Abstract
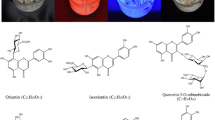
The buckwheat is a plant species with a high ability to produce secondary metabolites such as flavonoids. Together with rutin, the dominant flavonoid of the Fagopyrum Mill genus representatives (The Common buckwheat (F. esculentum Moench), the Tartary buckwheat (F. tataricum (L.) Gaertn.) and the Perennial buckwheat (F. cymosum Meissn.)), quercetin, vitexin, quercitrin, orientin, anthocyanides, phenolcarbonic acids and other compounds were found in buckwheat plants. Due to their considerable reactivity, flavonoids participate in the processes of photosynthesis, respiration, growth, reproduction and defence against different stresses. The purpose of this article is to overview the processes of flavonoid biosynthesis and accumulation during the growth and development stages of buckwheat plants under the influence of external factors regarding its species diversity. Flavonoids content and induction were examined in vitro culture.

Similar content being viewed by others
References
Ahlgrimm ED (1955) Uber den Gesamt flavonol sowie den Rutin gehalt verschiedener alten Blatter bei Fagopyrum. Arten Naturwiss 42:465–466
Alekseeva EC, Shevchuk VК, Shevchuk TE (1991) Selection of buckwheat on resistance to pathogens. Agropromizdat, Moscow
Alekseyeva EC (1999) Selection of Podolsky varieties of buckwheat. Chernovtsy, Ruta
Andersen OM, Markham KR (2005) Flavonoids: chemistry, biochemistry and application. Taylor & Francis, New York, pp 397–441
Anisimova MM (2011) Pharmacological researchers of the grass of Common Buckwheat (Fagopyrum sagittatum Gilib.). Samara 4:17–32
Barsukova EN, Кlykov AG, Chaikina EL (2019) Usage of the method of tissure culture for creation new forms of Fagopirum esculentum Moench. Rus Agric Sci 5:3–6. https://doi.org/10.31857/S2500-2627201953-6
Bate-Smith EC (1962) The phenolic constituents of plants and their taxonomic significance Dicotyledons. J Lin Soc Bot 58:95–173
Bohm BA (1998) Introduction of flavonoids. Harwood Academic Publishers, Singapore
Brunori A, Vegvari G (2007) Variety and location influence on the rutin content of the grain of Buckwheat (Fagopyrum esculentum Moench and Fagopyrum tataricum Gaertn.) Grown in Central and Southern Italy. In: Advances in buckwheat research: proceedings of the 10th International Symposium on buckwheat. Yangling, Shaanxi, China, pp 349‒357
Christensen KB, Kaemper M, Loges R, Fretté XC, Christensen LP, Grevsen K (2010) Effects of nitrogen fertilization, harvest time, and species on the concentration of polyphenols in aerial parts and seeds of normal and tartary buckwheat (Fagopyrum sp.). Europ J Hort Sci 75:153–164
Debeaujon I, Nesi N, Perez P, Devic M, Grandjean O, Caboche M, Lepiniec L (2003) Proanthocyanidin-accumulating cells in Arabidopsis testa: regulation of differentiation and role in seed development. Plant Cell 15:2514–2531. https://doi.org/10.1105/tpc.014043
Debski H, Szwed M, Wiczkowski W, Szawara-Nowak D, Baczek N, Horbowicz M (2016) UV-B radiation increases anthocyanin levels in cotyledons and inhibits the growth of common buckwheat seedlings. Acta Biol Hung 67:403–411. https://doi.org/10.1556/018.67.2016.4.6
Debski H, Wiczkowski W, Szawara-Nowak D, Baczek N, Chrzanowski G, Horbowicz M (2018) Effects of glyphosate and fluazifop-P-butyl on flavonoids content and growth of common buckwheat (Fagopyrum esculentum Moench). Fresen Environ Bul 27:91–97
