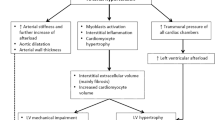
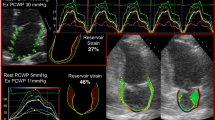
Abstract
Purpose of Review
This review aims to summarize recent developments in identifying and quantifying both the presence and amount of myocardial fibrosis by imaging and biomarkers. Further, this review seeks to describe in general ways how this information may be used to identify hypertension and the transition to heart failure with preserved ejection fraction.
Recent Findings
Recent studies using cardiac magnetic resonance imaging highlight the progressive nature of fibrosis from normal individuals to those with hypertension to those with clinical heart failure. However, separating hypertensive patients from those with heart failure remains challenging. Recent studies involving echocardiography show the subclinical myocardial strain changes between hypertensive heart disease and heart failure. Lastly, recent studies highlight the potential use of biomarkers to identify those with hypertension at the greatest risk of developing heart failure.
Summary
In light of the heterogeneous nature between hypertension and heart failure with preserved ejection fraction, an integrated approach with cardiac imaging and biomarker analysis may enable clinicians and investigators to more accurately characterize, prevent, and treat heart failure in those with hypertension.



Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Benjamin EJ, Muntner P, Alonso A, Bittencourt MS, Callaway CW, Carson AP, et al. Heart disease and stroke statistics-2019 update: a report from the American Heart Association. Circulation. 2019;139(10):e56–e528. https://doi.org/10.1161/CIR.0000000000000659.
Muntner P, Hardy ST, Fine LJ, Jaeger BC, Wozniak G, Levitan EB, et al. Trends in blood pressure control among US adults with hypertension, 1999-2000 to 2017-2018. Jama. 2020;324(12):1190–200. https://doi.org/10.1001/jama.2020.14545.
Levy D, Larson MG, Vasan RS, Kannel WB, Ho KK. The progression from hypertension to congestive heart failure. Jama. 1996;275(20):1557–62.
Drazner MH. The progression of hypertensive heart disease. Circulation. 2011;123(3):327–34. https://doi.org/10.1161/circulationaha.108.845792.
Messerli FH, Rimoldi SF, Bangalore S. The transition from hypertension to heart failure: contemporary update. JACC Heart Fail. 2017;5(8):543–51. https://doi.org/10.1016/j.jchf.2017.04.012.
Díez J, Frohlich ED. A translational approach to hypertensive heart disease. Hypertension (Dallas, Tex : 1979). 2010;55(1):1–8. https://doi.org/10.1161/hypertensionaha.109.141887.
Yang EY, Khan MA, Graviss EA, Nguyen DT, Bhimaraj A, Nambi V, et al. Relationship of extracellular volume assessed on cardiac magnetic resonance and serum cardiac troponins and natriuretic peptides with heart failure outcomes. Sci Rep. 2019;9(1):20168. https://doi.org/10.1038/s41598-019-56213-4.
Tadic M, Cuspidi C, Plein S, Milivojevic IG, Wang DW, Grassi G, et al. Comprehensive assessment of hypertensive heart disease: cardiac magnetic resonance in focus. Heart Fail Rev. 2020. https://doi.org/10.1007/s10741-020-09943-x.
Mewton N, Liu CY, Croisille P, Bluemke D, Lima JA. Assessment of myocardial fibrosis with cardiovascular magnetic resonance. J Am Coll Cardiol. 2011;57(8):891–903. https://doi.org/10.1016/j.jacc.2010.11.013.
Krittayaphong R, Boonyasirinant T, Chaithiraphan V, Maneesai A, Saiviroonporn P, Nakyen S, et al. Prognostic value of late gadolinium enhancement in hypertensive patients with known or suspected coronary artery disease. Iint J Cardiovasc Imaging. 2010;26(Suppl 1):123–31. https://doi.org/10.1007/s10554-009-9574-7.
Hinojar R, Varma N, Child N, Goodman B, Jabbour A, Yu CY, et al. T1 mapping in discrimination of hypertrophic phenotypes: hypertensive heart disease and hypertrophic cardiomyopathy: findings from the International T1 Multicenter Cardiovascular Magnetic Resonance Study. Circ Cardiovasc Imaging. 2015;8(12):e003285. https://doi.org/10.1161/circimaging.115.003285.
