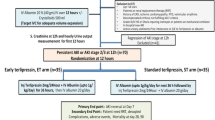
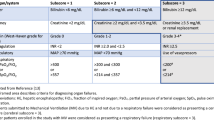
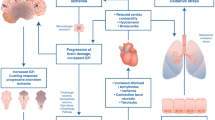
Abstract
Purpose of Review
Over the past decade, donation after circulatory death (DCD) liver transplantation has expanded in the United States due to improved surgical experience and perioperative management. Despite these advances, there remains a reluctance towards broader utilization of DCD liver allografts due to lack of standardized donation process, concern for inferior graft survival, and risk of ischemic cholangiopathy associated with temporary lack of oxygenated perfusion during withdrawal of life-supporting treatment during procurement.
Recent Findings
New perfusion technologies offer potential therapeutic options to mitigate biliary complications and expand utilization of marginal DCD grafts. As these modalities enter routine clinical practice, DCD utilization will continue to increase, and liver allocation policies in turn will evolve to reflect this growing practice.
Summary
This review describes recent progress in DCD LT, current challenges with utilization of DCD liver allografts, and how novel technologies and policies could impact the future of the field.



Similar content being viewed by others
Abbreviations
- CIT:
-
Cold ischemia time
- DBD:
-
Donation after brain death
- DCD:
-
Donation after circulatory death
- ERCP:
-
Endoscopic retrograde cholangiopancreatography
- EVMP:
-
Ex vivo machine perfusion
- HOPE:
-
Hypothermic oxygenated machine perfusion
- ITBS:
-
Ischemic type biliary strictures
- IC:
-
Ischemic cholangiopathy
- LT:
-
Liver transplantation
- MMaT:
-
Median MELD at transplant
- NRP:
-
Normothermic regional perfusion
- NMP:
-
Normothermic machine perfusion
- PTC:
-
Percutaneous transhepatic cholangiogram
- SCS:
-
Static cold storage
- Tpa:
-
Tissue plasminogen activator
- fWIT:
-
Functional warm ischemia time
- WIT:
-
Warm ischemia time
- WLST:
-
Withdrawal of life supporting treatment
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Thuong M, Ruiz A, Evrard P, et al. New classification of donation after circulatory death donors definitions and terminology. Transpl Int Off J Eur Soc Organ Transplant. 2016;29(7):749–59.
Foley DP, Fernandez LA, Leverson G, et al. Donation after cardiac death: the University of Wisconsin experience with liver transplantation. Ann Surg. 2005;242(5):724–31.
Skaro AI, Jay CL, Baker TB, et al. The impact of ischemic cholangiopathy in liver transplantation using donors after cardiac death: the untold story. Surg. 2009;146(4):543–52 (discussion 552–553).
Jay C, Ladner D, Wang E, et al. A comprehensive risk assessment of mortality following donation after cardiac death liver transplant - an analysis of the national registry. J Hepatol. 2011;55(4):808–13.
Haque O, Yuan Q, Uygun K, Markmann JF. Evolving utilization of donation after circulatory death livers in liver transplantation: The day of DCD has come. Clin Transplant. 2021;35(3): e14211.
Mathur AK, Heimbach J, Steffick DE, Sonnenday CJ, Goodrich NP, Merion RM. Donation after cardiac death liver transplantation: predictors of outcome. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2010;10(11):2512–9.
Cappucci SP, Smith WS, Schwartzstein R, White DB, Mitchell SL, Fehnel CR. End-Of-Life Care in the Potential Donor after Circulatory Death: A Systematic Review. The Neurohospitalist. 2023;13(1):61–8.
Haque OJ, Roth EM, Fleishman A, Eckhoff DE, Khwaja K. Long-Term Outcomes of Early Experience in Donation After Circulatory Death Liver Transplantation: Outcomes at 10 Years. Ann Transplant. 2021;26: e930243.
Mateo R, Cho Y, Singh G, et al. Risk factors for graft survival after liver transplantation from donation after cardiac death donors: an analysis of OPTN/UNOS data. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2006;6(4):791–6.
Croome KP, Taner CB. The Changing Landscapes in DCD Liver Transplantation. Curr Transplant Rep. 2020;7(3):194–204.