Dietrych-Szostak D, Oleszek W (1999) Effect of processing on the flavonoid content in buckwheat (Fagopyrum esculentum Moench) grain. J Agric Food Chem 47:4384–4387
Druzhkov AA (1972) Rutin content in diploid and tetraploid forms of the two varieties of buckwheat in connection with the heterostyle. News Academy of Sciences of the Turkmen SSR. Ser Biol Sci 1:9–14
Fabjan N, Rode J, Kosǐr IJ, Wang Z, Zhang Z, Kreft I (2003) Tartary buckwheat (Fagopyrum tataricum Gaertn) as a source of dietary rutin and quercitrin. J Agric Food Chem 51(22):6452–6455. https://doi.org/10.1021/jf034543e
Fesenko NV, Fesenko NN, Romanova OI, Alekseeva EU, Suvorova GN (2006) Buckwheat. ARIPG, Sankt-Peterburg
Forkmann G, Heller W (1999) Biosynthesis of flavonoids. Comp Nat Prod Chem 1:713–748
Gorkova IV (2002) Flavonoid accumulation dynamics in the ontogenesis of bucwheat and biochemical changes of seeds during the process of storage. Abstract dis cand agri-sciences, Oryol
Grotewold E (2006) The science of flavonoids. Springer, New York
Gumerova EA, utkina DB, Rumyanseva NI (2010) Phenolic compounds and antioxidant activity in suspension and cullus culture of tartary buckwheat. In: Bioantioxidant: theses of the report VIII International conference. RUDN, Moscow, pp 129‒131
Gupta N, Sharma SK, Rana JC, Chauhan RS (2011) Expression of flavonoid biosynthesis genes vis-à-vis rutin content variation in different growth stages of Fagopyrum specie. J Plant Physiol 168:2117–2123. https://doi.org/10.1016/j.jplph.2011.06.018
Holasova M, Fiedlerova V, Smrcinova H, Orsak LJ, Vavreinova S (2002) Buckwheat—the source of antioxidant activity in functional foods. Food Res Int 35:207–211
Hollman PC, Arts CW (2000) Flavonols, flavons and flavanols—nature, occurrence and dietary burden. J Sci Food Agric 80:1081–1093
Horbowicz M, Wiczkowski W, Koczkodaj D, Saniewski M (2011) Effects of methyl jasmonate on accumulation of flavonoids in seedlings of common buckwheat (Fagopyrum esculentum Moench). Acta Biol Hung 62:265–278. https://doi.org/10.1556/ABiol.62.2011.3.6
Horbowicz M, Wiczkowski W, Szawara-Nowak D, Sawicki T, Kosson R, Sytykiewicz H (2015) The level of flavonoids and amines in de-etiolated and methyl jasmonate treated seedling of common buckwheat. Phytoch Lett 13:15–19. https://doi.org/10.1016/j.phytol.2015.05.011
Jeong H, Sung J, Yang J, Kim Y, Jeong HS, Lee J (2018) Effect of sucrose on the functional composition and antioxidant capacity of buckwheat (Fagopyrum esculentum M.) sprouts. J Func Foods 43:70–76
Jiang P, Burczynski F, Campbell C, Pierce G, Austria JA, Briggs CJ (2007) Rutin and flavonoid contents in three buckwheat species Fagopyrumesculentum, F. tataricum and F. homotropicum and their protective effects against lipid peroxidation. Food Res Int 40(3):356–364. https://doi.org/10.1016/j.foodres.2006.10.009
Jovanovic ZS, Milosevic JD, Radovic SR (2006) Antioxidative enzymes in the response of buckwheat (Fagopyrum esculentum Moench) to ultraviolet B radiation. J Agric Food Chem 54:9472–9478. https://doi.org/10.1021/jf061324v
Kalinova J, Dadakova E (2006) Varietal and year variation of rutin content in common buckwheat (Fagopyrum esculentum Moench). Cer Res Com 34:1315–1321. https://doi.org/10.1556/CRC.34.2006.4.274
Kalinova JP, Vrchotova N, Tríska J (2019) Phenolics levels in different parts of common buckwheat (Fagopyrum esculentum) achenes. J Cer Sci 85:243–248
Karabanov IA (1981) Flavonoids in the plant world. Uragi, Minsk
