Abstract
Purpose of Review
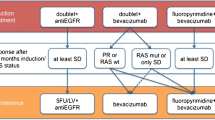
Current systemic management of MCRC should include periods of intensive and less intensive treatment or even complete stop. Different systemic post-induction strategies have been evaluated in many trials. The aim of this article is to review the available data on maintenance strategies in MCRC and potential options to personalize choice of the respective strategy.
Recent Findings
Despite the large variability of clinical trials conducted in this setting, it can be concluded that intermittent chemotherapy does not seem to be inferior to continuous chemotherapy if at least 3 months of intensive induction treatment is applied, and active maintenance seem to be superior to complete stop after at least 3 months of induction treatment in terms of PFS and may add some benefit in terms of OS. The choice of the respective maintenance strategy may be personalized taking into account disease and patient characteristic, choice of induction treatment and response, treatment tolerability and quality of life.
Summary
Patients with metastatic colorectal cancer and no options of secondary resection or local ablation should be considered for a personalized maintenance approach.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Global Burden of Disease Cancer C, Fitzmaurice C, Dicker D, Pain A, Hamavid H, Moradi-Lakeh M, et al. The global burden of cancer 2013. JAMA oncology. 2015;1(4):505–27. doi:10.1001/jamaoncol.2015.0735.
Malvezzi M, Carioli G, Bertuccio P, Rosso T, Boffetta P, Levi F, et al. European cancer mortality predictions for the year 2016 with focus on leukaemias. Annals of oncology : official journal of the European Society for Medical Oncology / ESMO. 2016;27(4):725–31. doi:10.1093/annonc/mdw022.
Siegel R, Desantis C, Jemal A. Colorectal cancer statistics, 2014. CA Cancer J Clin. 2014;64(2):104–17. doi:10.3322/caac.21220.
• Van Cutsem E, Cervantes A, Adam R, Sobrero A, Van Krieken JH, Aderka D, et al. ESMO consensus guidelines for the management of patients with metastatic colorectal cancer. Ann Oncol. 2016;27(8):1386–422. doi:10.1093/annonc/mdw235. The ESMO consensus guidelines have been developed based on the current available evidence to provide a series of expert recommendations to assist in the treatment and management of patients with metastatic colorectal cancer in this rapidly evolving treatment setting.
Cremolini C, Loupakis F, Antoniotti C, Lupi C, Sensi E, Lonardi S, et al. FOLFOXIRI plus bevacizumab versus FOLFIRI plus bevacizumab as first-line treatment of patients with metastatic colorectal cancer: updated overall survival and molecular subgroup analyses of the open-label, phase 3 TRIBE study. Lancet Oncol. 2015; doi:10.1016/S1470-2045(15)00122-9.
Douillard JY, Oliner KS, Siena S, Tabernero J, Burkes R, Barugel M, et al. Panitumumab-FOLFOX4 treatment and RAS mutations in colorectal cancer. N Engl J Med. 2013;369(11):1023–34. doi:10.1056/NEJMoa1305275.
Van Cutsem E, Lenz HJ, Kohne CH, Heinemann V, Tejpar S, Melezinek I, et al. Fluorouracil, leucovorin, and irinotecan plus cetuximab treatment and RAS mutations in colorectal cancer. J Clin Oncol. 2015;33(7):692–700. doi:10.1200/JCO.2014.59.4812.
Heinemann V, von Weikersthal LF, Decker T, Kiani A, Vehling-Kaiser U, Al-Batran SE, et al. FOLFIRI plus cetuximab versus FOLFIRI plus bevacizumab as first-line treatment for patients with metastatic colorectal cancer (FIRE-3): a randomised, open-label, phase 3 trial. Lancet Oncol. 2014;15(10):1065–75. doi:10.1016/S1470-2045(14)70330-4.
Saltz LB, Clarke S, Diaz-Rubio E, Scheithauer W, Figer A, Wong R, et al. Bevacizumab in combination with oxaliplatin-based chemotherapy as first-line therapy in metastatic colorectal cancer: a randomized phase III study. J Clin Oncol. 2008;26(12):2013–9.
Maughan TS, James RD, Kerr DJ, Ledermann JA, Seymour MT, Topham C, et al. Comparison of intermittent and continuous palliative chemotherapy for advanced colorectal cancer: a multicentre randomised trial. Lancet. 2003;361(9356):457–64.