Duca F, Kammerlander AA, Zotter-Tufaro C, Aschauer S, Schwaiger ML, Marzluf BA, et al. Interstitial fibrosis, functional status, and outcomes in heart failure with preserved ejection fraction: insights from a prospective cardiac magnetic resonance imaging study. Circ Cardiovasc Imaging. 2016;9(12):e005277. https://doi.org/10.1161/circimaging.116.005277.
Sibley CT, Noureldin RA, Gai N, Nacif MS, Liu S, Turkbey EB, et al. T1 mapping in cardiomyopathy at cardiac MR: comparison with endomyocardial biopsy. Radiology. 2012;265(3):724–32. https://doi.org/10.1148/radiol.12112721.
Flett AS, Hayward MP, Ashworth MT, Hansen MS, Taylor AM, Elliott PM, et al. Equilibrium contrast cardiovascular magnetic resonance for the measurement of diffuse myocardial fibrosis: preliminary validation in humans. Circulation. 2010;122(2):138–44. https://doi.org/10.1161/circulationaha.109.930636.
Müller-Brunotte R, Kahan T, López B, Edner M, González A, Díez J, et al. Myocardial fibrosis and diastolic dysfunction in patients with hypertension: results from the Swedish Irbesartan Left Ventricular Hypertrophy Investigation versus Atenolol (SILVHIA). J Hypertens. 2007;25(9):1958–66. https://doi.org/10.1097/HJH.0b013e3282170ada.
Díez J. Mechanisms of cardiac fibrosis in hypertension. J Clin Hypertens (Greenwich, Conn). 2007;9(7):546–50. https://doi.org/10.1111/j.1524-6175.2007.06626.x.
Kockova R, Kacer P, Pirk J, Maly J, Sukupova L, Sikula V, et al. Native T1 relaxation time and extracellular volume fraction as accurate markers of diffuse myocardial fibrosis in heart valve disease - comparison with targeted left ventricular myocardial biopsy. Circ J : official journal of the Japanese Circulation Society. 2016;80(5):1202–9. https://doi.org/10.1253/circj.CJ-15-1309.
•• Kuruvilla S, Janardhanan R, Antkowiak P, Keeley EC, Adenaw N, Brooks J, et al. Increased extracellular volume and altered mechanics are associated with LVH in hypertensive heart disease, not hypertension alone. J Am Coll Cardiol Img. 2015;8(2):172–80. https://doi.org/10.1016/j.jcmg.2014.09.020This study highlights the ability of T1 mapping and calculation of extracellular volume to identify cardiac remodeling in the setting of hypertension.
• Wu LM, Wu R, Ou YR, Chen BH, Yao QY, Lu Q, et al. Fibrosis quantification in hypertensive heart disease with LVH and non-LVH: findings from T1 mapping and contrast-free cardiac diffusion-weighted imaging. Sci Rep. 2017;7(1):559. https://doi.org/10.1038/s41598-017-00627-5This study highlights the ability of T1 mapping, calculation of extracellular volume, and diffusion-weighted imaging to identify cardiac remodeling in the setting of hypertension.
Mordi IR, Singh S, Rudd A, Srinivasan J, Frenneaux M, Tzemos N, et al. Comprehensive echocardiographic and cardiac magnetic resonance evaluation differentiates among heart failure with preserved ejection fraction patients, hypertensive patients, and healthy control subjects. J Am Coll Cardiol Img. 2018;11(4):577–85. https://doi.org/10.1016/j.jcmg.2017.05.022.
Rommel KP, von Roeder M, Latuscynski K, Oberueck C, Blazek S, Fengler K, et al. Extracellular volume fraction for characterization of patients with heart failure and preserved ejection fraction. J Am Coll Cardiol. 2016;67(15):1815–25. https://doi.org/10.1016/j.jacc.2016.02.018.
Kanagala P, Cheng ASH, Singh A, Khan JN, Gulsin GS, Patel P, et al. Relationship between focal and diffuse fibrosis assessed by cmr and clinical outcomes in heart failure with preserved ejection fraction. J Am Coll Cardiol Img. 2019;12(11 Pt 2):2291–301. https://doi.org/10.1016/j.jcmg.2018.11.031.