Kazantseva VV, Goncharuk EA, Fesenko AN, Shirokova AV, Zagoskina NV (2015) Features of the Phenolics' formation in seedlings of different varieties of buckwheat (Fagopyrum esculentum Moench). Agric Biol 50:611–619
Kim DO, Lee CY (2004) Comprehensive study on vitamin C equivalent antioxidant capacity (VCEAC) of various polyphenolics in scavenging a free radical and its structural relationship. Crit Rev Food Sci Nutr 44:253–273
Kim S-J, Kawaharada C, Suzuki T, Saito K, Hashimoto N, Takigawa S, Noda T, Matsuura-Endo C, Yamauchi H (2006) Effect of natural light periods on rutin, free amino acid and vitamin C contents in the sprouts of common (Fagopyrum esculentum Moench) and tartary (F. tataricum Gaertn.) buckwheats. Food Sci Tech Res 12:199–205. https://doi.org/10.3136/fstr.12.199
Kim S-J, Maeda T, Sarker MZI, Takigawa S, Matsuura-Endo C, Yamauchi H, Mukasa Y, Saito K, Hashimoto N, Noda T, Saito T, Suzuki T (2007) Identification of anthocyanins in the sprouts of buckwheat. J Agric Food Chem 55:6314–6318. https://doi.org/10.1021/jf0704716
Kim S-J, Zaidul ISM, Suzuki T, Mukasa Y, Hashimoto N, Takigawa S, Noda T, Matsuura-Endo C, Yamauchi H (2008) Comparison of phenolic compositions between common and tartary buckwheat (Fagopyrum) sprouts. Food Chem 110:814–820. https://doi.org/10.1016/j.foodchem.2008.02.050
Kiprovski B, Mikulic-Petkovsek M, Slatnar A, Veberic R, Stampar F, Malencic D, Latkovic D (2015) Comparison of phenolic profiles and antioxidant properties of European Fagopyrum esculentum cultivars. Food Chem 185:41–47. https://doi.org/10.1016/j.foodchem.2015.03.137
Kiselev VE, Kovalenko VN, Minaeva VG (1985) Buckwheat as a source of flavonoids. Science, Novosibirsk
Kitabayashi H, Ujihant A, Hirose T, Minami M (1995) Varietal differences and heritability for rutin content in common buckwheat, Fagopyrum esculentum Moench. Breed Sci 45:75–79
Klykov AG, Moiseenko LM, Gorovoy PG (2018) Biological resources of species of the genus Fagopyrum Mill. (Buckwheat) in the Russian Far East. Vladivostok
Korotkov AV (2001) Formation of yield and quality of buckwheat grain using growth regulators. Abstract dis cand agri-sciences, Moscow
Krasilnikova LO, Avksentieva OO, Zhmurko VV (2007) Plant biochemistry. Kolorit, Harkiv
Krause J (1978) Uber die bedeutung des endosperms fur die flavonoidsynthese in den kotyle nen von Fagopyrum esculentum Moench. Z Pflanzenphysiol 90:319–330
Krause J, Reznik H (1972) Identefezierung von Phenylpropanderivaten aus den Laubblattem Fagopyrum esculentum Moench. Z Pflanzenphysiol 68:115–120
Kreft S, Knapp M, Kreft I (1999) Extraction of rutin from buckwhet (Fagopyrum esculentum Moench) seeds and determination by capillary electrophoresis. J Agr Food Chem 47:4649–4652. https://doi.org/10.1021/jf990186p
Kreft S, Strukelj B, Gaberscik A, Kreft I (2002) Rutin in buckwheat herbs grown at different UV-B radiation levels: comparison of two UV spectrophotometric and an HPLC method. J Exp Bot 53:1801–1804. https://doi.org/10.1093/jxb/erf032
Kreft I, Fabjan N, Yasumoto K (2006) Rutin content in buckwheat (Fagopyrum esculentum Moench) food materials and products. Food Chem 98:508–512
Lee S-Y, Cho S-I, Park M-H, Kim Y-K, Choi J-E, Park S-U (2007) Growth and rutin production in hairy root cultures of buckwheat (Fagopyrum esculentum M.). Prep Biochem Biotechnol 37:239–246. https://doi.org/10.1080/10826060701386729
Li S-Q, Zhang QH (2001) Advances in the development of functional foods from buckwheat. Crit Rev Food Sci Nut 41:451–464. https://doi.org/10.1080/20014091091887
