Abstract
Purposes of Review
Th9 cells are recognized as a novel subset of effector T helper cells that preferentially produce IL-9. Here, we provide a current update on the reports related to the function of Th9 cells in allergic inflammatory diseases.
Recent Findings
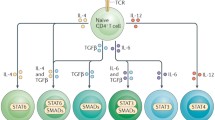
The effector Th9 cells differentiating from naïve T helper cells have recently been identified. Because of accumulating findings of Th9 cells in many inflammatory diseases, including allergic diseases, diverse functions of Th9 cells in regulating immune responses have been suggested. Related reports indicate multiple sources of IL-9 besides Th9 cells and their association with the pathogenesis of allergic rhinitis, asthma, atopic dermatitis, contact dermatitis, and food allergy. More recently, elements of the epigenetic landscape involving in the regulation of IL-9 by Th9 cells have been identified to be the potential target for allergic inflammation.
Summary
This review provides the most recent information about Th9 cells and their contribution in airway allergic disease, skin, and food allergy.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Veldhoen M, Uyttenhove C, van Snick J, Helmby H, Westendorf A, Buer J, et al. Transforming growth factor-beta 'reprograms' the differentiation of T helper 2 cells and promotes an interleukin 9-producing subset. Nat Immunol. 2008;9(12):1341–6. https://doi.org/10.1038/ni.1659.
Dardalhon V, Awasthi A, Kwon H, Galileos G, Gao W, Sobel RA, et al. IL-4 inhibits TGF-beta-induced Foxp3(+) T cells and, together with TGF-beta, generates IL-9(+) IL-10(+) Foxp3(−) effector T cells. Nat Immunol. 2008;9(12):1347–55. https://doi.org/10.1038/ni.1677.
Jabeen R, Goswami R, Awe O, Kulkarni A, Nguyen ET, Attenasio A, et al. Th9 cell development requires a BATF-regulated transcriptional network. J Clin Invest. 2013;123(11):4641–53. https://doi.org/10.1172/JCI69489.
•• Micosse C, von Meyenn L, Steck O, Kipfer E, Adam C, Simillion C, et al. Human "TH9" cells are a subpopulation of PPAR-gamma(+) TH2 cells. Sci Immunol. 2019;4(31). https://doi.org/10.1126/sciimmunol.aat5943 COMMENT: Study showing that Th9 subset transiently induced during skin inflammation is a subpopulation of Th2 lineage.
Clark RA, Schlapbach C. TH9 cells in skin disorders. Semin Immunopathol. 2017;39(1):47–54. https://doi.org/10.1007/s00281-016-0607-8.
Licona-Limon P, Henao-Mejia J, Temann AU, Gagliani N, Licona-Limon I, Ishigame H, et al. Th9 cells drive host immunity against gastrointestinal worm infection. Immunity. 2013;39(4):744–57. https://doi.org/10.1016/j.immuni.2013.07.020.
Angkasekwinai P, Srimanote P, Wang YH, Pootong A, Sakolvaree Y, Pattanapanyasat K, et al. Interleukin-25 (IL-25) promotes efficient protective immunity against Trichinella spiralis infection by enhancing the antigen-specific IL-9 response. Infect Immun. 2013;81(10):3731–41. https://doi.org/10.1128/IAI.00646-13.
Jones CP, Gregory LG, Causton B, Campbell GA, Lloyd CM. Activin a and TGF-beta promote T(H)9 cell-mediated pulmonary allergic pathology. J Allergy Clin Immunol. 2012;129(4):1000–10 e3. https://doi.org/10.1016/j.jaci.2011.12.965.
Angkasekwinai P, Chang SH, Thapa M, Watarai H, Dong C. Regulation of IL-9 expression by IL-25 signaling. Nat Immunol. 2010;11(3):250–6. https://doi.org/10.1038/ni.1846.
Chang HC, Sehra S, Goswami R, Yao W, Yu Q, Stritesky GL, et al. The transcription factor PU.1 is required for the development of IL-9-producing T cells and allergic inflammation. Nat Immunol. 2010;11(6):527–34. https://doi.org/10.1038/ni.1867.
