Abstract
Purpose of Review
Hypertension is a leading risk factor for all-cause mortality in adults; however, medication non-adherence and intolerance present an enormous treatment challenge. Given the critical role of renal sympathetic nerves in neurogenic control of blood pressure and pathophysiology of hypertension, renal sympathetic denervation (RDN) has been explored as a therapeutic strategy in hypertension treatment over the last 15 years. In this review, we will discuss the role of renal sympathetic nerves in the pathophysiology of hypertension, provide an update on the available evidence regarding the short- and long-term safety and effectiveness of RDN in the treatment of hypertension, and consider its future perspectives.
Recent Findings
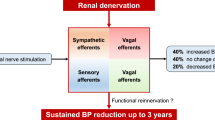
RDN is a percutaneous endovascular catheter–based neuromodulation approach that enables ablation of renal sympathetic nerve fibers within the adventitial layer of the renal arteries using radiofrequency (most extensively studied), ultrasound energy, or neurolytics (e.g., alcohol). In the last decade, advancements in procedural techniques and well-designed sham-controlled trials utilizing 24-h ambulatory blood pressure measurements have demonstrated that RDN has an excellent safety profile and results in a modest reduction of blood pressure, in a wide range of hypertensive phenotypes (mild to resistant), irrespective of antihypertensive drug use and this effect is sustained over a 3-year period. Superiority of a particular RDN modality has not been yet established.
Summary
Despite strong evidence demonstrating efficacy and safety of RDN, current data does not support its use as a primary approach in the treatment of hypertension due to its modest treatment effect and concerns around long-term sustainability. Perhaps the best utility of RDN is in hypertensives intolerant to antihypertensive medications or as an adjunct to aldosterone antagonists in the management of resistant hypertension. Patient selection will be critical to demonstrate a meaningful benefit of RDN. Future well-designed studies are necessary to determine predictors and measures of response to RDN, long-term efficacy given question of renal nerve regeneration, comparison of available technologies, safety in patients with advanced kidney disease, and improvement in patient quality of life measures.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Collaborators GBDRF. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392(10159):1923–1994.
Mills KT, et al. Global disparities of hypertension prevalence and control: a systematic analysis of population-based studies from 90 countries. Circulation. 2016;134(6):441–50.
Group SR, et al. A randomized trial of intensive versus standard blood-pressure control. N Engl J Med. 2015;373(22):2103–2116.
Whelton PK, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension. 2018;71(6):e13–115.
Smith SM, et al. Incidence, prevalence, and predictors of treatment-resistant hypertension with intensive blood pressure lowering. J Clin Hypertens (Greenwich). 2019;21(6):825–34.
Kirkland EB, et al. Trends in healthcare expenditures among US adults with hypertension: national estimates, 2003–2014. J Am Heart Assoc. 2018;7(11).
Durand H, et al. Medication adherence among patients with apparent treatment-resistant hypertension: systematic review and meta-analysis. J Hypertens. 2017;35(12):2346–57.
Ritchey M, et al. Vital signs: disparities in antihypertensive medication nonadherence among medicare part D beneficiaries - United States, 2014. MMWR Morb Mortal Wkly Rep. 2016;65(36):967–76.
DiBona GF. Neural control of the kidney: functionally specific renal sympathetic nerve fibers. Am J Physiol Regul Integr Comp Physiol. 2000;279(5):R1517–24.
Sakakura K, et al. Anatomic assessment of sympathetic peri-arterial renal nerves in man. J Am Coll Cardiol. 2014;64(7):635–43.
Schlaich MP, et al. Renal sympathetic-nerve ablation for uncontrolled hypertension. N Engl J Med. 2009;361(9):932–4.
Campese VM, Kogosov E. Renal afferent denervation prevents hypertension in rats with chronic renal failure. Hypertension. 1995;25(4 Pt 2):878–82.
Converse RL Jr, et al. Sympathetic overactivity in patients with chronic renal failure. N Engl J Med. 1992;327(27):1912–8.
Barrett CJ. Renal sympathetic nerves - what have they got to do with cardiovascular disease? Exp Physiol. 2015;100(4):359–65.
Myat A, et al. Renal sympathetic denervation therapy for resistant hypertension: a contemporary synopsis and future implications. Circ Cardiovasc Interv. 2013;6(2):184–97.
Smithwick RH, Thompson JE. Splanchnicectomy for essential hypertension; results in 1,266 cases. J Am Med Assoc. 1953;152(16):1501–4.
Krum H, et al. Percutaneous renal denervation in patients with treatment-resistant hypertension: final 3-year report of the Symplicity HTN-1 study. Lancet. 2014;383(9917):622–9.
Symplicity HTNI, et al. Renal sympathetic denervation in patients with treatment-resistant hypertension (the Symplicity HTN-2 trial): a randomised controlled trial. Lancet. 2010;376(9756):1903–9.