Li X, Park NI, Xu H, Woo S-H, Park CH, Park SU (2010) Differential expression of flavonoid biosynthesis genes and accumulation of phenolic compounds in common buckwheat (Fagopyrum esculentum). J Agric Food Chem 58:12176–12181. https://doi.org/10.1021/jf103310
Li X, Park NI, Park CH, Kim SG, Lee SY, Park SU (2011) Influence of sucrose on rutin content and flavonoid biosynthetic gene expression in seedlings of common buckwheat (Fagopyrum esculentum Moench). Plant OMICS 4:215–219
Liu L-P, Zang X-Y, Yuan Q-Y, Cai Q-S (2006) Mitigating effect of exogenous sucrose on root growth of buckwheat (Fagopyrum esculentum Moench) seedlings under salt stress. Plant Physiol Commun 42:847–850
Makarenko OA, Levitsky AP (2013) Physiological functions of flavonoids in plants. Physiol Biochem Cult Plants 45:100–112
Margna UV (1990) Interrelation of the flavonoids biosynthesis with the primary plants metabolism. VINITI, Moscow
Margna U, Laanest L, Margna E, Vainjärv T (1985) L-Tyrosine as a precursor of flavonoids in buckwheat cotyledons. Zeitschrift fur Naturforschung Sect C J Biosci 40:154–159
Margna U, Margna E, Paluteder A (1990) Localization and distribution of flavonoids in buckwheat seedling cotyledons. J Plant Physiol 136:166–171
Metzger BT, Robbins MG, Barnes DM (2010) Longitudinal expression of antioxidant phytochemicals in buckwheat (Fagopyrum esculentum Moench). J Herbs Spices Med Plants 16:106–118
Mierziak J, Kostyn K, Kulma A (2014) Flavonoids as important molecules of plant interactions with the environment. Molecules 19:16240–16265. https://doi.org/10.3390/molecules191016240
Minaeva VG (1978) Flavonoids in plant ontogenesis and their practical use. Science, Novosibirsk
Moumou Y, Trotin F, Dubois J, Vasseur J, El-Boustani E (1992) Influence of culture conditions on polyphenol production by Fagopyrum esculentum tissue cultures. J Nat Prod 55:33–38
Murphy A, Peer WA, Taiz L (2000) Regulation of auxin transport by aminopeptidases and endogenous flavonoids. Planta 211:315–324
Murri IK (1958) Biochemistry buckwheat. In: Biochemistry of the cultural plant. Argistate edition, Moscow, Leningrad, pp 642‒698
Nam T-G, Lim YJ, Eom SH (2018) Flavonoid accumulation in common buckwheat (Fagopyrum esculentum) sprout tissues in response to light. Hort Environ Biotech 59:19–27. https://doi.org/10.1007/s13580-018-0003-5
Ndakidemi PA, Dakora FD (2003) Legume seed flavonoids and nitrogenous metabolites as signals and protectants in early seedling development. Funct Plant Biol 30:729–745. https://doi.org/10.1071/FP03042
Olschlager C, Regos I, Zeller FJ, Treutter D (2008) Identification of galloylated propelargonidins and procyanidins in buckwheat grain and quantification of rutin and flavanols from homostylous hybrids originating from F. esculentum x F. homotropicum. Phytochem 69:1389–1397. https://doi.org/10.1016/j.phytochem.2008.01.001
Oomah BD, Mazza G (1996) Flavonoids and antioxidative activities in buckwheat. J Agric Food Chem 44:1746–1750
Park BJ, Park JI, Chang KJ, Park CH (2004) Comparison in rutin content in seed and plant of tartary buckwheat (Fagopyrum tartaricum). In: Proceedings of the 9th international symposium on Buckwheat, Prague, pp 626‒629
Park CH, Yeo HJ, Park YJ, Morgan AM, Valan Arasu M, Al-Dhabi NA, Park SU (2017) Influence of indole-3-acetic acid and gibberellic acid on phenylpropanoid accumulation in common buckwheat (Fagopyrum esculentum Moench) sprouts. Molecules 22:374. https://doi.org/10.3390/molecules22030374
