Abstract
Purpose of Review
Targeting traditional cardiovascular risk factors is effective in reducing recurrent cardiovascular events, yet the presence of residual cardiovascular risk due to underlying systemic inflammation is a largely unaddressed opportunity. This review aims to comprehensively assess the evolving role of colchicine as a therapeutic approach targeting residual inflammatory risk in the context of those with coronary artery disease (CAD).
Recent Findings
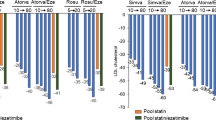
Inflammation plays a significant role in promoting atherosclerosis, and targeting anti-inflammatory pathways has the potential to decrease cardiovascular events. Low-dose colchicine (0.5 mg/day orally), when added to guideline-directed medical care for CAD, safely decreases major adverse cardiovascular events (MACE) by 31% in stable atherosclerosis patients and 23% in those after recent myocardial infarctions. Meta-analyses of recent randomized control trials further support both the efficacy and safety of colchicine, particularly when added to other standard cardiovascular therapies, including statin therapy. The European Society of Cardiology and other national guidelines endorse the use of low-dose colchicine in patients across the spectrum of CAD. Recently, colchicine was FDA-approved in the United States as the first anti-inflammatory therapy for the reduction of cardiovascular events. In a period of a rising incidence of CAD across the globe, colchicine represents a unique opportunity to decrease MACE due to its large magnitude of benefits and general affordability. However, challenges with drug interactions must be addressed, especially in those regions where HIV, hepatitis, and tuberculosis are prevalent.
Summary
Colchicine is safe and effective at reducing cardiovascular events across a broad spectrum of coronary syndromes. The ability to simultaneously target traditional risk factors and mitigate residual inflammatory risk marks a substantial advancement in cardiovascular prevention strategies, heralding a new era in the global battle against CAD.



Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Roth GA, Mensah GA, Johnson CO, Addolorato G, Ammirati E, Baddour LM, et al. Global burden of cardiovascular diseases and risk factors, 1990–2019: update from the GBD 2019 Study. J Am Coll Cardiol. 2020;76(25):2982–3021. https://doi.org/10.1016/j.jacc.2020.11.010.
Zhao D. Epidemiological features of cardiovascular disease in Asia. JACC: Asia. 2021;1(1):1–13. https://doi.org/10.1016/j.jacasi.2021.04.007.
Das Pradhan A, Glynn RJ, Fruchart J-C, MacFadyen JG, Zaharris ES, Everett BM, et al. Triglyceride lowering with pemafibrate to reduce cardiovascular risk. N Engl J Med. 2022;387(21):1923–34. https://doi.org/10.1056/NEJMoa2210645.
Bhatt DL, Steg PG, Miller M, Brinton EA, Jacobson TA, Ketchum SB, et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med. 2018;380(1):11–22. https://doi.org/10.1056/NEJMoa1812792.
Nicholls SJ, Lincoff AM, Garcia M, Bash D, Ballantyne CM, Barter PJ, et al. Effect of high-dose omega-3 fatty acids vs corn oil on major adverse cardiovascular events in patients at high cardiovascular risk: the STRENGTH randomized clinical trial. JAMA. 2020;324(22):2268–80. https://doi.org/10.1001/jama.2020.22258.
Ridker PM, Danielson E, Fonseca FAH, Genest J, Gotto AM, Kastelein JJP, et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med. 2008;359(21):2195–207. https://doi.org/10.1056/NEJMoa0807646.
Ridker PM, Everett BM, Pradhan A, MacFadyen JG, Solomon DH, Zaharris E, et al. Low-dose methotrexate for the prevention of atherosclerotic events. N Engl J Med. 2018;380(8):752–62. https://doi.org/10.1056/NEJMoa1809798.
Nicholls SJ, Kastelein JJP, Schwartz GG, Bash D, Rosenson RS, Cavender MA, et al. Varespladib and cardiovascular events in patients with an acute coronary syndrome: the VISTA-16 randomized clinical trial. JAMA. 2014;311(3):252–62. https://doi.org/10.1001/jama.2013.282836.
O’Donoghue ML, Braunwald E, White HD, Steen DL, Lukas MA, Tarka E, et al. Effect of darapladib on major coronary events after an acute coronary syndrome: the SOLID-TIMI 52 randomized clinical trial. JAMA. 2014;312(10):1006–15. https://doi.org/10.1001/jama.2014.11061.
Darapladib for preventing ischemic events in stable coronary heart disease. New Eng J Med. 2014;370(18):1702–11. https://doi.org/10.1056/NEJMoa1315878.
Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med. 2017;377(12):1119–31. https://doi.org/10.1056/NEJMoa1707914.