Schelbert EB, Fridman Y, Wong TC, Abu Daya H, Piehler KM, Kadakkal A, et al. Temporal relation between myocardial fibrosis and heart failure with preserved ejection fraction: association with baseline disease severity and subsequent outcome. JAMA Cardiol. 2017;2(9):995–1006. https://doi.org/10.1001/jamacardio.2017.2511.
Tastet L, Kwiecinski J, Pibarot P, Capoulade R, Everett RJ, Newby DE, et al. Sex-related differences in the extent of myocardial fibrosis in patients with aortic valve stenosis. J Am Coll Cardiol Img. 2020;13(3):699–711. https://doi.org/10.1016/j.jcmg.2019.06.014.
Everett RJ, Tastet L, Clavel MA, Chin CWL, Capoulade R, Vassiliou VS, et al. Progression of hypertrophy and myocardial fibrosis in aortic stenosis: a multicenter cardiac magnetic resonance study. Circ Cardiovasc Imaging. 2018;11(6):e007451. https://doi.org/10.1161/circimaging.117.007451.
Braga JR, Leong-Poi H, Rac VE, Austin PC, Ross HJ, Lee DS. Trends in the use of cardiac imaging for patients with heart failure in Canada. JAMA Netw Open. 2019;2(8):e198766. https://doi.org/10.1001/jamanetworkopen.2019.8766.
Collier P, Phelan D, Klein A. A test in context: myocardial strain measured by speckle-tracking echocardiography. J Am Coll Cardiol. 2017;69(8):1043–56. https://doi.org/10.1016/j.jacc.2016.12.012.
Levy PT, Machefsky A, Sanchez AA, Patel MD, Rogal S, Fowler S, et al. Reference ranges of left ventricular strain measures by two-dimensional speckle-tracking echocardiography in children: a systematic review and meta-analysis. J Am Soc Echocardiogr : official publication of the American Society of Echocardiography. 2016;29(3):209–25.e6. https://doi.org/10.1016/j.echo.2015.11.016.
Nagata Y, Wu VC, Otsuji Y, Takeuchi M. Normal range of myocardial layer-specific strain using two-dimensional speckle tracking echocardiography. PLoS One. 2017;12(6):e0180584. https://doi.org/10.1371/journal.pone.0180584.
Chan J, Shiino K, Obonyo NG, Hanna J, Chamberlain R, Small A, et al. Left ventricular global strain analysis by two-dimensional speckle-tracking echocardiography: the learning curve. J Am Soc Echocardiogr : official publication of the American Society of Echocardiography. 2017;30(11):1081–90. https://doi.org/10.1016/j.echo.2017.06.010.
Quintana RA, Bui LP, Moudgil R, Palaskas N, Hassan S, Abe JI, et al. Speckle-tracking echocardiography in cardio-oncology and beyond. Tex Heart Inst J. 2020;47(2):96–107. https://doi.org/10.14503/thij-18-6736.
Saito M, Khan F, Stoklosa T, Iannaccone A, Negishi K, Marwick TH. Prognostic implications of LV strain risk score in asymptomatic patients with hypertensive heart disease. JACC Cardiovasc Imaging. 2016;9(8):911–21. https://doi.org/10.1016/j.jcmg.2015.09.027.
Przewlocka-Kosmala M, Marwick TH, Dabrowski A, Kosmala W. Contribution of cardiovascular reserve to prognostic categories of heart failure with preserved ejection fraction: a classification based on machine learning. J Am Soc Echocardiogr : official publication of the American Society of Echocardiography. 2019;32(5):604–15.e6. https://doi.org/10.1016/j.echo.2018.12.002.
Okamoto R, Ali Y, Hashizume R, Suzuki N, Ito M. BNP as a major player in the heart-kidney connection. Int J Mol Sci. 2019;20(14):3581. https://doi.org/10.3390/ijms20143581.
Vinnakota S, Chen HH. The importance of natriuretic peptides in cardiometabolic diseases. J Endocrine Soc. 2020;4(6):bvaa052. https://doi.org/10.1210/jendso/bvaa052.