Parr AJ, Bolwell GP (2000) Phenols in the plant and man. The potential for possible nutritional enhancement of the diet by modifying the phenols content or profile. J Sci Food Agric 80:985–1012
Pavlovskaya NE, Zotikov VI, Kopniyenko NN (2010) Biochemistry of legumes and cereals: monograph. Orel SAU, Orel
Polekhina NN (2013) Dynamics of flavonoid accumulation in ontogenesis of varieties of buckwheat (Fagopyrum esculentum Moench) zoned in the Orel region. Orel SAU, Orel
Quettier-Deleu C, Gressier B, Vasseur J, Dine T, Brunet C, Luyckx M, Cazin M, Cazin J-C, Bailleul F, Trotin F (2000) Phenolic compounds and antioxidant activities of buckwheat (Fagopyrum esculentum Moench) hulls and flour. J Ethnopharm 72:35–42. https://doi.org/10.1016/S0378-8741(00)00196-3
Quian JY, Mayer D, Kuhn M (1999) Flavonoids in fine buckwheat (Fagopyrum esculentum Moench) flour and their free radical scavanging activities. Dtsch Lebensm-Rundsch 95:343–349
Regvar M, Bukovnik U, Likar M, Kreft I (2012) UV-B radiation affects flavonoids and fungal colonisation in Fagopyrum esculentum and F. tataricum. Cent Eur J Biol 7:275–283
Ren S-C, Sun J-T (2014) Changes in phenolic content, phenylalanine ammonia-lyase (PAL) activity, and antioxidant capacity of two buckwheat sprouts in relation to germination. J Funct Foods 7:298–304. https://doi.org/10.1016/j.jff.2014.01.031
Sakuta M (2014) Diversity in plant red pigments: anthocyanins and betacyanins. Plant Biotechnol Rep 8:37–48. https://doi.org/10.1007/s11816-013-0294-z
Santos-Buelga C, Scalbert A (2000) Proanthocyanidins and tannin-like compounds—nature, occurrence, dietary intake and effects on nutrition and health. J Sci Food Agric 80:1094–1117
Sato H, Sakamura S (1975) Isolation and identification of flavonoids in immature buckwheat seed (Fagopyrum esculentum Moench). Agric Chem Soc Jpn 49:53–55
Sawai Y (2016) Effect of light intensity on flavonoid contents of common and tartary buckwheat sprouts. Nippon Shokuhin Kagaku Kogaku Kaishi 63:382–387. https://doi.org/10.3136/nskkk.63.382
Sedej I, Sakac M, Mandic A, Misan A, Tumbas V, Canadanovic-Brunet J (2012) Buckwheat (Fagopyrum esculentum Moench) grain and fractions: antioxidant compounds and activities. J Food Sci 77:954–959. https://doi.org/10.1111/j.1750-3841.2012.02867.x
Sidorchuk ON (2002) Red-flowered mutants in the world gene pool collection of the genus Fagopyrum Mill. In: Collection of scientific papers of the international conference dedicated to the 30th anniversary of the research Institute of cereals. Kemenets-Podolsky, pp 169‒176
Smirnov OE, Kosyan AM, Kosyk OI, Taran NY (2015) Response of phenolic metabolism induced by aluminium toxicity in Fagopyrum esculentum Moench. Plants Ukrain Bioch J 87:129–135. https://doi.org/10.15407/ubj87.06.129
Suzuki T, Kim S-J, Yamauchi H, Takigawa S, Honda Y, Mukasa Y (2005) Characterization of a flavonoid 3-O-glucosyltransferase and its activity during cotyledon growth in buckwheat (Fagopyrum esculentum). Plant Sci 169:943–948. https://doi.org/10.1016/j.plantsci.2005.06.01
Suzuki T, Kim S-J, Mohamed ZIS, Mukasa Y, Takigaw S, Matsuura-Endo C, Yamauchi H, Hashimoto N, Noda T, Saito T (2007) Structural identification of anthocyanins and analysis of concentrations during growth and flowering in buckwheat (Fagopyrum esculentum Moench) petals. J Agric Food Chem 55:9571–9575. https://doi.org/10.1021/jf071474l