Vargas TR, Humblin E, Vegran F, Ghiringhelli F, Apetoh L. T(H)9 cells in anti-tumor immunity. Semin Immunopathol. 2017;39(1):39–46. https://doi.org/10.1007/s00281-016-0599-4.
Gerlach K, McKenzie AN, Neurath MF, Weigmann B. IL-9 regulates intestinal barrier function in experimental T cell-mediated colitis. Tissue Barriers. 2015;3(1–2):e983777. https://doi.org/10.4161/21688370.2014.983777.
Romagnani S. Immunologic influences on allergy and theT(H)1/T(H)2 balance. J Allergy Clin Immunol. 2004;113(3):395–400. https://doi.org/10.1016/j.jaci.2003.11.025.
Moro K, Yamada T, Tanabe M, Takeuchi T, Ikawa T, Kawamoto H, et al. Innate production of T(H)2 cytokines by adipose tissue-associated c-kit(+)Sca-1(+) lymphoid cells. Nature. 2010;463(7280):540–4. https://doi.org/10.1038/nature08636.
Price AE, Liang HE, Sullivan BM, Reinhardt RL, Eisley CJ, Erle DJ, et al. Systemically dispersed innate IL-13-expressing cells in type 2 immunity. Proc Natl Acad Sci U S A. 2010;107(25):11489–94. https://doi.org/10.1073/pnas.1003988107.
Neill DR, Wong SH, Bellosi A, Flynn RJ, Daly M, Langford TK, et al. Nuocytes represent a new innate effector leukocyte that mediates type-2 immunity. Nature. 2010;464(7293):1367–70. https://doi.org/10.1038/nature08900.
Halim TY, Steer CA, Matha L, Gold MJ, Martinez-Gonzalez I, McNagny KM, et al. Group 2 innate lymphoid cells are critical for the initiation of adaptive T helper 2 cell-mediated allergic lung inflammation. Immunity. 2014;40(3):425–35. https://doi.org/10.1016/j.immuni.2014.01.011.
Liu B, Lee JB, Chen CY, Hershey GK, Wang YH. Collaborative interactions between type 2 innate lymphoid cells and antigen-specific CD4+ Th2 cells exacerbate murine allergic airway diseases with prominent eosinophilia. J Immunol. 2015;194(8):3583–93. https://doi.org/10.4049/jimmunol.1400951.
Choy DF, Hart KM, Borthwick LA, Shikotra A, Nagarkar DR, Siddiqui S, et al. TH2 and TH17 inflammatory pathways are reciprocally regulated in asthma. Sci Transl Med. 2015;7(301):301ra129. https://doi.org/10.1126/scitranslmed.aab3142.
Sehra S, Yao W, Nguyen ET, Glosson-Byers NL, Akhtar N, Zhou B, et al. TH9 cells are required for tissue mast cell accumulation during allergic inflammation. J Allergy Clin Immunol. 2015;136(2):433–40 e1. https://doi.org/10.1016/j.jaci.2015.01.021.
Kim BS, Kim IK, Park YJ, Kim YS, Kim YJ, Chang WS, et al. Conversion of Th2 memory cells into Foxp3+ regulatory T cells suppressing Th2-mediated allergic asthma. Proc Natl Acad Sci U S A. 2010;107(19):8742–7. https://doi.org/10.1073/pnas.0911756107.
Murugaiyan G, Beynon V, Da Cunha AP, Joller N, Weiner HL. IFN-gamma limits Th9-mediated autoimmune inflammation through dendritic cell modulation of IL-27. J Immunol. 2012;189(11):5277–83. https://doi.org/10.4049/jimmunol.1200808.
Anuradha R, George PJ, Hanna LE, Chandrasekaran V, Kumaran P, Nutman TB, et al. IL-4-, TGF-beta-, and IL-1-dependent expansion of parasite antigen-specific Th9 cells is associated with clinical pathology in human lymphatic Filariasis. J Immunol. 2013;191(5):2466–73. https://doi.org/10.4049/jimmunol.1300911.