Bakris GL, et al. 12-month blood pressure results of catheter-based renal artery denervation for resistant hypertension: the SYMPLICITY HTN-3 trial. J Am Coll Cardiol. 2015;65(13):1314–21.
Azizi M, et al. Optimum and stepped care standardised antihypertensive treatment with or without renal denervation for resistant hypertension (DENERHTN): a multicentre, open-label, randomised controlled trial. Lancet. 2015;385(9981):1957–65.
•• Mahfoud F, et al. Effects of renal denervation on kidney function and long-term outcomes: 3-year follow-up from the Global SYMPLICITY Registry. Eur Heart J. 2019;40(42):3474–3482. This is a prospective, open-label registry of 1742 high cardiovascular risk patients (treated with Symplicity Flex or Symplicity Spyral RF-RDN) that provides 3-year follow-up data on RDN, the largest and longest so far. This study demonstrates BP-lowering efficacy and procedural safety of RDN over the 3-year period.
Krum H, et al. Catheter-based renal sympathetic denervation for resistant hypertension: a multicentre safety and proof-of-principle cohort study. Lancet. 2009;373(9671):1275–81.
Kandzari DE, et al. Predictors of blood pressure response in the SYMPLICITY HTN-3 trial. Eur Heart J. 2015;36(4):219–27.
Kandzari DE, et al. Effect of renal denervation on blood pressure in the presence of antihypertensive drugs: 6-month efficacy and safety results from the SPYRAL HTN-ON MED proof-of-concept randomised trial. Lancet. 2018;391(10137):2346–55.
Bohm M, et al. Efficacy of catheter-based renal denervation in the absence of antihypertensive medications (SPYRAL HTN-OFF MED Pivotal): a multicentre, randomised, sham-controlled trial. Lancet. 2020;395(10234):1444–51.
Townsend RR, et al. Catheter-based renal denervation in patients with uncontrolled hypertension in the absence of antihypertensive medications (SPYRAL HTN-OFF MED): a randomised, sham-controlled, proof-of-concept trial. Lancet. 2017;390(10108):2160–70.
Weber MA, et al. The REDUCE HTN: REINFORCE: randomized, sham-controlled trial of bipolar radiofrequency renal denervation for the treatment of hypertension. JACC Cardiovasc Interv. 2020;13(4):461–70.
Azizi M, et al. Endovascular ultrasound renal denervation to treat hypertension (RADIANCE-HTN SOLO): a multicentre, international, single-blind, randomised, sham-controlled trial. Lancet. 2018;391(10137):2335–45.
Azizi M, et al. Six-month results of treatment-blinded medication titration for hypertension control following randomization to endovascular ultrasound renal denervation or a sham procedure in the RADIANCE-HTN SOLO trial. Circulation. 2019.
Azizi M, et al. 12-month results from the unblinded phase of the RADIANCE-HTN SOLO trial of ultrasound renal denervation. JACC Cardiovasc Interv. 2020;13(24):2922–33.
Azizi M, et al. Ultrasound renal denervation for hypertension resistant to a triple medication pill (RADIANCE-HTN TRIO): a randomised, multicentre, single-blind, sham-controlled trial. Lancet. 2021;397(10293):2476–86.
Kario K, et al. Catheter-based ultrasound renal denervation in patients with resistant hypertension: the randomized, controlled REQUIRE trial. Hypertens Res. 2022;45(2):221–31.
Mahfoud F, et al. Alcohol-mediated renal denervation using the Peregrine system infusion catheter for treatment of hypertension. JACC Cardiovasc Interv. 2020;13(4):471–84.
• Mahfoud F, et al. Long-term results up to 12 months after catheter-based alcohol-mediated renal denervation for treatment of resistant hypertension. Circ Cardiovasc Interv. 2021;14(9):e010075. This 3-arm randomized, single-blind trial in patients with resistant hypertension compares the safety and efficacy of (a) RF-RDN of the main renal artery, (b) RF-RDN of the main and distal arterial branches, or (c) US-RDN of the main renal artery. All 3 modalities lowered BP effectively; however, US-RDN performed better than RF-RDN in this study. The small sample size limits the reliability of the results.
Fengler K, et al. A three-arm randomized trial of different renal denervation devices and techniques in patients with resistant hypertension (RADIOSOUND-HTN). Circulation. 2019;139(5):590–600.
Pekarskiy SE, et al. Denervation of the distal renal arterial branches vs. conventional main renal artery treatment: a randomized controlled trial for treatment of resistant hypertension. J Hypertens. 2017;35(2):369–375.
Gosse P, et al. Twenty-four-hour blood pressure monitoring to predict and assess impact of renal denervation: the DENERHTN study (renal denervation for hypertension). Hypertension. 2017;69(3):494–500.