Dasgeb B, Kornreich D, McGuinn K, Okon L, Brownell I, Sackett DL. Colchicine: an ancient drug with novel applications. Br J Dermatol. 2018;178(2):350–6. https://doi.org/10.1111/bjd.15896.
Nerlekar N, Beale A, Harper RW. Colchicine–a short history of an ancient drug. Med J Aust. 2014;201(11):687–8. https://doi.org/10.5694/mja14.00846.
Adler Y, Charron P, Imazio M, Badano L, Barón-Esquivias G, Bogaert J, et al. 2015 ESC Guidelines for the diagnosis and management of pericardial diseases: the task force for the diagnosis and management of pericardial diseases of the European Society of Cardiology (ESC)endorsed by: The European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2015;36(42):2921–64. https://doi.org/10.1093/eurheartj/ehv318.
Esatoglu SN, Hatemi G. Update on the treatment of Behçet’s syndrome. Intern Emerg Med. 2019;14(5):661–75. https://doi.org/10.1007/s11739-019-02035-1.
Ozen S, Demirkaya E, Erer B, Livneh A, Ben-Chetrit E, Giancane G, et al. EULAR recommendations for the management of familial Mediterranean fever. Ann Rheum Dis. 2016;75(4):644–51. https://doi.org/10.1136/annrheumdis-2015-208690.
Chiabrando JG, Bonaventura A, Vecchié A, Wohlford GF, Mauro AG, Jordan JH, et al. Management of acute and recurrent pericarditis. J Am Coll Cardiol. 2020;75(1):76–92. https://doi.org/10.1016/j.jacc.2019.11.021.
Libby P, Bonow R, Mann D, Tomaselli G, Bhatt D, Solomon S. Braunwald’s heart disease : a textbook of cardiovascular medicine. 12th ed. Philadelphia: Elsevier; 2021.
Fernandez DM, Rahman AH, Fernandez NF, Chudnovskiy A, Amir ED, Amadori L, et al. Single-cell immune landscape of human atherosclerotic plaques. Nat Med. 2019;25(10):1576–88. https://doi.org/10.1038/s41591-019-0590-4.
Leung YY, Yao Hui LL, Kraus VB. Colchicine–update on mechanisms of action and therapeutic uses. Semin Arthritis Rheum. 2015;45(3):341–50. https://doi.org/10.1016/j.semarthrit.2015.06.013.
Martínez GJ, Celermajer DS, Patel S. The NLRP3 inflammasome and the emerging role of colchicine to inhibit atherosclerosis-associated inflammation. Atherosclerosis. 2018;269:262–71. https://doi.org/10.1016/j.atherosclerosis.2017.12.027.
Richette P, Frazier A, Bardin T. Pharmacokinetics considerations for gout treatments. Expert Opin Drug Metab Toxicol. 2014;10(7):949–57. https://doi.org/10.1517/17425255.2014.915027.
Slobodnick A, Shah B, Pillinger MH, Krasnokutsky S. Colchicine: old and new. Am J Med. 2015;128(5):461–70. https://doi.org/10.1016/j.amjmed.2014.12.010.
Niel E, Scherrmann J-M. Colchicine today. Joint Bone Spine. 2006;73(6):672–8. https://doi.org/10.1016/j.jbspin.2006.03.006.
Chappey ON, Niel E, Wautier JL, Hung PP, Dervichian M, Cattan D, et al. Colchicine disposition in human leukocytes after single and multiple oral administration. Clin Pharmacol Ther. 1993;54(4):360–7. https://doi.org/10.1038/clpt.1993.161.
Robinson PC, Terkeltaub R, Pillinger MH, Shah B, Karalis V, Karatza E, et al. Consensus statement regarding the efficacy and safety of long-term low-dose colchicine in gout and cardiovascular disease. Am J Med. 2022;135(1):32–8. https://doi.org/10.1016/j.amjmed.2021.07.025.
Rochdi M, Sabouraud A, Girre C, Venet R, Scherrmann JM. Pharmacokinetics and absolute bioavailability of colchicine after i.v. and oral administration in healthy human volunteers and elderly subjects. Eur J Clin Pharmacol. 1994;46(4):351–4. https://doi.org/10.1007/bf00194404.
Wiggins BS, Saseen JJ, Page RL, Reed BN, Sneed K, Kostis JB, et al. Recommendations for management of clinically significant drug-drug interactions with statins and select agents used in patients with cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2016;134(21):e468–95. https://doi.org/10.1161/CIR.0000000000000456.
