneumonia, and regulatory mechanisms were not enough to control the inflammatory response.
Travis MA, Sheppard D. TGF-beta activation and function in immunity. Annu Rev Immunol. 2014;32:51–82.
Rendon-Ramirez EJ, Ortiz-Stern A, Martinez-Mejia C, et al. TGF-beta blood levels distinguish between influenza A (H1N1)pdm09 virus sepsis and sepsis due to other forms of community-acquired pneumonia. Viral Immunol. 2015;28(5):248–54. Important article: this study found a negative correlation between the best score (SOFA) to evaluate influenza and TGF-beta.
Arruvito L, Raiden S, Geffner J. Host response to respiratory syncytial virus infection. Curr Opin Infect Dis. 2015;28(3):259–66.
Gonzalez PA, Bueno SM, Carreno LJ, Riedel CA, Kalergis AM. Respiratory syncytial virus infection and immunity. Rev Med Virol. 2012;22(4):230–44.
Ye Q, Shao WX, Shang SQ, Pan YX, Shen HQ, Chen XJ. Epidemiological characteristics and immune status of children with respiratory syncytial virus. J Med Virol. 2015;87(2):323–9.
Viasus D, Simonetti A, Garcia-Vidal C, Carratala J. Prediction of prognosis by markers in community-acquired pneumonia. Expert Rev Anti Infect Ther. 2013;11(9):917–29. Important article. It talks about several nonspecific non-cytokines biomarkers that we can use together with the severity pneumoniae scores.
Pieralli F, Vannucchi V, Mancini A, et al. Procalcitonin kinetics in the first 72 hours predicts 30-day mortality in severely ill septic patients admitted to an intermediate care unit. J Clin Med Res. 2015;7(9):706–13.
Ugajin M, Yamaki K, Hirasawa N, Yagi T. Predictive values of semi-quantitative procalcitonin test and common biomarkers for the clinical outcomes of community-acquired pneumonia. Respir Care. 2014;59(4):564–73.
Jabaudon M, Blondonnet R, Roszyk L, et al. Soluble forms and ligands of the receptor for advanced glycation end-products in patients with acute respiratory distress syndrome: an observational prospective study. PLoS One. 2015;10(8):e0135857.
Narvaez-Rivera RM, Rendon A, Salinas-Carmona MC, Rosas-Taraco AG. Soluble RAGE as a severity marker in community acquired pneumonia associated sepsis. BMC Infect Dis. 2012;12:15. Important article: an association between elevated sRAGE and fatal outcomes suggests that it may have an independent causal effect in CAP.
Justel M, Socias L, Almansa R, et al. IgM levels in plasma predict outcome in severe pandemic influenza. J Clin Virol. 2013;58(3):564–7.
Torres A, Sibila O, Ferrer M, et al. Effect of corticosteroids on treatment failure among hospitalized patients with severe community-acquired pneumonia and high inflammatory response: a randomized clinical trial. JAMA. 2015;313(7):677–86. Major importance: systemic corticosteroids have anti-inflammatory effects that may attenuate the local and systemic inflammatory process observed in CAP. Corticosteroids reduced the risk of treatment failure, but in-hospital mortality was not different.
Nie W, Zhang Y, Cheng J, Xiu Q. Corticosteroids in the treatment of community-acquired pneumonia in adults: a meta-analysis. PLoS One. 2012;7(10):e47926. Major importance: this is a meta-analysis that did not find a benefit with the inclusion of corticosteroids in the treatment of CAP in adults, but it was very heterogeneous and not restricted to severe CAP.
Blum CA, Nigro N, Briel M, et al. Adjunct prednisone therapy for patients with community-acquired pneumonia: a multicentre, double-blind, randomised, placebo-controlled trial. Lancet. 2015;385(9977):1511–8. Major importance: this randomized multicenter study compared seven days of prednisone with a placebo in adult patients hospitalized with CAP and concluded that prednisone shortens time to clinical stability without an increase in complications.
Ferrer M, Torres A, Martinez R, et al. Inhaled corticosteroids and systemic inflammatory response in community-acquired pneumonia: a prospective clinical study. Respirology. 2014;19(6):929–35.
Kovaleva A, Remmelts HH, Rijkers GT, Hoepelman AI, Biesma DH, Oosterheert JJ. Immunomodulatory effects of macrolides during community-acquired pneumonia: a literature review. J Antimicrob Chemother. 2012;67(3):530–40.
Raz-Pasteur A, Shasha D, Paul M. Fluoroquinolones or macrolides alone versus combined with beta-lactams for adults with community-acquired pneumonia: systematic review and meta-analysis. Int J Antimicrob Agents. 2015;46(3):242–8.
Sligl WI, Asadi L, Eurich DT, Tjosvold L, Marrie TJ, Majumdar SR. Macrolides and mortality in critically ill patients with community-acquired pneumonia: a systematic review and meta-analysis. Crit Care Med. 2014;42(2):420–32. Important article: this is a systematic review and meta-analysis that evaluated macrolide use and mortality and found a statistically significant lower risk of mortality compared with non-macrolide but the heterogeneity among the studies was substantial.
Kakeya H, Seki M, Izumikawa K, et al. Efficacy of combination therapy with oseltamivir phosphate and azithromycin for influenza: a multicenter, open-label, randomized study. PLoS One. 2014;9(3):e91293.
Dobesh PP, Olsen KM. Statins role in the prevention and treatment of sepsis. Pharmacol Res. 2014;88:31–40.
Khan AR, Riaz M, Bin Abdulhak AA, et al. The role of statins in prevention and treatment of community acquired pneumonia: a systematic review and meta-analysis. PLoS One. 2013;8(1):e52929.
National Heart L, Blood Institute ACTN, Truwit JD, et al. Rosuvastatin for sepsis-associated acute respiratory distress syndrome. N Engl J Med. 2014;370(23):2191–200. Important article: this randomized study showed no mortality benefit and higher degree of hepatic and renal dysfunction.
Bhan U, Podsiad AB, Kovach MA, Ballinger MN, Keshamouni V, Standiford TJ. Linezolid has unique immunomodulatory effects in post-influenza community acquired MRSA pneumonia. PLoS One. 2015;10(1):e0114574.
Acknowledgments
This work was supported by CONACYT, PAICYT-UANL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Drs Rendon, Rendon-Ramirez, and Rosas-Taraco declare no conflicts of interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by the author.
Additional information
This article is part of the Topical Collection on Respiratory Infections
Rights and permissions
About this article
Cite this article
Rendon, A., Rendon-Ramirez, E.J. & Rosas-Taraco, A.G. Relevant Cytokines in the Management of Community-Acquired Pneumonia. Curr Infect Dis Rep 18, 10 (2016). https://doi.org/10.1007/s11908-016-0516-y
Published:
DOI: https://doi.org/10.1007/s11908-016-0516-y