Reich DJ, Mulligan DC, Abt PL, et al. ASTS recommended practice guidelines for controlled donation after cardiac death organ procurement and transplantation. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2009;9(9):2004–11.
Andrews PA, Burnapp L, Manas D. British Transplantation Society. Summary of the British Transplantation Society guidelines for transplantation from donors after deceased circulatory death. Transplant. 2014;97(3):265–70.
Braat AE, Blok JJ, Putter H, et al. The Eurotransplant donor risk index in liver transplantation: ET-DRI. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2012;12(10):2789–96.
Kalisvaart M, Croome KP, Hernandez-Alejandro R, et al. Donor Warm Ischemia Time in DCD Liver Transplantation-Working Group Report From the ILTS DCD, Liver Preservation, and Machine Perfusion Consensus Conference. Transplant. 2021;105(6):1156–64.
Schlegel A, van Reeven M, Croome K, et al. A multicentre outcome analysis to define global benchmarks for donation after circulatory death liver transplantation. J Hepatol. 2022;76(2):371–82.
Croome KP, Lee DD, Perry DK, et al. Comparison of longterm outcomes and quality of life in recipients of donation after cardiac death liver grafts with a propensity-matched cohort. Liver Transplant Off Publ Am Assoc Study Liver Dis Int Liver Transplant Soc. 2017;23(3):342–51.
Laing RW, Scalera I, Isaac J, et al. Liver Transplantation Using Grafts From Donors After Circulatory Death: A Propensity Score-Matched Study From a Single Center. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2016;16(6):1795–804.
Croome KP, Mathur AK, Aqel B, et al. Classification of Distinct Patterns of Ischemic Cholangiopathy Following DCD Liver Transplantation: Distinct Clinical Courses and Long-term Outcomes From a Multicenter Cohort. Transplantation. 2022;106(6):1206–1214. This article describes the spectrum of ischemic cholangiopathy patterns and clarifies that not all instances of cholangiopathy result in graft failure.
Tung BY, Kimmey MB. Biliary complications of orthotopic liver transplantation. Dig Dis Basel Switz. 1999;17(3):133–44.
Mourad MM, Algarni A, Liossis C, Bramhall SR. Aetiology and risk factors of ischaemic cholangiopathy after liver transplantation. World J Gastroenterol. 2014;20(20):6159–69.
Allen AM, Kim WR, Xiong H, et al. Survival of recipients of livers from donation after circulatory death who are relisted and undergo retransplant for graft failure. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2014;14(5):1120–8.
Croome KP, Lee DD, Nguyen JH, Keaveny AP, Taner CB. Waitlist Outcomes for Patients Relisted Following Failed Donation After Cardiac Death Liver Transplant: Implications for Awarding Model for End-Stage Liver Disease Exception Scores. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2017;17(9):2420–7.
Hashimoto K, Eghtesad B, Gunasekaran G, et al. Use of tissue plasminogen activator in liver transplantation from donation after cardiac death donors. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2010;10(12):2665–72.
Seal JB, Bohorquez H, Reichman T, et al. Thrombolytic protocol minimizes ischemic-type biliary complications in liver transplantation from donation after circulatory death donors. Liver Transplant Off Publ Am Assoc Study Liver Dis Int Liver Transplant Soc. 2015;21(3):321–8.
Haque O, Raigani S, Rosales I, et al. Thrombolytic Therapy During ex-vivo Normothermic Machine Perfusion of Human Livers Reduces Peribiliary Vascular Plexus Injury. Front Surg. 2021;8: 644859.
Halldorson JB, Bakthavatsalam R, Montenovo M, et al. Differential rates of ischemic cholangiopathy and graft survival associated with induction therapy in DCD liver transplantation. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2015;15(1):251–8.
Ig-Izevbekhai K, Goldberg DS, Karp SJ, Foley DP, Abt PL. Immunosuppression in Donation After Circulatory Death Liver Transplantation: Can Induction Modify Graft Survival? Liver Transplant Off Publ Am Assoc Study Liver Dis Int Liver Transplant Soc. 2020;26(9):1154–66.
Starzl TE, Groth CG, Brettschneider L, et al. Orthotopic homotransplantation of the human liver. Ann Surg. 1968;168(3):392–415.