Adams RA, Meade AM, Seymour MT, Wilson RH, Madi A, Fisher D, et al. Intermittent versus continuous oxaliplatin and fluoropyrimidine combination chemotherapy for first-line treatment of advanced colorectal cancer: results of the randomised phase 3 MRC COIN trial. The Lancet Oncology. 2011;12(7):642–53. doi:10.1016/S1470-2045(11)70102-4.
Tournigand C, Cervantes A, Figer A, Lledo G, Flesch M, Buyse M, et al. OPTIMOX1: a randomized study of FOLFOX4 or FOLFOX7 with oxaliplatin in a stop-and-go fashion in advanced colorectal cancer—a GERCOR study. J Clin Oncol. 2006;24(3):394–400. doi:10.1200/JCO.2005.03.0106.
Labianca R, Sobrero A, Isa L, Cortesi E, Barni S, Nicolella D, et al. Intermittent versus continuous chemotherapy in advanced colorectal cancer: a randomised ‘GISCAD’ trial. Annals of oncology : official journal of the European Society for Medical Oncology / ESMO. 2011;22(5):1236–42. doi:10.1093/annonc/mdq580.
Diaz-Rubio E, Gomez-Espana A, Massuti B, Sastre J, Abad A, Valladares M, et al. First-line XELOX plus bevacizumab followed by XELOX plus bevacizumab or single-agent bevacizumab as maintenance therapy in patients with metastatic colorectal cancer: the phase III MACRO TTD study. Oncologist. 2012;17(1):15–25. doi:10.1634/theoncologist.2011-0249.
Yalcin S, Uslu R, Dane F, Yilmaz U, Zengin N, Buyukunal E, et al. Bevacizumab + capecitabine as maintenance therapy after initial bevacizumab + XELOX treatment in previously untreated patients with metastatic colorectal cancer: phase III ‘Stop and Go’ study results—a Turkish oncology group trial. Oncology. 2013;85(6):328–35. doi:10.1159/000355914.
Hochster HS, Grothey A, Hart L, Rowland K, Ansari R, Alberts S, et al. Improved time to treatment failure with an intermittent oxaliplatin strategy: results of CONcePT. Ann Oncol. 2014;25(6):1172–8. doi:10.1093/annonc/mdu107.
Pfeiffer P, Sorbye H, Qvortrup C, Karlberg M, Kersten C, Vistisen K, et al. Maintenance therapy with cetuximab every second week in the first-line treatment of metastatic colorectal cancer: the NORDIC-7.5 study by the nordic colorectal cancer biomodulation group. Clin Colorectal Cancer. 2015;14(3):170–6. doi:10.1016/j.clcc.2015.03.002.
Tveit KM, Guren T, Glimelius B, Pfeiffer P, Sorbye H, Pyrhonen S, et al. Phase III trial of cetuximab with continuous or intermittent fluorouracil, leucovorin, and oxaliplatin (Nordic FLOX) versus FLOX alone in first-line treatment of metastatic colorectal cancer: the NORDIC-VII study. J Clin Oncol. 2012;30(15):1755–62. doi:10.1200/JCO.2011.38.0915.
Chibaudel B, Maindrault-Goebel F, Lledo G, Mineur L, Andre T, Bennamoun M, et al. Can chemotherapy be discontinued in unresectable metastatic colorectal cancer? The GERCOR OPTIMOX2 study. J Clin Oncol. 2009;27(34):5727–33. doi:10.1200/JCO.2009.23.4344.
• Simkens LH, van Tinteren H, May A, Ten Tije AJ, Creemers GJ, Loosveld OJ, et al. Maintenance treatment with capecitabine and bevacizumab in metastatic colorectal cancer (CAIRO3): a phase 3 randomised controlled trial of the Dutch colorectal cancer group. Lancet. 2015;385(9980):1843–52. doi:10.1016/S0140-6736(14)62004-3. The CAIRO3 study was designed to determine the efficacy of maintenance treatment with capecitabine plus bevacizumab versus observation. It showed that maintenance treatment with capecitabine plus bevacizumab after six cycles of CAPOX-B in patients with metastatic colorectal cancer prolongs progression free survival and does not compromise quality of life.