Liao W, Spolski R, Li P, Du N, West EE, Ren M, et al. Opposing actions of IL-2 and IL-21 on Th9 differentiation correlate with their differential regulation of BCL6 expression. Proc Natl Acad Sci U S A. 2014;111(9):3508–13. https://doi.org/10.1073/pnas.1301138111.
Yao WG, Zhang YL, Jabeen R, Nguyen ET, Wilkes DS, Tepper RS, et al. Interleukin-9 is required for allergic airway inflammation mediated by the cytokine TSLP. Immunity. 2013;38(2):360–72. https://doi.org/10.1016/j.immuni.2013.01.007.
Wong MT, Ye JJ, Alonso MN, Landrigan A, Cheung RK, Engleman E, et al. Regulation of human Th9 differentiation by type I interferons and IL-21. Immunol Cell Biol. 2010;88(6):624–31. https://doi.org/10.1038/icb.2010.53.
Xiao X, Balasubramanian S, Liu WT, Chu XF, Wang HB, Taparowsky EJ, et al. OX40 signaling favors the induction of T(H)9 cells and airway inflammation. Nat Immunol. 2012;13(10):981–90. https://doi.org/10.1038/ni.2390.
Kerzerho J, Maazi H, Speak AO, Szely N, Lombardi V, Khoo B, et al. Programmed cell death ligand 2 regulates TH9 differentiation and induction of chronic airway hyperreactivity. J Allergy Clin Immunol. 2013;131(4):1048–57, 57 e1–2. https://doi.org/10.1016/j.jaci.2012.09.027.
Kim IK, Kim BS, Koh CH, Seok JW, Park JS, Shin KS, et al. Glucocorticoid-induced tumor necrosis factor receptor-related protein co-stimulation facilitates tumor regression by inducing IL-9-producing helper T cells. Nat Med. 2015;21(9):1010–7. https://doi.org/10.1038/nm.3922.
Xiao X, Shi X, Fan Y, Zhang X, Wu M, Lan P, et al. GITR subverts Foxp3(+) Tregs to boost Th9 immunity through regulation of histone acetylation. Nat Commun. 2015;6:8266. https://doi.org/10.1038/ncomms9266.
Elyaman W, Bassil R, Bradshaw EM, Orent W, Lahoud Y, Zhu B, et al. Notch receptors and Smad3 signaling cooperate in the induction of Interleukin-9-producing T cells. Immunity. 2012;36(4):623–34. https://doi.org/10.1016/j.immuni.2012.01.020.
Richard AC, Tan CY, Hawley ET, Gomez-Rodriguez J, Goswami R, Yang XP, et al. The TNF-family ligand TL1A and its receptor DR3 promote T cell-mediated allergic immunopathology by enhancing differentiation and pathogenicity of IL-9-producing T cells. J Immunol. 2015;194(8):3567–82. https://doi.org/10.4049/jimmunol.1401220.
Goswami R, Jabeen R, Yagi R, Pham D, Zhu JF, Goenka S, et al. STAT6-dependent regulation of Th9 development. J Immunol. 2012;188(3):968–75. https://doi.org/10.4049/jimmunol.1102840.
Staudt V, Bothur E, Klein M, Lingnau K, Reuter S, Grebe N, et al. Interferon-regulatory factor 4 is essential for the developmental program of T helper 9 cells. Immunity. 2010;33(2):192–202. https://doi.org/10.1016/j.immuni.2010.07.014.
Zhu YX, Kang LY, Luo W, Li CC, Yang L, Yang YC. Multiple transcription factors are required for activation of human interleukin 9 gene in T cells. J Biol Chem. 1996;271(26):15815–22.
Jash A, Sahoo A, Kim GC, Chae CS, Hwang JS, Kim JE, et al. Nuclear factor of activated T cells 1 (NFAT1)-induced permissive chromatin modification facilitates nuclear factor-kappaB (NF-kappaB)-mediated interleukin-9 (IL-9) transactivation. J Biol Chem. 2012;287(19):15445–57. https://doi.org/10.1074/jbc.M112.340356.