Ahmad Y, et al. Renal denervation for hypertension: a systematic review and meta-analysis of randomized, blinded, placebo-controlled trials. JACC Cardiovasc Interv. 2021;14(23):2614–24.
Mahfoud F, et al. Long-term efficacy and safety of renal denervation in the presence of antihypertensive drugs (SPYRAL HTN-ON MED): a randomised, sham-controlled trial. Lancet. 2022;399(10333):1401–10.
Ott C, et al. Renal denervation in patients with versus without chronic kidney disease: results from the Global SYMPLICITY Registry with follow-up data of 3 years. Nephrol Dial Transplant. 2022;37(2):304–10.
Booth LC, et al. Reinnervation of renal afferent and efferent nerves at 5.5 and 11 months after catheter-based radiofrequency renal denervation in sheep. Hypertension. 2015;65(2):393–400.
Chen P, et al. The influence of inhibiting renal neural regeneration on the efficacy of renal denervation to chronic heart failure. ESC Heart Fail. 2021;8(6):4760–71.
Rodionova K, et al. Complex reinnervation pattern after unilateral renal denervation in rats. Am J Physiol Regul Integr Comp Physiol. 2016;310(9):R806–18.
Sanders MF, et al. Renal safety of catheter-based renal denervation: systematic review and meta-analysis. Nephrol Dial Transplant. 2017;32(9):1440–7.
Mahfoud F, et al. Proceedings from the 3rd European Clinical Consensus Conference for clinical trials in device-based hypertension therapies. Eur Heart J. 2020;41(16):1588–99.
Townsend RR, et al. Review and meta-analysis of renal artery damage following percutaneous renal denervation with radiofrequency renal artery ablation. EuroIntervention. 2020;16(1):89–96.
Schmieder RE. Renal denervation: where do we stand and what is the relevance to the nephrologist? Nephrol Dial Transplant. 2020.
Xia M, et al. Efficacy and safety of renal denervation for hypertension in patients with chronic kidney disease: a meta-analysis. Int J Hyperthermia. 2021;38(1):732–42.
Marin F, et al. Insights on safety and efficacy of renal artery denervation for uncontrolled-resistant hypertension in a high risk population with chronic kidney disease: first Italian real-world experience. J Nephrol. 2021;34(5):1445–55.
Singh RR, et al. Catheter-based renal denervation exacerbates blood pressure fall during hemorrhage. J Am Coll Cardiol. 2017;69(8):951–64.
Chowdhury EK, et al. Cost-effectiveness of renal denervation therapy for treatment-resistant hypertension: a best case scenario. Am J Hypertens. 2018;31(10):1156–63.
Mahfoud F, et al. Changes in plasma renin activity after renal artery sympathetic denervation. J Am Coll Cardiol. 2021;77(23):2909–19.
Vaclavik J, et al. Addition of spironolactone in patients with resistant arterial hypertension (ASPIRANT): a randomized, double-blind, placebo-controlled trial. Hypertension. 2011;57(6):1069–75.
Williams B, et al. Spironolactone versus placebo, bisoprolol, and doxazosin to determine the optimal treatment for drug-resistant hypertension (PATHWAY-2): a randomised, double-blind, crossover trial. Lancet. 2015;386(10008):2059–68.
Oliveras A, et al. Spironolactone versus sympathetic renal denervation to treat true resistant hypertension: results from the DENERVHTA study - a randomized controlled trial. J Hypertens. 2016;34(9):1863–71.
Peixoto AJ. Is there a role for device therapies in resistant hypertension?: The CON side. Kidney360. 2020;1(1):9–13.
Sharp TE 3rd, et al. Renal denervation prevents heart failure progression via inhibition of the renin-angiotensin system. J Am Coll Cardiol. 2018;72(21):2609–21.
Fukuta H, et al. Effects of catheter-based renal denervation on heart failure with reduced ejection fraction: a meta-analysis of randomized controlled trials. Heart Fail Rev. 2022;27(1):29–36.
Younis A, Steinberg JS. Renal denervation for patients with atrial fibrillation. Curr Cardiol Rep. 2021;23(9):126.
Steinberg JS, et al. Effect of renal denervation and catheter ablation vs catheter ablation alone on atrial fibrillation recurrence among patients with paroxysmal atrial fibrillation and hypertension: the ERADICATE-AF randomized clinical trial. JAMA. 2020;323(3):248–55.
Nawar K, et al. Renal denervation for atrial fibrillation: a comprehensive updated systematic review and meta-analysis. J Hum Hypertens. 2022.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Hypertension
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rao, A., Krishnan, N. Update on Renal Sympathetic Denervation for the Treatment of Hypertension. Curr Cardiol Rep 24, 1261–1271 (2022). https://doi.org/10.1007/s11886-022-01753-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11886-022-01753-x