König P, Mayer O, Bruthans J, Seidlerová J, Mateřánková M, Gelžinský J, et al. The prognostic importance of subclinical heart failure in stable coronary heart disease patients. Acta Cardiol. 2020;75(4):329–36. https://doi.org/10.1080/00015385.2019.1590958.
Kubo T, Ochi Y, Baba Y, Sugiura K, Takahashi A, Hirota T, et al. Elevation of high-sensitivity cardiac troponin T and left ventricular remodelling in hypertrophic cardiomyopathy. ESC Heart Fail. 2020;7(6):3593–600. https://doi.org/10.1002/ehf2.12852.
Aimo A, Gaggin HK, Barison A, Emdin M, Januzzi JL Jr. Imaging, biomarker, and clinical predictors of cardiac remodeling in heart failure with reduced ejection fraction. JACC Heart Fail. 2019;7(9):782–94. https://doi.org/10.1016/j.jchf.2019.06.004.
Nordestgaard BG, Zacho J. Lipids, atherosclerosis and CVD risk: is CRP an innocent bystander? Nutr, Metab, Cardiovasc Diseases : NMCD. 2009;19(8):521–4. https://doi.org/10.1016/j.numecd.2009.07.005.
• Chirinos JA, Orlenko A, Zhao L, Basso MD, Cvijic ME, Li Z, et al. Multiple plasma biomarkers for risk stratification in patients with heart failure and preserved ejection fraction. J Am Coll Cardiol. 2020;75(11):1281–95. https://doi.org/10.1016/j.jacc.2019.12.069This study highlights the emerging significance of using biomarkers in aggregate to identify potential heart failure phenotypes.
Barman SA, Li X, Haigh S, Kondrikov D, Mahboubi K, Bordan Z, et al. Galectin-3 is expressed in vascular smooth muscle cells and promotes pulmonary hypertension through changes in proliferation, apoptosis, and fibrosis. Am J Physiol Lung Cell Mol Physiol. 2019;316(5):L784–l97. https://doi.org/10.1152/ajplung.00186.2018.
Yao Y, Shen D, Chen R, Ying C, Wang C, Guo J, et al. Galectin-3 predicts left ventricular remodeling of hypertension. Jf ClinHypertens (Greenwich, Conn). 2016;18(6):506–11. https://doi.org/10.1111/jch.12757.
Ghorbani A, Bhambhani V, Christenson RH, Meijers WC, de Boer RA, Levy D, et al. Longitudinal change in Galectin-3 and incident cardiovascular outcomes. J Am Coll Cardiol. 2018;72(25):3246–54. https://doi.org/10.1016/j.jacc.2018.09.076.
López B, González A, Ravassa S, Beaumont J, Moreno MU, San José G, et al. Circulating biomarkers of myocardial fibrosis: the need for a reappraisal. J Am Coll Cardiol. 2015;65(22):2449–56. https://doi.org/10.1016/j.jacc.2015.04.026.
Zile MR, Baicu CF, Ikonomidis JS, Stroud RE, Nietert PJ, Bradshaw AD, et al. Myocardial stiffness in patients with heart failure and a preserved ejection fraction: contributions of collagen and titin. Circulation. 2015;131(14):1247–59. https://doi.org/10.1161/circulationaha.114.013215.
Duprez DA, Gross MD, Ix JH, Kizer JR, Tracy RP, Shea S, et al. Collagen biomarkers predict new onset of hypertension in normotensive participants: the Multi-Ethnic Study of Atherosclerosis. J Hypertens. 2018;36(11):2245–50. https://doi.org/10.1097/hjh.0000000000001793.
López B, Ravassa S, González A, Zubillaga E, Bonavila C, Bergés M, et al. Myocardial collagen cross-linking is associated with heart failure hospitalization in patients with hypertensive heart failure. J Am Coll Cardiol. 2016;67(3):251–60. https://doi.org/10.1016/j.jacc.2015.10.063.
Ravassa S, Trippel T, Bach D, Bachran D, González A, López B, et al. Biomarker-based phenotyping of myocardial fibrosis identifies patients with heart failure with preserved ejection fraction resistant to the beneficial effects of spironolactone: results from the Aldo-DHF trial. Eur J Heart Fail. 2018;20(9):1290–9. https://doi.org/10.1002/ejhf.1194.