Tamiya T, Ichiyama K, Kotani H, Fukaya T, Sekiya T, Shichita T, et al. Smad2/3 and IRF4 play a cooperative role in IL-9-producing T cell induction. J Immunol. 2013;191(5):2360–71. https://doi.org/10.4049/jimmunol.1301276.
Humblin E, Thibaudin M, Chalmin F, Derangere V, Limagne E, Richard C, et al. IRF8-dependent molecular complexes control the Th9 transcriptional program. Nat Commun. 2017;8(1):2085. https://doi.org/10.1038/s41467-017-01070-w.
Malik S, Sadhu S, Elesela S, Pandey RP, Chawla AS, Sharma D, et al. Transcription factor Foxo1 is essential for IL-9 induction in T helper cells. Nat Commun. 2017;8(1):815. https://doi.org/10.1038/s41467-017-00674-6.
•• Xiao X, Fan Y, Li J, Zhang X, Lou X, Dou Y, et al. Guidance of super-enhancers in regulation of IL-9 induction and airway inflammation. J Exp Med. 2018;215(2):559–74. https://doi.org/10.1084/jem.20170928 COMMENT: Study identifying the elements for the epigenetic landscape regulating IL-9 expression by Th9 cells that could be another target for allergic airway inflammation.
•• Koh B, Abdul Qayum A, Srivastava R, Fu Y, Ulrich BJ, Janga SC, et al. A conserved enhancer regulates Il9 expression in multiple lineages. Nat Commun. 2018;9(1):4803. https://doi.org/10.1038/s41467-018-07202-0 COMMENT: Study identifying a conserved IL-9 regulatory element, CNS-25 as enhancer for IL-9 expression in multiple cell types, including Th9 cells.
Chen CY, Lee JB, Liu B, Ohta S, Wang PY, Kartashov AV, et al. Induction of Interleukin-9-producing mucosal mast cells promotes susceptibility to IgE-mediated experimental food allergy. Immunity. 2015;43(4):788–802. https://doi.org/10.1016/j.immuni.2015.08.020.
Turner JE, Morrison PJ, Wilhelm C, Wilson M, Ahlfors H, Renauld JC, et al. IL-9-mediated survival of type 2 innate lymphoid cells promotes damage control in helminth-induced lung inflammation. J Exp Med. 2013;210(13):2951–65. https://doi.org/10.1084/jem.20130071.
Rauber S, Luber M, Weber S, Maul L, Soare A, Wohlfahrt T, et al. Resolution of inflammation by interleukin-9-producing type 2 innate lymphoid cells. Nat Med. 2017;23(8):938–44. https://doi.org/10.1038/nm.4373.
Visekruna A, Ritter J, Scholz T, Campos L, Guralnik A, Poncette L, et al. Tc9 cells, a new subset of CD8(+) T cells, support Th2-mediated airway inflammation. Eur J Immunol. 2013;43(3):606–18. https://doi.org/10.1002/eji.201242825.
Nowak EC, Weaver CT, Turner H, Begum-Haque S, Becher B, Schreiner B, et al. IL-9 as a mediator of Th17-driven inflammatory disease. J Exp Med. 2009;206(8):1653–60. https://doi.org/10.1084/jem.20090246.
Elyaman W, Bradshaw EM, Uyttenhove C, Dardalhon V, Awasthi A, Imitola J, et al. IL-9 induces differentiation of TH17 cells and enhances function of FoxP3+ natural regulatory T cells. Proc Natl Acad Sci U S A. 2009;106(31):12885–90. https://doi.org/10.1073/pnas.0812530106.
Doull IJ, Lawrence S, Watson M, Begishvili T, Beasley RW, Lampe F, et al. Allelic association of gene markers on chromosomes 5q and 11q with atopy and bronchial hyperresponsiveness. Am J Respir Crit Care Med. 1996;153(4 Pt 1):1280–4. https://doi.org/10.1164/ajrccm.153.4.8616554.
