Abstract
Purpose of Review
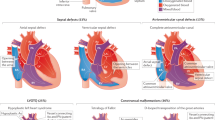
Approximately 30% of syndromic cases diagnosed with CHD, which lure us to further investigate the molecular and clinical challenges behind syndromic CHD (sCHD). The aetiology of sCHD in a majority of cases remains enigmatic due to involvement of multiple factors, namely genetic, epigenetic and environmental modifiable risk factors for the development of the disease. Here, we aim to update the role of genetic contributors including chromosomal abnormalities, copy number variations (CNVs) and single gene mutations in cardiac specific genes, maternal lifestyle conditions, environmental exposures and epigenetic modifiers in causing CHD in different genetic syndromes.
Recent Findings
The exact aetiology of sCHD is still unknown. With the advancement of next-generation technologies including WGS, WES, transcriptome, proteome and methylome study, numerous novel genes and pathways have been identified. Moreover, our recent knowledge regarding epigenetic and environmental regulation during cardiogenesis is still evolving and may solve some of the mystery behind complex sCHD.
Summary
Here, we focus to understand how the complex combination of genetic, environmental and epigenetic factors interact to interfere with developmental pathways, culminating into cardiac and extracardiac defects in sCHD.
Similar content being viewed by others
Data Availability
There are different databases such as American and Denmark databases which keep record for the birth surveillance as well as genetic counselling. However, post natal-care as well as improvement in clinical research may aid in better understanding the etiology of disease. The susceptibility to disease may also decrease.
Abbreviations
- ALGS:
-
Alagille syndrome
- AS:
-
Aortic stenosis
- ASD:
-
Atrial septal defect
- AVSD:
-
Atrioventricular septal defect
- BAV:
-
Bicuspid aortic valve
- CdC:
-
Cri-du-chat syndrome
- CNVs:
-
Copy number variations
- CoA:
-
Coarctation of aorta
- DCM:
-
Dilated cardiomyopathy
- DILV:
-
Double inlet left ventricle
- DORV:
-
Double outlet right ventricle
- DS:
-
Down syndrome
- EA:
-
Ebstein’s anomaly
- HCM:
-
Hypertrophic cardiomyopathy
- HLHS:
-
Hypoplastic left heart syndrome
- HOS:
-
Holt-Oram syndrome
- IAA:
-
Interrupted aortic arch
- JS:
-
Jacobsen syndrome
- LVOTO:
-
Left ventricular outflow tract obstruction
- NS:
-
Noonan syndrome
- NGS:
-
Next-generation sequencing
- OFT:
-
Outflow tract
- PAPVR:
-
Partial anomalous pulmonary venous return
- PDA:
-
Patent ductus arteriosus
- PS:
-
Pulmonary stenosis
- PTA:
-
Persistent truncus arteriosus
- SNVs:
-
Single nucleotide variants
- SVAS:
-
Supravalvular aortic stenosis
- TA:
-
Tricuspid atresia
- TAPVR:
-
Total anomalous pulmonary venous return
- TGA:
-
Transposition of great arteries
- ToF:
-
Tetralogy of Fallot
- VSD:
-
Ventricular septal defect
- WS:
-
Williams syndrome
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Van Der Linde D, Konings EE, Slager MA, Witsenburg M, Helbing WA, Takkenberg JJ, Roos-Hesselink JW. Birth prevalence of congenital heart disease worldwide: a systematic review and meta-analysis. J Am Coll Cardiol. 2011;58:2241–7.
Thomford NE, Dzobo K, Yao NA, Chimusa E, Evans J, Okai E, Kruszka P, Muenke M, Awandare G, Wonkam A. Genomics and epigenomics of congenital heart defects: expert review and lessons learned in Africa. OMICS: A J Integr Biol. 2018;22:301–321.
Ito S, Chapman KA, Kisling M, John AS. Genetic considerations for adults with congenital heart disease. In: American Journal of Medical Genetics Part C: Seminars in Medical Genetics. Wiley Online Library. 2020:149–153.
Nees SN, Chung WK. Genetic basis of human congenital heart disease. Cold Spring Harb Perspect Biol. 2020;12: a036749.
Nakajima Y. Retinoic acid signaling in heart development. Genesis. 2019;57: e23300.
Linglart L, Bonnet D. Epigenetics and congenital heart diseases. Journal of Cardiovascular Development and Disease. 2022;9:185.
