Abstract
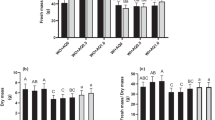
The enhancement of plant resistance to pest infestation, improving plant growth parameters and productivity is in sustainable agriculture programs. Hydrogen peroxide (H2O2) is among the most effective compounds acts as a messenger molecule involved in acclimatory signaling and triggering tolerance against various biotic and abiotic stresses. In this study, three cultivars of common bean (Phaseolus vulgaris L.), Nebraska, Paulista, and Valentino were treated with H2O2 to evaluate some vegetative parameters, yield and the natural infestations Tetranychus urticae Koch. Leaf area scored the highest size in Valentino cultivar in (742.5 and 722.3 cm2) in response to 1.5 and 1 mM of H2O2 respectively compared to 416.3 cm2 in the control. However, the Paulista cultivar reached the highest size 722.3 cm2 in response to 1.5 mM H2O2 followed by 702.3 cm2 in response to 1 mM H2O2 compared to 401.8 cm2 in the control. Valentino cultivar scored the highest plant heights 81.8 cm in response to 1.5 mM H2O2 followed by 79.2 cm in response to 1 mM H2O2 compared to 43.2 cm in the untreated plants. Valentino cultivar scored the highest number of seeds, pods, and branches compared to the other two cultivars, while Nebraska scored the lowest number of seeds, pods, and branches in response to 1.5 mM hydrogen peroxide compared to control. Nebraska cultivar scored the lowest number of spider mites, T. urticae compared to the other two cultivars in response to 1.5 mM compared to control. An increase in ROS or hydrogen peroxide in the cells in response to exogenous application of hydrogen peroxide could be toxic to the T. urticae and affect its feeding and subsequently mite reproduction. Results from the existing investigation showed that exogenic application of hydrogen peroxide enhanced the resistance of common beans in response to T. urticae feeding.











Similar content being viewed by others
References
Aidemark M, Andersson C, Rasmusson AG, Widell S (2009) Regulation of callose synthase activity in situ in alamethicin-permeabilized Arabidopsis and tobacco suspension cells. BMC Plant Biol 9(1):27. https://doi.org/10.1186/1471-2229-9-27
Alakhdar HH (2020) Efficacy of Chitosan Nano-particles Against Two Tetranychid mites and Two Associated Predaceous Mites (Acari: Tetranychidae: Phytoseiidae). Egy. Sci. J. Pestic. 6(1):8–13. https://doi.org/10.13140/RG.2.2.20972.31363/3
Alakhdar HH, Elsamahy MFM (2016) Relative Toxicity of Silica Nanoparticles to Two Tetranychids and Three Associated Predators. Egy J Biol Pest Control 26(2):283–286
Alakhdar HH, Zeinab E, Rabie EM (2015) Evaluation Some Genotypes of Soybeans Yield Under Pest Infestation. Inter J of Sci Res Agric Sci 2:007–017. https://doi.org/10.13140/RG.2.2.15939.14888
Alvarez ME, Pennell RI, Meijer PJ, Ishikawa A, Dixon RA, Lamb C (1998) Reactive oxygen intermediates mediate a systemic signal network in the establishment of plant immunity. Cell 92(6):773–784. https://doi.org/10.1016/S0092-8674(00)81405-1
Anonymous (2003) SAS Statistics and graphics guide, release 9.1. SAS Institute, Cary, North Carolina 27513, USA.
Asai T, Tena G, Plotnikova J, Willmann MR, Chiu W-L, Gomez- Gomez L, Boller T, Ausebel FM, Sheen J (2002) MAP kinase signaling cascade in Arabidopsis innate immunity. Nature 415:977–983. https://doi.org/10.1038/415977a
Bacic A, Fincher GB, Stone BA (2009) Chemistry, Biochemistry and Biology 1,3-β-Glucans, and Related Polysaccharides. Elsevier, Amsterdam, Boston, p 677
Dat JF, Pellinen R, Beeckman T, Kangasja¨rvi J, Langebartels C, Inze D, Van BF (2003) Changes in hydrogen peroxide homeostasis trigger active cell death in tobacco. Plant J. 33:621–632. https://doi.org/10.1046/j.1365-313X.2003.01655.x
El-Noemani AA, El-Zeiny HA, El-Gindy AM, El-Sahhar EA, El-Shawadfy MA (2010) Performance of some bean (Phaseolus vulgaris L.) varieties under different irrigation systems and regimes. Aust. J. Basic Appl. Sci. 4(12):6185–6196
FAOSTAT (2017) Green Bean World Statistics. Major food and agricultural commodities producers—Countries by commodity. Available online: www.faostat.fao.org (accessed on 23 October 2017).