Ulbrecht M, Eisenhut T, Bonisch J, Kruse R, Wjst M, Heinrich J, et al. High serum IgE concentrations: association with HLA-DR and markers on chromosome 5q31 and chromosome 11q13. J Allergy Clin Immunol. 1997;99(6 Pt 1):828–36.
Dong Q, Louahed J, Vink A, Sullivan CD, Messler CJ, Zhou Y, et al. IL-9 induces chemokine expression in lung epithelial cells and baseline airway eosinophilia in transgenic mice. Eur J Immunol. 1999;29(7):2130–9. https://doi.org/10.1002/(SICI)1521-4141(199907)29:07<2130::AID-IMMU2130>3.0.CO;2-S.
Longphre M, Li D, Gallup M, Drori E, Ordonez CL, Redman T, et al. Allergen-induced IL-9 directly stimulates mucin transcription in respiratory epithelial cells. J Clin Invest. 1999;104(10):1375–82. https://doi.org/10.1172/JCI6097.
Temann UA, Ray P, Flavell RA. Pulmonary overexpression of IL-9 induces Th2 cytokine expression, leading to immune pathology. J Clin Invest. 2002;109(1):29–39. https://doi.org/10.1172/JCI13696.
Townsend JM, Fallon GP, Matthews JD, Smith P, Jolin EH, McKenzie NA. IL-9-deficient mice establish fundamental roles for IL-9 in pulmonary mastocytosis and goblet cell hyperplasia but not T cell development. Immunity. 2000;13(4):573–83.
Kung TT, Luo B, Crawley Y, Garlisi CG, Devito K, Minnicozzi M, et al. Effect of anti-mIL-9 antibody on the development of pulmonary inflammation and airway hyperresponsiveness in allergic mice. Am J Respir Cell Mol Biol. 2001;25(5):600–5. https://doi.org/10.1165/ajrcmb.25.5.4533.
Cheng G, Arima M, Honda K, Hirata H, Eda F, Yoshida N, et al. Anti-interleukin-9 antibody treatment inhibits airway inflammation and hyperreactivity in mouse asthma model. Am J Respir Crit Care Med. 2002;166(3):409–16. https://doi.org/10.1164/rccm.2105079.
Angkasekwinai P. Allergic inflammation and atopic disease: role of Th9 cells. Methods Mol Biol. 2017;1585:189–99. https://doi.org/10.1007/978-1-4939-6877-0_15.
Wilhelm C, Hirota K, Stieglitz B, Van Snick J, Tolaini M, Lahl K, et al. An IL-9 fate reporter demonstrates the induction of an innate IL-9 response in lung inflammation. Nat Immunol. 2011;12(11):1071–7. https://doi.org/10.1038/ni.2133.
•• Moretti S, Renga G, Oikonomou V, Galosi C, Pariano M, Iannitti RG, et al. A mast cell-ILC2-Th9 pathway promotes lung inflammation in cystic fibrosis. Nat Commun. 2017;8:14017. https://doi.org/10.1038/ncomms14017 COMMENT: Study indicating the collaborative network of Th9, ILC2 and mast cells in amplifying lung inflammation.
Shimbara A, Christodoulopoulos P, Soussi-Gounni A, Olivenstein R, Nakamura Y, Levitt RC, et al. IL-9 and its receptor in allergic and nonallergic lung disease: increased expression in asthma. J Allergy Clin Immunol. 2000;105(1 Pt 1):108–15.
Erpenbeck VJ, Hohlfeld JM, Volkmann B, Hagenberg A, Geldmacher H, Braun A, et al. Segmental allergen challenge in patients with atopic asthma leads to increased IL-9 expression in bronchoalveolar lavage fluid lymphocytes. J Allergy Clin Immunol. 2003;111(6):1319–27.
Nouri-Aria KT, Pilette C, Jacobson MR, Watanabe H, Durham SR. IL-9 and c-kit(+) mast cells in allergic rhinitis during seasonal allergen exposure: effect of immunotherapy. J Allergy Clin Immunol. 2005;116(1):73–9. https://doi.org/10.1016/j.jaci.2005.03.011.