Huang AC, Olson SB, Maslen CL. A review of recent developments in Turner syndrome research. Journal of cardiovascular development and disease. 2021;8:138.
Gelb BD, Chung WK. Complex genetics and the etiology of human congenital heart disease. Cold Spring Harb Perspect Med. 2014;4: a013953.
A Richards A, Garg V. Genetics of congenital heart disease. Curr Cardiol Rev. 2010;6:91-97.
Formigari R, Michielon G, Digilio MC, Piacentini G, Carotti A, Giardini A, Di Donato RM, Marino B. Genetic syndromes and congenital heart defects: how is surgical management affected? Eur J Cardiothorac Surg. 2009;35:606–14.
Digilio MC, Marino B. What is new in genetics of congenital heart defects? Front Pediatr. 2016;4:120.
Goldmuntz E. The epidemiology and genetics of congenital heart disease. Clin Perinatol. 2001;28:1–10.
Russell MW, Chung WK, Kaltman JR, Miller TA. Advances in the understanding of the genetic determinants of congenital heart disease and their impact on clinical outcomes. J Am Heart Assoc. 2018;7: e006906.
Down JLH. Observations on an ethnic classification of idiots. London hospital reports. 1866;3:259–62.
Bergström S, Carr H, Petersson G, Stephansson O, Bonamy A-KE, Dahlström A, Halvorsen CP, Johansson S. Trends in congenital heart defects in infants with Down syndrome. Pediatrics 2016:138.
Freeman SB, Bean LH, Allen EG, Tinker SW, Locke AE, Druschel C, Hobbs CA, Romitti PA, Royle MH, Torfs CP. Ethnicity, sex, and the incidence of congenital heart defects: a report from the National Down Syndrome Project. Genet Med. 2008;10:173–80.
Ackerman C, Locke AE, Feingold E, Reshey B, Espana K, Thusberg J, Mooney S, Bean LJ, Dooley KJ, Cua CL. An excess of deleterious variants in VEGF-A pathway genes in Down-syndrome-associated atrioventricular septal defects. Am J Hum Genet. 2012;91:646–59.
Moran R, Robin NH. Congenital heart defects. In: Emery and Rimoin’s Principles and practice of medical genetics and genomics. Elsevier. 2020:3–75.
• Trevino CE, Holleman AM, Corbitt H, Maslen CL, Rosser TC, Cutler DJ, Johnston HR, Rambo-Martin BL, Oberoi J, Dooley KJ. Identifying genetic factors that contribute to the increased risk of congenital heart defects in infants with Down syndrome. Sci Rep. 2020;10:1–12. Finding from this study showed that Nitch4 as well as genes involved in the ciliome might play a role in causing AVSD in Down Syndrome.
•• Alharbi KM, Al-Mazroea AH, Abdallah AM, Almohammadi Y, Carlus SJ, Basit S. Targeted next-generation sequencing of 406 genes identified genetic defects underlying congenital heart disease in Down syndrome patients. Pediatr Cardiol. 2018;39:1676–80. This study revealed the role of mutations in different cardiac specific genes such as GATA3, KCNH2, ENG, FLNA and GUSB as an underlying risk factor for CHD in DS cases.
Pierpont ME, Brueckner M, Chung WK, Garg V, Lacro RV, McGuire AL, Mital S, Priest JR, Pu WT, Roberts A. Genetic basis for congenital heart disease: revisited: a scientific statement from the American Heart Association. Circulation. 2018;138:e653–711.
Peterson JK, Kochilas LK, Catton KG, Moller JH, Setty SP. Long-term outcomes of children with trisomy 13 and 18 after congenital heart disease interventions. Ann Thorac Surg. 2017;103:1941–9.
Redon R, Ishikawa S, Fitch KR, Feuk L, Perry GH, Andrews TD, Fiegler H, Shapero MH, Carson AR, Chen W. Global variation in copy number in the human genome. nature 2006;444:444–454.
Warejko JK, Schueler M, Vivante A, et al. Whole exome sequencing reveals a monogenic cause of disease in ≈43% of 35 families with midaortic syndrome. Hypertension. 2018;71:691–9.
Grossfeld PD, Mattina T, Lai Z, Favier R, Jones KL, Cotter F, Jones C, Consortium 11q. The 11q terminal deletion disorder: a prospective study of 110 cases. Am J Med Genet A. 2004;129:51–61.