Wu CK, Su MM, Wu YF, Hwang JJ, Lin LY. Combination of plasma biomarkers and clinical data for the detection of myocardial fibrosis or aggravation of heart failure symptoms in heart failure with preserved ejection fraction patients. J Clin Med. 2018;7(11):427. https://doi.org/10.3390/jcm7110427.
Duprez DA, Gross MD, Kizer JR, Ix JH, Hundley WG, Jacobs DR Jr. Predictive value of collagen biomarkers for heart failure with and without preserved ejection fraction: MESA (Multi-Ethnic Study of Atherosclerosis). J Am Heart Assoc. 2018;7(5):e007885. https://doi.org/10.1161/jaha.117.007885.
Pichler G, Redon J, Martínez F, Solaz E, Calaforra O, Andrés MS, et al. Cardiac magnetic resonance-derived fibrosis, strain and molecular biomarkers of fibrosis in hypertensive heart disease. J Hypertens. 2020;38:2036–42. https://doi.org/10.1097/hjh.0000000000002504.
Moore-Morris T, Guimarães-Camboa N, Yutzey KE, Pucéat M, Evans SM. Cardiac fibroblasts: from development to heart failure. J Mol Med (Berlin, Germany). 2015;93(8):823–30. https://doi.org/10.1007/s00109-015-1314-y.
Frangogiannis NG. The extracellular matrix in ischemic and nonischemic heart failure. Circ Res. 2019;125(1):117–46. https://doi.org/10.1161/circresaha.119.311148.
Travers JG, Kamal FA, Robbins J, Yutzey KE, Blaxall BC. Cardiac fibrosis: the fibroblast awakens. Circ Res. 2016;118(6):1021–40. https://doi.org/10.1161/circresaha.115.306565.
Drew DA, Katz R, Kritchevsky S, Ix JH, Shlipak MG, Newman AB, et al. Fibroblast growth factor 23 and blood pressure in older adults: the health, aging, and body composition study. Hypertension (Dallas, Tex : 1979). 2020;76(1):236–43. https://doi.org/10.1161/hypertensionaha.120.14703.
Akhabue E, Montag S, Reis JP, Pool LR, Mehta R, Yancy CW, et al. FGF23 (Fibroblast Growth Factor-23) and incident hypertension in young and middle-aged adults: the CARDIA Study. Hypertension (Dallas, Tex : 1979). 2018;72(1):70–6. https://doi.org/10.1161/hypertensionaha.118.11060.
Mirza MA, Larsson A, Melhus H, Lind L, Larsson TE. Serum intact FGF23 associate with left ventricular mass, hypertrophy and geometry in an elderly population. Atherosclerosis. 2009;207(2):546–51. https://doi.org/10.1016/j.atherosclerosis.2009.05.013.
• Roy C, Slimani A, de Meester C, Amzulescu M, Pasquet A, Vancraeynest D, et al. Associations and prognostic significance of diffuse myocardial fibrosis by cardiovascular magnetic resonance in heart failure with preserved ejection fraction. J Cardiovasc Magnetic Resonance : official journal of the Society for Cardiovascular Magnetic Resonance. 2018;20(1):55. https://doi.org/10.1186/s12968-018-0477-4This study highlights the potential of the relationship between biomarkers and cardiac imaging that may lead to better ways to identify and treat different heart failure patients.
Almahmoud MF, Soliman EZ, Bertoni AG, Kestenbaum B, Katz R, Lima JAC, et al. Fibroblast Growth Factor-23 and heart failure with reduced versus preserved ejection fraction: MESA. J Am Heart Assoc. 2018;7(18):e008334. https://doi.org/10.1161/jaha.117.008334.
Akhabue E, Vu TT, Vaidya A, Michos ED, de Boer IH, Kestenbaum B, et al. Fibroblast Growth Factor-23, heart failure risk, and renin-angiotensin-aldosterone-system blockade in hypertension: the MESA Study. Am J Hypertens. 2019;32(1):18–25. https://doi.org/10.1093/ajh/hpy142.
Roselló-Lletí E, Rivera M, Martínez-Dolz L, González Juanatey JR, Cortés R, Jordán A, et al. Inflammatory activation and left ventricular mass in essential hypertension. Am J Hypertens. 2009;22(4):444–50. https://doi.org/10.1038/ajh.2008.369.