Devos S, Cormont F, Vrtala S, Hooghe-Peters E, Pirson F, Van Snick J. Allergen-induced interleukin-9 production in vitro: correlation with atopy in human adults and comparison with interleukin-5 and interleukin-13. Clin Exp Allergy. 2006;36(2):174–82. https://doi.org/10.1111/j.1365-2222.2006.02422.x.
Umezu-Goto M, Kajiyama Y, Kobayashi N, Kaminuma O, Suko M, Mori A. IL-9 production by peripheral blood mononuclear cells of atopic asthmatics. Int Arch Allergy Immunol. 2007;143:76–9. https://doi.org/10.1159/000101410.
Yao WG, Tepper RS, Kaplan MH. Predisposition to the development of IL-9-secreting T cells in atopic infants. J Allergy Clin Immunol. 2011;128(6):1357–60. https://doi.org/10.1016/j.jaci.2011.06.019.
Iinuma T, Okamoto Y, Yamamoto H, Inamine-Sasaki A, Ohki Y, Sakurai T, et al. Interleukin-25 and mucosal T cells in noneosinophilic and eosinophilic chronic rhinosinusitis. Ann Allergy Asthma Immunol. 2015;114(4):289–98. https://doi.org/10.1016/j.anai.2015.01.013.
Hoppenot D, Malakauskas K, Lavinskiene S, Bajoriuniene I, Kalinauskaite V, Sakalauskas R. Peripheral blood Th9 cells and eosinophil apoptosis in asthma patients. Medicina-Lithuania. 2015;51(1):10–7. https://doi.org/10.1016/j.medici.2015.01.001.
Jia L, Wang Y, Li J, Li S, Zhang Y, Shen J, et al. Detection of IL-9 producing T cells in the PBMCs of allergic asthmatic patients. BMC Immunol. 2017;18(1):38. https://doi.org/10.1186/s12865-017-0220-1.
Bieber T. Atopic dermatitis. N Engl J Med. 2008;358(14):1483–94. https://doi.org/10.1056/NEJMra074081.
Lowe AJ, Leung DYM, Tang MLK, Su JC, Allen KJ. The skin as a target for prevention of the atopic march. Ann Allergy Asthma Immunol. 2018;120(2):145–51. https://doi.org/10.1016/j.anai.2017.11.023.
Beck LA, Thaci D, Hamilton JD, Graham NM, Bieber T, Rocklin R, et al. Dupilumab treatment in adults with moderate-to-severe atopic dermatitis. N Engl J Med. 2014;371(2):130–9. https://doi.org/10.1056/NEJMoa1314768.
•• Thaci D, Simpson EL, Beck LA, Bieber T, Blauvelt A, Papp K, et al. Efficacy and safety of dupilumab in adults with moderate-to-severe atopic dermatitis inadequately controlled by topical treatments: a randomised, placebo-controlled, dose-ranging phase 2b trial. Lancet. 2016;387(10013):40–52. https://doi.org/10.1016/S0140-6736(15)00388-8 COMMENT: Clinical trial study indicating the effect of targeting Th2 cytokine IL-4 and IL-13 in atopic dermatitis.
Werfel T, Allam JP, Biedermann T, Eyerich K, Gilles S, Guttman-Yassky E, et al. Cellular and molecular immunologic mechanisms in patients with atopic dermatitis. J Allergy Clin Immunol. 2016;138(2):336–49. https://doi.org/10.1016/j.jaci.2016.06.010.
Lin JY, Chen JS, Hsu CJ, Miaw SC, Liu CY, Lee SJ, et al. Epicutaneous sensitization with protein antigen induces Th9 cells. J Invest Dermatol. 2012;132(3 Pt 1):739–41. https://doi.org/10.1038/jid.2011.382.
Namkung JH, Lee JE, Kim E, Park GT, Yang HS, Jang HY, et al. An association between IL-9 and IL-9 receptor gene polymorphisms and atopic dermatitis in a Korean population. J Dermatol Sci. 2011;62(1):16–21. https://doi.org/10.1016/j.jdermsci.2011.01.007.