Favier R, Akshoomoff N, Mattson S, Grossfeld P. Jacobsen syndrome: advances in our knowledge of phenotype and genotype. In: Am J Med Genet C Semin Med. Wiley Online Library. 2015:239–250.
Battaglia 1p36 deletion syndrome – retired chapter, for historical reference only, in GeneReviews(®), M.P. Adam, et al., (Eds). University of Washington, Seattle. 1993.
Battaglia A. Del 1p36 syndrome: a newly emerging clinical entity. Brain Develop. 2005;27:358–61.
Hills C, Moller JH, Finkelstein M, Lohr J, Schimmenti L. Cri du chat syndrome and congenital heart disease: a review of previously reported cases and presentation of an additional 21 cases from the Pediatric Cardiac Care Consortium. Pediatrics. 2006;117:e924–7.
Peng Y, Pang J, Hu J, Jia Z, Xi H, Ma N, Yang S, Liu J, Huang X, Tang C. Clinical and molecular characterization of 12 prenatal cases of Cri-du-chat syndrome. Mol Genet Genomic Med. 2020;8: e1312.
Yi Li Q, Newbury-Ecob RA, Terrett JA, Wilson DI, Curtis AR, Ho Yi C, Gebuhr T, Bullen PJ, Robson SC, Strachan T. Holt-Oram syndrome is caused by mutations in TBX5, a member of the Brachyury (T) gene family. Nat Genet. 1997;15:21–9.
Li L, Krantz ID, Deng YU, Genin A, Banta AB, Collins CC, Qi M, Trask BJ, Kuo WL, Cochran J. Alagille syndrome is caused by mutations in human Jagged1, which encodes a ligand for Notch1. Nat Genet. 1997;16:243–51.
Emerick KM, Rand EB, Goldmuntz E, Krantz ID, Spinner NB, Piccoli DA. Features of Alagille syndrome in 92 patients: frequency and relation to prognosis. Hepatology. 1999;29:822–9.
Roberts AE, Allanson JE, Tartaglia M, Gelb BD. Noonan syndrome. The Lancet. 2013;381:333–42.
Gelb BD, Roberts AE, Tartaglia M. Cardiomyopathies in Noonan syndrome and the other RASopathies. Prog Pediatr Cardiol. 2015;39:13–9.
Srivastava D. Genetic regulation of cardiogenesis and congenital heart disease. ANNUAL REVIEW OF PATHOLOG. 2006;1:199.
Vissers LE, van Ravenswaaij C, Admiraal R, Hurst JA, de Vries B, Janssen IM, van der Vliet WA, Huys EH, de Jong PJ, Hamel BC. Mutations in a new member of the chromodomain gene family cause CHARGE syndrome. Nat Genet. 2004;36:955–7.
Weismann CG, Gelb BD. The genetics of congenital heart disease: a review of recent developments. Curr Opin Cardiol. 2007;22:200–6.
Corona-Rivera JR, Nieto-García R, Gutiérrez-Chávez AS, Bobadilla-Morales L, Rios-Flores IM, Corona-Rivera A, Fabián-Morales GE, Zavala-Cortés I, Lugo-Iglesias C, Peña-Padilla C. Maternal risk factors for congenital heart defects in infants with Down syndrome from Western Mexico. Am J Med Genet A. 2019;179:1857–65.
Khoury MJ, Erickson JD. Can maternal risk factors influence the presence of major birth defects in infants with Down syndrome? Am J Med Genet. 1992;43:1016–22.
Torfs CP, Christianson RE. Maternal risk factors and major associated defects in infants with Down syndrome. Epidemiology. 1999:264–270.
Fixler DE, Threlkeld N. Prenatal exposures and congenital heart defects in Down syndrome infants. Teratology. 1998;58:6–12.
H. Bean LJ, Allen EG, Tinker SW, Hollis ND, Locke AE, Druschel C, Hobbs CA, O’Leary L, Romitti PA, Royle MH. Lack of maternal folic acid supplementation is associated with heart defects in Down syndrome: A report from the National Down Syndrome Project Birth Defects Res A. 2011;91:885-893.
Dolk H, McCullough N, Callaghan S, Casey F, Craig B, Given J, Loane M, Lagan BM, Bunting B, Boyle B. Risk factors for congenital heart disease: the Baby Hearts Study, a population-based case-control study. PLoS ONE. 2020;15: e0227908.