•• Tardif J-C, Kouz S, Waters DD, Bertrand OF, Diaz R, Maggioni AP, et al. Efficacy and safety of low-dose colchicine after myocardial infarction. N Engl J Med. 2019;381(26):2497–505. https://doi.org/10.1056/NEJMoa1912388. In this randomized control trial, among patients who experienced myocardial infarction within the past 30 days, the use of colchicine was associated with a decrease in the primary composite endpoint, encompassing death from cardiovascular causes, resuscitated cardiac arrest, recurrent MI, stroke, or urgent coronary revascularization, over a median follow-up period of 22.6 months.
•• Nidorf SM, Fiolet ATL, Mosterd A, Eikelboom JW, Schut A, Opstal TSJ, et al. Colchicine in patients with chronic coronary disease. N Engl J Med. 2020;383(19):1838–47. https://doi.org/10.1056/NEJMoa2021372. The results of this randomized control trial reveal that among individuals with chronic coronary disease, colchicine led to a significant decrease in the composite outcome encompassing cardiovascular death, spontaneous myocardial infarction, ischemic stroke, or ischemia-driven coronary revascularization throughout a median follow-up period of 28.6 months.
Samuel M, Tardif JC, Bouabdallaoui N, Khairy P, Dubé MP, Blondeau L, et al. Colchicine for secondary prevention of cardiovascular disease: a systematic review and meta-analysis of randomized controlled trials. Can J Cardiol. 2021;37(5):776–85. https://doi.org/10.1016/j.cjca.2020.10.006.
Andreis A, Imazio M, Avondo S, Casula M, Paneva E, Piroli F, et al. Adverse events of colchicine for cardiovascular diseases: a comprehensive meta-analysis of 14 188 patients from 21 randomized controlled trials. J Cardiovasc Med. 2021;22(8):637–44. https://doi.org/10.2459/jcm.0000000000001157.
Stewart S, Yang KCK, Atkins K, Dalbeth N, Robinson PC. Adverse events during oral colchicine use: a systematic review and meta-analysis of randomised controlled trials. Arthritis Res Ther. 2020;22(1):28. https://doi.org/10.1186/s13075-020-2120-7.
Tong DC, Quinn S, Nasis A, Hiew C, Roberts-Thomson P, Adams H, et al. Colchicine in Patients with acute coronary syndrome. Circulation. 2020;142(20):1890–900. https://doi.org/10.1161/CIRCULATIONAHA.120.050771.
Galli M, Princi G, Crea F, D’Amario D. Colchicine and risk of non-cardiovascular death in patients with coronary artery disease: a pooled analysis underling possible safety concerns. European Heart Journal - Cardiovascular Pharmacotherapy. 2020;7(3):e18–9. https://doi.org/10.1093/ehjcvp/pvaa137.
Finkelstein Y, Aks SE, Hutson JR, Juurlink DN, Nguyen P, Dubnov-Raz G, et al. Colchicine poisoning: the dark side of an ancient drug. Clin Toxicol (Phila). 2010;48(5):407–14. https://doi.org/10.3109/15563650.2010.495348.
Crittenden DB, Lehmann RA, Schneck L, Keenan RT, Shah B, Greenberg JD, et al. Colchicine use is associated with decreased prevalence of myocardial infarction in patients with gout. J Rheumatol. 2012;39(7):1458–64. https://doi.org/10.3899/jrheum.111533.
Shah B, Toprover M, Crittenden DB, Jeurling S, Pike VC, Krasnokutsky S, et al. Colchicine use and incident coronary artery disease in male patients with gout. Can J Cardiol. 2020;36(11):1722–8. https://doi.org/10.1016/j.cjca.2020.05.026.
Solomon DH, Liu CC, Kuo IH, Zak A, Kim SC. Effects of colchicine on risk of cardiovascular events and mortality among patients with gout: a cohort study using electronic medical records linked with Medicare claims. Ann Rheum Dis. 2016;75(9):1674–9. https://doi.org/10.1136/annrheumdis-2015-207984.
Langevitz P, Livneh A, Neumann L, Buskila D, Shemer J, Amolsky D, et al. Prevalence of ischemic heart disease in patients with familial Mediterranean fever. Isr Med Assoc J. 2001;3(1):9–12.
Vaidya K, Arnott C, Martínez GJ, Ng B, McCormack S, Sullivan DR, et al. Colchicine therapy and plaque stabilization in patients with acute coronary syndrome: a CT coronary angiography study. JACC Cardiovasc Imaging. 2018;11(2 Pt 2):305–16. https://doi.org/10.1016/j.jcmg.2017.08.013.