Granito M, Paolini M, Pérez S (2008) Polyphenols and antioxidant capacity of Phaseolus vulgaris stored under extreme conditions and processed. LWT - Food Sci Technol 41:994–999. https://doi.org/10.1016/j.lwt.2007.07.014
Jacobs AK, Lipka V, Burton RA, Panstruga R, Strizhov N, Schulze-Lefert P, Fincher GB (2003) An arabidopsis callose synthase, GSL5, is required for wound and papillary callose formation. Plant Cell 15:2503–2513. https://doi.org/10.1105/tpc.016097
Kutoš T, Golob T, Kač M, Plestenjak A (2003) Dietary fibre content of dry and processed beans. Food Chem 80(02):231–235. https://doi.org/10.1016/S0308-8146(02)00258-3
Kwak JM, Mori IC, Pei ZM, Leonhardt N, Torres MA, Dangl JL, Bloom RE, Bodde S, Jones JDG, Schroeder JI (2003) NADPH oxidase Atrboh D and Atrboh F genes function in ROS-dependent ABA signaling in Arabidopsis. EMBO J 22:2623–2633. https://doi.org/10.1093/emboj/cdg277
Levine A, Tenhaken R, Dixon RA, Lamb C (1994) H2O2 from the oxidative burst orchestrates the plant hypersensitive disease resistance response. Cell 79:583–593. https://doi.org/10.1016/0092-8674(94)90544-4
Martinez-Ferrer MT, Jacas JA, Piolles-Moles JL, Aucejo-Romero S (2006) Approaches for sampling the two-spotted spider mite (Acari: Tetranychidae) on clementines in Spain. J Econ Entomol 99:1490–1499. https://doi.org/10.1603/0022-0493-99.4.1490
Miller G, Suzuki N, Ciftci-Yilmaz S, Mittler R (2010) Reactive oxygen species homeostasis and signaling during drought and salinity stresses. Plant Cell Environ 33:453–467. https://doi.org/10.1111/j.1365-3040.2009.02041.x
Mozaffaria F, Abbasipoura H, Garjanb AS, Mahmoud M (2013) Toxicity and Oviposition Deterrence and Repellency of Mentha pulegium (Lamiacaeae) Essential Oils against Tetranychus urticae Koch (Tetranychidae). J Essen Oil B Plants 16(5):575–581. https://doi.org/10.1080/0972060X.2013.854500
Neill SJ, Desikan R, Clarke A, Hurst RD, Hancock JT (2002) Hydrogen peroxide and nitric oxide as signaling molecules in plants. J Exp Bot 53(1237):1247. https://doi.org/10.1093/jexbot/53.372.1237
Petersen LN, Ingle RA, Knight MR, Denby KJ (2009) OXI1 protein kinase is required for plant immunity against Pseudomonas syringae in Arabidopsis. J Exp Bot 60:3727–3735. https://doi.org/10.1093/jxb/erp219
Rentel MC, Lecourieux D, Ouaked F, Usher SL, Petersen L, Okamoto H, Knight H, Peck SC, Grierson CS, Hirt H (2004) Oxi1 kinase is necessary for oxidative burst-mediated signaling in Arabidopsis. Nature 427:858–861. https://doi.org/10.1038/nature02353
Van-Camp W, Van-Montagu M, Inze D (1998) H2O2 and NO: redox signals in disease resistance. Trends in Plant Sci 3:330–334. https://doi.org/10.1016/S1360-1385(98)01297-7
Zhang X, Dong FC, Gao JF, Song CP (2001) Hydrogen peroxide-induced changes in intracellular pH of guard cells precede stomatal closure. Cell Res 1:37–43. https://doi.org/10.1038/sj.cr.7290064
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Ritu Chaudhary.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Alakhdar, H.H., Shoala, T. Exogenous application of hydrogen peroxide in different resistant bean cultivars of Phaseolus vulgaris to Tetranychus urticae (Acari: Tetranychidae). Arthropod-Plant Interactions 15, 439–445 (2021). https://doi.org/10.1007/s11829-021-09829-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-021-09829-1