Magliocca JF, Magee JC, Rowe SA, et al. Extracorporeal support for organ donation after cardiac death effectively expands the donor pool. J Trauma. 2005;58(6):1095–101 (discussion 1101–1102).
Hunt F, Johnston CJC, Coutts L, et al. From Haphazard to a Sustainable Normothermic Regional Perfusion Service: A Blueprint for the Introduction of Novel Perfusion Technologies. Transpl Int Off J Eur Soc Organ Transplant. 2022;35:10493.
Hessheimer AJ, de la Rosa G, Gastaca M, et al. Abdominal normothermic regional perfusion in controlled donation after circulatory determination of death liver transplantation: Outcomes and risk factors for graft loss. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2022;22(4):1169–1181. This manuscript presents one of the largest experiences from the Spanish transplant registry, demonstrating superior outcomes.
Hessheimer AJ, Coll E, Torres F, et al. Normothermic regional perfusion vs. super-rapid recovery in controlled donation after circulatory death liver transplantation. J Hepatol. 2019;70(4):658–65.
Watson CJE, Hunt F, Messer S, et al. In situ normothermic perfusion of livers in controlled circulatory death donation may prevent ischemic cholangiopathy and improve graft survival. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2019;19(6):1745–58.
Mergental H, Perera MTPR, Laing RW, et al. Transplantation of Declined Liver Allografts Following Normothermic Ex-Situ Evaluation. Am J Transplant Off J Am Soc Transplant Am Soc Transpl Surg. 2016;16(11):3235–45.
Jassem W, Xystrakis E, Ghnewa YG, et al. Normothermic Machine Perfusion (NMP) Inhibits Proinflammatory Responses in the Liver and Promotes Regeneration. Hepatol Baltim Md. 2019;70(2):682–95.
Nasralla D, Coussios CC, Mergental H, et al. A randomized trial of normothermic preservation in liver transplantation. Nature. 2018;557(7703):50–56. This paper reports the first randomized controlled trial using ex vivo normothermic perfusion in liver transplantation.
Markmann JF, Abouljoud MS, Ghobrial RM, et al. Impact of Portable Normothermic Blood-Based Machine Perfusion on Outcomes of Liver Transplant: The OCS Liver PROTECT Randomized Clinical Trial. JAMA Surg. 2022;157(3):189–198. This paper rerports the first american randomized trial using normothermic perfusion in liver transplantation.
Czigany Z, Schöning W, Ulmer TF, et al. Hypothermic oxygenated machine perfusion (HOPE) for orthotopic liver transplantation of human liver allografts from extended criteria donors (ECD) in donation after brain death (DBD): a prospective multicentre randomised controlled trial (HOPE ECD-DBD). BMJ Open. 2017;7(10):e017558. This paper reports the first randomized trial using hypothermic machine perfusion in liver transplantation.
Schlegel A, Porte R, Dutkowski P. Protective mechanisms and current clinical evidence of hypothermic oxygenated machine perfusion (HOPE) in preventing post-transplant cholangiopathy. J Hepatol. 2022;76(6):1330–47.
Eden J, Sousa Da Silva R, Cortes-Cerisuelo M, et al. Utilization of livers donated after circulatory death for transplantation - An international comparison. J Hepatol. 2023;78(5):1007–1016. This excellent international comparison outlines some of the challenges that impede broader utilization of DCD.
Hessheimer AJ, Gastaca M, Miñambres E, Colmenero J, Fondevila C. in representation of the SETH Working Group on DCD. Donation after circulatory death liver transplantation: consensus statements from the Spanish Liver Transplantation Society. Transpl Int Off J Eur Soc Organ Transplant. 2020;33(8):902–16.
Funding
The authors have nothing to disclose.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to the review of the literature, writing of the manuscript and final review and approval of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Human and animals rights and informed consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Disclosures
None of the authors have any relevant potential conflicts of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Haque, O.J., Roth, E.M. & Lee, D.D. Modern-Day Practice of DCD Liver Transplantation: Controversies, Innovations, and Future Directions. Curr Gastroenterol Rep 25, 413–420 (2023). https://doi.org/10.1007/s11894-023-00902-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11894-023-00902-8