Nidorf SM, Eikelboom JW, Budgeon CA, Thompson PL. Low-dose colchicine for secondary prevention of cardiovascular disease. J Am Coll Cardiol. 2013;61(4):404–10. https://doi.org/10.1016/j.jacc.2012.10.027.
Visseren FLJ, Mach F, Smulders YM, Carballo D, Koskinas KC, Bäck M, et al. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice: developed by the Task Force for cardiovascular disease prevention in clinical practice with representatives of the European Society of Cardiology and 12 medical societies With the special contribution of the European Association of Preventive Cardiology (EAPC). Eur Heart J. 2021;42(34):3227–337. https://doi.org/10.1093/eurheartj/ehab484.
Ponte-Negretti CI, Wyss FS, Piskorz D, Santos RD, Villar R, Lorenzatti A, et al. Latin American Consensus on management of residual cardiometabolic risk. A consensus paper prepared by the Latin American Academy for the Study of Lipids and Cardiometabolic Risk (ALALIP) endorsed by the Inter-American Society of Cardiology (IASC), the International Atherosclerosis Society (IAS), and the Pan-American College of Endothelium (PACE). Arch Cardiol Mex. 2022;92(1):99–112. https://doi.org/10.24875/acm.21000005.
Sabatine MS, Giugliano RP, Keech AC, Honarpour N, Wiviott SD, Murphy SA, et al. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med. 2017;376(18):1713–22. https://doi.org/10.1056/NEJMoa1615664.
Schwartz GG, Steg PG, Szarek M, Bhatt DL, Bittner VA, Diaz R, et al. Alirocumab and cardiovascular outcomes after acute coronary syndrome. N Engl J Med. 2018;379(22):2097–107. https://doi.org/10.1056/NEJMoa1801174.
Cannon CP, Blazing MA, Giugliano RP, McCagg A, White JA, Theroux P, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372(25):2387–97. https://doi.org/10.1056/NEJMoa1410489.
Nissen SE, Lincoff AM, Brennan D, Ray KK, Mason D, Kastelein JJP, et al. Bempedoic acid and cardiovascular outcomes in statin-intolerant patients. N Engl J Med. 2023;388(15):1353–64. https://doi.org/10.1056/NEJMoa2215024.
Marx N, Husain M, Lehrke M, Verma S, Sattar N. GLP-1 receptor agonists for the reduction of atherosclerotic cardiovascular risk in patients with type 2 diabetes. Circulation. 2022;146(24):1882–94. https://doi.org/10.1161/CIRCULATIONAHA.122.059595.
Cowie MR, Fisher M. SGLT2 inhibitors: mechanisms of cardiovascular benefit beyond glycaemic control. Nat Rev Cardiol. 2020;17(12):761–72. https://doi.org/10.1038/s41569-020-0406-8.
Eikelboom JW, Connolly SJ, Bosch J, Dagenais GR, Hart RG, Shestakovska O, et al. Rivaroxaban with or without aspirin in stable cardiovascular disease. N Engl J Med. 2017;377(14):1319–30. https://doi.org/10.1056/NEJMoa1709118.
Beezer J, Al Hatrushi M, Husband A, Kurdi A, Forsyth P. Polypharmacy definition and prevalence in heart failure: a systematic review. Heart Fail Rev. 2022;27(2):465–92. https://doi.org/10.1007/s10741-021-10135-4.
Ly DP, Giuriato MA, Song Z. Changes in prescription drug and health care use over 9 years after the large drug price increase for colchicine. JAMA Intern Med. 2023;183(7):670–6. https://doi.org/10.1001/jamainternmed.2023.0898.
Funding
The authors did not receive support from any organization for the submitted work. Brittany Weber reports support from NIH K23 and AHA Career Developmental Award, outside the submitted work.
Author information
Authors and Affiliations
Contributions
R.S.Z wrote the main manuscript text and prepared the figures. All authors reviewed the manuscript and made meaningful contributions.
Corresponding author
Ethics declarations
Conflict of Interest
Brittany Weber reports being on the Scientific Advisory Boards for Kiniksa, NovoNordisk, BMS, Horizon, and Agepha; honoraria from AWIR and ACR 2023 and ACR 2024; and Esperion Women Leadership Board. Michael S. Garshick reports consultant fees from Agepha, Kiniksa, BMS, and Horizon Pharmaceuticals (part of Amgen). The other authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
No animal or human subjects by the authors were used in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, R.S., Weber, B.N., Araiza-Garaygordobil, D. et al. Colchicine for the Prevention of Cardiovascular Disease: Potential Global Implementation. Curr Cardiol Rep (2024). https://doi.org/10.1007/s11886-024-02049-y
Accepted:
Published:
DOI: https://doi.org/10.1007/s11886-024-02049-y