Brandalize APC, Bandinelli E, dos Santos PA, Roisenberg I, Schüler-Faccini L. Evaluation of C677T and A1298C polymorphisms of the MTHFR gene as maternal risk factors for Down syndrome and congenital heart defects. Am J Med Genet A. 2009;149:2080–7.
Nakajima Y. Second lineage of heart forming region provides new understanding of conotruncal heart defects. Congenit Anom. 2010;50:8–14.
Roberts C, Ivins SM, James CT, Scambler PJ. Retinoic acid down-regulates Tbx1 expression in vivo and in vitro. Developmental dynamics: an official publication of the American Association of Anatomists. 2005;232:928–38.
Guris DL, Duester G, Papaioannou VE, Imamoto A. Dose-dependent interaction of Tbx1 and Crkl and locally aberrant RA signaling in a model of del22q11 syndrome. Dev Cell. 2006;10:81–92.
Sailani MR. Genetic variability and epigenetic alterations in Down syndrome with congenital heart defects. PhD Thesis, éditeur non identifié. 2013.
• Mouat JS, Li S, Myint SS, Laufer BI, Lupo PJ, Schraw JM, Woodhouse JP, de Smith AJ, LaSalle JM. Epigenomic signature of major congenital heart defects in newborns with Down syndrome. medRxiv. 2023;2023–05. This study finds a sex-specific signature DNA methylatin in DS-CHD individuals compared to DS non-CHD.
Rachamadugu SI, Miller KA, Lee IH, Zou YS. Genetic detection of congenital heart disease. Gynecology and Obstetrics Clinical Medicine. 2022.
Matsumoto N, Niikawa N. Kabuki make-up syndrome: a review. In: American Journal of Medical Genetics Part C: Seminars in Medical Genetics. Wiley Online Library. 2003:57–65.
Zaidi S, Brueckner M. Genetics and genomics of congenital heart disease. Circ Res. 2017;120:923–40.
Wyllie JP, Wright MJ, Burn J, Hunter S. Natural history of trisomy 13. Arch Dis Child. 1994;71:343–5.
Nguyen JM, Qualmann KJ, Okashah R, Reilly A, Alexeyev MF, Campbell DJ. 5p deletions: current knowledge and future directions. In: Am J Med Genet Part C: Seminars in Medical Genetics. Wiley Online Library. 2015:224–238.
Peyvandi F, Kunicki T, Lillicrap D. Genetic sequence analysis of inherited bleeding diseases. Blood, The Journal of the American Society of Hematology. 2013;122:3423–31.
Koolen DA, Sharp AJ, Hurst JA, Firth HV, Knight SJ, Goldenberg A, Saugier-Veber P, Pfundt R, Vissers LE, Destrée A. Clinical and molecular delineation of the 17q21. 31 microdeletion syndrome. J Med Genet. 2008;45:710–20.
Phelan K, McDermid H. The 22q13. 3 deletion syndrome (Phelan-McDermid syndrome). Molecular syndromology. 2011;2:186–201.
Hassed S, Li S, Mulvihill J, Aston C, Palmer S. Adams-Oliver syndrome review of the literature: refining the diagnostic phenotype. Am J Med Genet A. 2017;173:790–800.
Alankarage D, Szot JO, Pachter N, Slavotinek A, Selleri L, Shieh JT, Winlaw D, Giannoulatou E, Chapman G, Dunwoodie SL. Functional characterization of a novel PBX1 de novo missense variant identified in a patient with syndromic congenital heart disease. Hum Mol Genet. 2020;29:1068–82.
Trider C-L, Arra-Robar A, van Ravenswaaij-Arts C, Blake K. Developing a CHARGE syndrome checklist: health supervision across the lifespan (from head to toe). Am J Med Genet A. 2017;173:684–91.
O’Connor MJ, Tang X, Collins RT. Cardiac diagnoses, procedures, and healthcare utilisation in inpatients with Ellis–van Creveld syndrome. Cardiol Young. 2015;25:95–101.
Lin AE, Krikov S, Riehle-Colarusso T, Frías JL, Belmont J, Anderka M, Geva T, Getz KD, Botto LD, Study NBDP. Laterality defects in the national birth defects prevention study (1998–2007): birth prevalence and descriptive epidemiology. Am J Med Genet A. 2014;164:2581–91.