Masiha S, Sundström J, Lind L. Inflammatory markers are associated with left ventricular hypertrophy and diastolic dysfunction in a population-based sample of elderly men and women. J Hum Hypertens. 2013;27(1):13–7. https://doi.org/10.1038/jhh.2011.113.
Cainzos-Achirica M, Enjuanes C, Greenland P, McEvoy JW, Cushman M, Dardari Z, et al. The prognostic value of interleukin 6 in multiple chronic diseases and all-cause death: The Multi-Ethnic Study of Atherosclerosis (MESA). Atherosclerosis. 2018;278:217–25. https://doi.org/10.1016/j.atherosclerosis.2018.09.034.
Marques MD, Nauffal V, Ambale-Venkatesh B, Vasconcellos HD, Wu C, Bahrami H, et al. Association between inflammatory markers and myocardial fibrosis. Hypertension (Dallas, Tex : 1979). 2018;72(4):902–8. https://doi.org/10.1161/hypertensionaha.118.11463.
Markousis-Mavrogenis G, Tromp J, Ouwerkerk W, Devalaraja M, Anker SD, Cleland JG, et al. The clinical significance of interleukin-6 in heart failure: results from the BIOSTAT-CHF study. Eur J Heart Fail. 2019;21(8):965–73. https://doi.org/10.1002/ejhf.1482.
Nguyen K, Fan W, Bertoni A, Budoff MJ, Defilippi C, Lombardo D, et al. N-terminal Pro B-type natriuretic peptide and high-sensitivity cardiac troponin as markers for heart failure and cardiovascular disease risks according to glucose status (from the Multi-Ethnic Study of Atherosclerosis [MESA]). Am J Cardiol. 2020;125(8):1194–201. https://doi.org/10.1016/j.amjcard.2020.01.025.
Devereux RB, Palmieri V, Sharpe N, De Quattro V, Bella JN, de Simone G, et al. Effects of once-daily angiotensin-converting enzyme inhibition and calcium channel blockade-based antihypertensive treatment regimens on left ventricular hypertrophy and diastolic filling in hypertension: the prospective randomized enalapril study evaluating regression of ventricular enlargement (preserve) trial. Circulation. 2001;104(11):1248–54. https://doi.org/10.1161/hc3601.095927.
Schneider MP, Klingbeil AU, Delles C, Ludwig M, Kolloch RE, Krekler M, et al. Effect of irbesartan versus atenolol on left ventricular mass and voltage: results of the CardioVascular Irbesartan Project. Hypertension (Dallas, Tex : 1979). 2004;44(1):61–6. https://doi.org/10.1161/01.HYP.0000133249.28773.d7.
Klingbeil AU, Schneider M, Martus P, Messerli FH, Schmieder RE. A meta-analysis of the effects of treatment on left ventricular mass in essential hypertension. Am J Med. 2003;115(1):41–6. https://doi.org/10.1016/s0002-9343(03)00158-x.
Upadhya B, Rocco MV, Pajewski NM, Morgan T, Blackshear J, Hundley WG, et al. Effect of intensive blood pressure reduction on left ventricular mass, structure, function, and fibrosis in the SPRINT-HEART. Hypertension (Dallas, Tex : 1979). 2019;74:276–84 Hypertensionaha11913073. https://doi.org/10.1161/hypertensionaha.119.13073.
Ekström M, Hellman A, Hasselström J, Hage C, Kahan T, Ugander M, et al. The transition from hypertension to hypertensive heart disease and heart failure: the PREFERS Hypertension study. ESC Heart Fail. 2020;7(2):737–46. https://doi.org/10.1002/ehf2.12612.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflicts of interest relevant to this manuscript.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Hypertension and the Heart
Rights and permissions
About this article
Cite this article
Stacey, R.B., Hundley, W.G. Integrating Measures of Myocardial Fibrosis in the Transition from Hypertensive Heart Disease to Heart Failure. Curr Hypertens Rep 23, 22 (2021). https://doi.org/10.1007/s11906-021-01135-8
Accepted:
Published:
DOI: https://doi.org/10.1007/s11906-021-01135-8