Sismanopoulos N, Delivanis DA, Alysandratos KD, Angelidou A, Vasiadi M, Therianou A, et al. IL-9 induces VEGF secretion from human mast cells and IL-9/IL-9 receptor genes are overexpressed in atopic dermatitis. PLoS One. 2012;7(3):e33271. https://doi.org/10.1371/journal.pone.0033271.
Ciprandi G, De Amici M, Giunta V, Marseglia A, Marseglia G. Serum Interleukin-9 levels are associated with clinical severity in children with atopic dermatitis. Pediatr Dermatol. 2013;30(2):222–5. https://doi.org/10.1111/j.1525-1470.2012.01766.x.
Hamza AM, Omar SS, Abo El-Wafa RA, Elatrash MJ. Expression levels of transcription factor PU.1 and interleukin-9 in atopic dermatitis and their relation to disease severity and eruption types. Int J Dermatol. 2017;56(5):534–9. https://doi.org/10.1111/ijd.13579.
Esaki H, Brunner PM, Renert-Yuval Y, Czarnowicki T, Huynh T, Tran G, et al. Early-onset pediatric atopic dermatitis is TH2 but also TH17 polarized in skin. J Allergy Clin Immunol. 2016;138(6):1639–51. https://doi.org/10.1016/j.jaci.2016.07.013.
Schlapbach C, Gehad A, Yang C, Watanabe R, Guenova E, Teague JE, et al. Human TH9 cells are skin-tropic and have autocrine and paracrine proinflammatory capacity. Sci Transl Med. 2014;6(219):219ra8. https://doi.org/10.1126/scitranslmed.3007828.
Ma L, Xue HB, Guan XH, Shu CM, Zhang JH, Yu J. Possible pathogenic role of T helper type 9 cells and interleukin (IL)-9 in atopic dermatitis. Clin Exp Immunol. 2014;175(1):25–31. https://doi.org/10.1111/cei.12198.
Coulter EM, Jenkinson C, Farrell J, Lavergne SN, Pease C, White A, et al. Measurement of CD4+ and CD8+ T-lymphocyte cytokine secretion and gene expression changes in p-phenylenediamine allergic patients and tolerant individuals. J Invest Dermatol. 2010;130(1):161–74. https://doi.org/10.1038/jid.2009.187.
Baeck M, Herman A, de Montjoye L, Hendrickx E, Cheou P, Cochez PM, et al. Increased expression of interleukin-9 in patients with allergic contact dermatitis caused by p-phenylenediamine. Contact Dermatitis. 2018;79(6):346–55. https://doi.org/10.1111/cod.13123.
Liu J, Harberts E, Tammaro A, Girardi N, Filler RB, Fishelevich R, et al. IL-9 regulates allergen-specific Th1 responses in allergic contact dermatitis. J Invest Dermatol. 2014;134(7):1903–11. https://doi.org/10.1038/jid.2014.61.
Eller K, Wolf D, Huber JM, Metz M, Mayer G, McKenzie AN, et al. IL-9 production by regulatory T cells recruits mast cells that are essential for regulatory T cell-induced immune suppression. J Immunol. 2011;186(1):83–91. https://doi.org/10.4049/jimmunol.1001183.
Valenta R, Hochwallner H, Linhart B, Pahr S. Food allergies: the basics. Gastroenterology. 2015;148(6):1120–31 e4. https://doi.org/10.1053/j.gastro.2015.02.006.
Forbes EE, Groschwitz K, Abonia JP, Brandt EB, Cohen E, Blanchard C, et al. IL-9- and mast cell-mediated intestinal permeability predisposes to oral antigen hypersensitivity. J Exp Med. 2008;205(4):897–913. https://doi.org/10.1084/jem.20071046.