Jin SC, Homsy J, Zaidi S, Lu Q, Morton S, DePalma SR, Zeng X, Qi H, Chang W, Sierant MC. Contribution of rare inherited and de novo variants in 2,871 congenital heart disease probands. Nat Genet. 2017;49:1593–601.
McDermott DA, Fong JC, Basson CT. Holt-Oram syndrome. 2019.
Hannibal MC, Buckingham KJ, Ng SB, Ming JE, Beck AE, McMillin MJ, Gildersleeve HI, Bigham AW, Tabor HK, Mefford HC. Spectrum of MLL2 (ALR) mutations in 110 cases of Kabuki syndrome. Am J Med Genet A. 2011;155:1511–6.
MacCarrick G, Black JH, Bowdin S, El-Hamamsy I, Frischmeyer-Guerrerio PA, Guerrerio AL, Sponseller PD, Loeys B, Dietz HC. Loeys-Dietz syndrome: a primer for diagnosis and management. Genet Med. 2014;16:576–87.
Loughborough WW, Minhas KS, Rodrigues JC, Lyen SM, Burt HE, Manghat NE, Brooks MJ, Stuart G, Hamilton MC. Cardiovascular manifestations and complications of Loeys-Dietz syndrome: CT and MR imaging findings. Radiographics. 2018;38:275–86.
Evans C-A, Pinner J, Chan CY, Bowyer L, Mowat D, Buckley MF, Roscioli T. Fetal diagnosis of Mowat-Wilson syndrome by whole exome sequencing. Am J Med Genet A. 2019;179:2152–7.
Mowat DR, Wilson MJ, Goossens M. Mowat-Wilson syndrome. J Med Genet. 2003;40:305–10.
Lin AE, Michot C, Cormier-Daire V, L’Ecuyer TJ, Matherne GP, Barnes BH, Humberson JB, Edmondson AC, Zackai E, O’Connor MJ. Gain-of-function mutations in SMAD4 cause a distinctive repertoire of cardiovascular phenotypes in patients with Myhre syndrome. Am J Med Genet A. 2016;170:2617–31.
Jhang WK, Choi J-H, Lee BH, Kim G-H, Yoo H-W. Cardiac manifestations and associations with gene mutations in patients diagnosed with RASopathies. Pediatr Cardiol. 2016;37:1539–47.
Meroni G. X-linked Opitz G/BBB syndrome synonyms: Opitz syndrome, X-linked; XLOS. 2018.
Konya MN, Elmas M, Erginoğlu SE, Yeşil M. A rare case of 3C disease: Ritscher-Schinzel syndrome presenting with recurrent talipes equinovarus. Int J Surg Case Rep. 2015;7:130–3.
Leonardi ML, Pai GS, Wilkes B, Lebel RR. Ritscher-Schinzel cranio-cerebello-cardiac (3C) syndrome: report of four new cases and review. Am J Med Genet. 2001;102:237–42.
Spena S, Gervasini C, Milani D. Ultra-rare syndromes: the example of Rubinstein-Taybi syndrome. Journal of pediatric genetics. 2015;4:177–86.
Jira PE, Waterham HR, Wanders RJA, Smeitink JAM, Sengers RCA, Wevers RA. Smith-Lemli-Opitz syndrome and the DHCR7 gene. Ann Hum Genet. 2003;67:269–80.
Leventopoulos G, Kitsiou-Tzeli S, Kritikos K, Psoni S, Mavrou A, Kanavakis E, Fryssira H. A clinical study of Sotos syndrome patients with review of the literature. Pediatr Neurol. 2009;40:357–64.
Acknowledgements
We are extremely thankful to Prof. Rajiva Raman, Department of Zoology, BHU, Varanasi, India for critically reviewing the manuscript, grammatical corrections and for his valuable suggestions.
Author information
Authors and Affiliations
Contributions
JM has performed the literature search and prepared the original draft including all figures and tables. BM sketched the outline for the article, critically supervised and revised the manuscript at every single step. Both authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Maddhesiya, J., Mohapatra, B. Understanding the Genetic and Non-Genetic Interconnections in the Aetiology of Syndromic Congenital Heart Disease: An Updated Review: Part 2. Curr Cardiol Rep 26, 167–178 (2024). https://doi.org/10.1007/s11886-024-02020-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11886-024-02020-x