Koeberle D, Betticher DC, von Moos R, Dietrich D, Brauchli P, Baertschi D, et al. Bevacizumab continuation versus no continuation after first-line chemotherapy plus bevacizumab in patients with metastatic colorectal cancer: a randomized phase III non-inferiority trial (SAKK 41/06). Ann Oncol. 2015;26(4):709–14. doi:10.1093/annonc/mdv011.
• Hegewisch-Becker S, Graeven U, Lerchenmuller CA, Killing B, Depenbusch R, Steffens CC, et al. Maintenance strategies after first-line oxaliplatin plus fluoropyrimidine plus bevacizumab for patients with metastatic colorectal cancer (AIO 0207): a randomised, non-inferiority, open-label, phase 3 trial. Lancet Oncol. 2015; doi:10.1016/S1470-2045(15)00042-X. Randomized trial investigating different maintenance strategies after first-line induction treatment with fluoropyrimidine, oxaliplatin and bevacizumab. An active maintenance treatment, either with a fluoropyrimidine plus bevacizumab or with bevacizumab alone, is associated with a longer progression free survival, but choice of maintenance or post-induction strategy does not affect overall survival.
Stein A, Schwenke C, Folprecht G, Arnold D. Effect of application and intensity of Bevacizumab-based maintenance after induction chemotherapy with Bevacizumab for metastatic colorectal cancer: a meta-analysis. Clin Colorectal Cancer. 2016;15(2):e29–39. doi:10.1016/j.clcc.2015.12.005.
Wasan H, Meade AM, Adams R, Wilson R, Pugh C, Fisher D, et al. Intermittent chemotherapy plus either intermittent or continuous cetuximab for first-line treatment of patients with KRAS wild-type advanced colorectal cancer (COIN-B): a randomised phase 2 trial. Lancet Oncol. 2014;15(6):631–9. doi:10.1016/S1470-2045(14)70106-8.
Tournigand C, Chibaudel B, Samson B, Scheithauer W, Vernerey D, Mesange P, et al. Bevacizumab with or without erlotinib as maintenance therapy in patients with metastatic colorectal cancer (GERCOR DREAM; OPTIMOX3): a randomised, open-label, phase 3 trial. Lancet Oncol. 2015;16(15):1493–505. doi:10.1016/S1470-2045(15)00216-8.
Johnsson A, Hagman H, Frodin JE, Berglund A, Keldsen N, Fernebro E, et al. A randomized phase III trial on maintenance treatment with bevacizumab alone or in combination with erlotinib after chemotherapy and bevacizumab in metastatic colorectal cancer: the Nordic ACT trial. Ann Oncol. 2013;24(9):2335–41. doi:10.1093/annonc/mdt236.
Goey KKH, Elias SG, Van Tinteren H. Predictive value of KRAS mutation status in metastatic colorectal cancer (mCRC) patients treated with capecitabine and bevacizumab (CAP-B) maintenance treatment vs observation in the phase III CAIRO3 study. J Clin Oncol. 2016;34(suppl):abstr 3525.
• Quidde J, Hegewisch-Becker S, Graeven U, Lerchenmuller CA, Killing B, Depenbusch R, et al. Quality of life assessment in patients with metastatic colorectal cancer receiving maintenance therapy after first-line induction treatment: a preplanned analysis of the phase III AIO KRK 0207 trial. Ann Oncol. 2016;27(12):2203–10. doi:10.1093/annonc/mdw425. This was one of the first studies analysing quality of life during maintenance treatment. The study showed that continuation of an active maintenance treatment with fluoropyrimidine and bevacizumab after induction treatment was neither associated with a detrimental effect on health related quality of life when compared to bevacizumab alone or no active treatment.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Julia Quidde has received compensation from Servier for service as a consultant.
Alexander Stein has received research funding through grants from Roche, Sanofi, and Merck, and has received compensation from Roche, Sanofi, Merck, Amgen, Lilly, Bayer, and Servier for service as a consultant.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Topical Collection on Personalized Medicine in Colorectal Cancer
Rights and permissions
About this article
Cite this article
Quidde, J., Stein, A. Personalizing Maintenance Therapy in Metastatic Colorectal Cancer. Curr Colorectal Cancer Rep 13, 205–211 (2017). https://doi.org/10.1007/s11888-017-0365-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11888-017-0365-6