Osterfeld H, Ahrens R, Strait R, Finkelman FD, Renauld JC, Hogan SP. Differential roles for the IL-9/IL-9 receptor alpha-chain pathway in systemic and oral antigen-induced anaphylaxis. J Allergy Clin Immunol. 2010;125(2):469–76 e2. https://doi.org/10.1016/j.jaci.2009.09.054.
He SH, Liu ZQ, Chen X, Song CH, Zhou LF, Ma WJ, et al. IL-9(+) IL-10(+) T cells link immediate allergic response to late phase reaction. Clin Exp Immunol. 2011;165(1):29–37. https://doi.org/10.1111/j.1365-2249.2011.04394.x.
Xie J, Lotoski LC, Chooniedass R, Su RC, Simons FE, Liem J, et al. Elevated antigen-driven IL-9 responses are prominent in peanut allergic humans. PLoS One. 2012;7(10):e45377. https://doi.org/10.1371/journal.pone.0045377.
Brough HA, Cousins DJ, Munteanu A, Wong YF, Sudra A, Makinson K, et al. IL-9 is a key component of memory TH cell peanut-specific responses from children with peanut allergy. J Allergy Clin Immunol. 2014;134(6):1329–38 e10. https://doi.org/10.1016/j.jaci.2014.06.032.
Shik D, Tomar S, Lee JB, Chen CY, Smith A, Wang YH. IL-9-producing cells in the development of IgE-mediated food allergy. Semin Immunopathol. 2017;39(1):69–77. https://doi.org/10.1007/s00281-016-0605-x.
Antoniu SA. MEDI-528, an anti-IL-9 humanized antibody for the treatment of asthma. Curr Opin Mol Ther. 2010;12(2):233–9.
Parker JM, Oh CK, LaForce C, Miller SD, Pearlman DS, Le C, et al. Safety profile and clinical activity of multiple subcutaneous doses of MEDI-528, a humanized anti-interleukin-9 monoclonal antibody, in two randomized phase 2a studies in subjects with asthma. BMC Pulm Med. 2011;11:14. https://doi.org/10.1186/1471-2466-11-14.
Oh CK, Leigh R, McLaurin KK, Kim K, Hultquist M, Molfino NA. A randomized, controlled trial to evaluate the effect of an anti-interleukin-9 monoclonal antibody in adults with uncontrolled asthma. Respir Res. 2013;14:93. https://doi.org/10.1186/1465-9921-14-93.
Muraro A, Lemanske RF Jr, Hellings PW, Akdis CA, Bieber T, Casale TB, et al. Precision medicine in patients with allergic diseases: airway diseases and atopic dermatitis-PRACTALL document of the European academy of allergy and clinical immunology and the American Academy of Allergy, Asthma & Immunology. J Allergy Clin Immunol. 2016;137(5):1347–58. https://doi.org/10.1016/j.jaci.2016.03.010.
•• Schwartz DM, Farley TK, Richoz N, Yao C, Shih HY, Petermann F, et al. Retinoic Acid Receptor Alpha Represses a Th9 Transcriptional and Epigenomic Program to Reduce Allergic Pathology. Immunity. 2019;50(1):106–20 e10. https://doi.org/10.1016/j.immuni.2018.12.014 COMMENT: Study indicating the effect of retinoic acid on transcriptome of Th9 cells and repressing Th9 epigenome can control allergic lung pathology.
Lloyd CM, Harker JA. Epigenetic control of Interleukin-9 in asthma. N Engl J Med. 2018;379(1):87–9. https://doi.org/10.1056/NEJMcibr1803610.
McGregor MC, Krings JG, Nair P, Castro M. Role of biologics in asthma. Am J Respir Crit Care Med. 2018;199:433–45. https://doi.org/10.1164/rccm.201810-1944CI.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflicting financial interests.
Human and Animal Rights
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Basic and Applied Science
Rights and permissions
About this article
Cite this article
Angkasekwinai, P. Th9 Cells in Allergic Disease. Curr Allergy Asthma Rep 19, 29 (2019). https://doi.org/10.1007/s11882-019-0860-8
Published:
DOI: https://doi.org/10.1007/s11882-019-0860-8