Suzuki T, Watanabe M, Iki M, Aoyagi Y, Kim S-J, Mukasa Y, Yokota S, Takigawa S, Hashimoto N, Noda T, Yamauchi H, Matsuura-Endo C (2009) Time-course study and effects of drying method on concentrations of γ-aminobutyric acid, flavonoids, anthocyanin, and 2″-hydroxynicotianamine in leaves of buckwheats. J Agric Food Chem 57:259–264. https://doi.org/10.1021/jf802731d
Taguchi G (2016) Flavonoid biosynthesis in buckwheat. In: Molecular breeding and nutritional aspects of buckwheat. Elsevier, London, pp 377‒386. https://doi.org/10.1016/B978-0-12-803692-1.00030-4
Tanaka N (1996) Rutin and other polyphenols in Fagopyrum esculentum hairy roots. Nat Med 50:269–273
Tien NNT, Trinh LND, Inoue N, Morita N, Van Hung P (2018) Nutritional composition, bioactive compounds, and diabetic enzyme inhibition capacity of three varieties of buckwheat in Japan. Cer Chem 95:615–624. https://doi.org/10.1134/S1062359018020140
Tomas-Barberan FA, Clifford MN (2000) Flavanones, chalcones and dihydrochalcones—nature, ocurrence and dietary burden. J Sci Food Agric 80:1073–1080
Tronchet J (1970) Evolution des flavonols dans les petioles et les limbes de Paeonia corallina Retz. au cours du development. Ann Sci Univ Besancon Bot 7:15–26
Troyer JR (1956) Quantitative changes in buckwheat flavonoid substances during seedling development. Plant Physiol 31:75–78i
Tsai H, Deng H, Tsai S, Hsu Y (2012) Bioactivity comparison of extracts from various parts of common and tartary buckwheats: evaluation of the antioxidant- and angiotensin-converting enzyme inhibitory activities. Chem Centr J 6:78. https://doi.org/10.1186/1752-153X-6-78
Tsurunaga Y, Takahashi T, Katsube T, Kudo A, Kuramitsu O, Ishiwata M, Matsumoto S (2013) Effects of UV-B irradiation on the levels of anthocyanin, rutin and radical scavenging activity of buckwheat sprouts. Food Chem 141:552–556. https://doi.org/10.1016/j.foodchem.2013.03.032
Tumova L, Tuma J (2009) The effect of paraquat on flavonoid production in Fagopyrum esculentum cultures in vitro. Cer Res Communic 37:557–560
Tumova L, Píchová M, Dušek J (2007) Fagopyrum esculentum in vitro. Ceska Slov Farm 56:125–128
Vegvari G, Brunori A, Sandor G, Jocsak I, Rabnecz G (2008) The influence of growing place on the rutin content on Fagopyrum esculentum and Fagopyrum tataricum varieties seeds. Cer Res Com 36:599–602
Vysochina GI (2004) Phenolic compounds in taxonomy and phylogeny of the buckwheat family. Novosibirsk
Watanabe M (1998) Catechins as antioxidants from buckwheat (Fagopyrum esculentum Moench) groats. J Agric Food Chem 46:839–845
Watanabe M, Ito M (2003) Effect of light on the content of phenolic compounds in buckwheat seedlings. Nippon Shokuhin Kagaku Kogaku Kaishi 50:32–34. https://doi.org/10.3136/nskkk.50.32
Watanabe M, Ohshita Y, Tsushida T (1997) Antioxidant compounds from Buckwheat (Fagopyrum esculentum Möench) hulls. J Agric Food Chem 45:1039–1044
Wiczkowski W, Szawara-Nowak D, Debski H, Mitrus J, Horbowicz M (2014) Comparison of flavonoids profile in sprouts of common buckwheat cultivars and wild tartary buckwheat. Int J Food Sci Technol 49:1977–1984
Wieslander G, Fabjan N, Kreft I et al. (2005) An experimental study on health effects of consumption of a new type of tartary buckwheat biscuits with high levels of the antioxidant rutin. In: III International symposium on buckwheat and the dietary culture, August 8–12, 2005, Xichang. China, pp 36‒37
Winkel-Shirley B (1999) Evidence for enzyme complexes in the phenylpropanoid and flavonoid path ways. Physiol Plant 107:142–155
Yan C, Dongli G, Baili F, Xiaoli G, Jinfeng G, Shouqiang A (2007) Flavonoides content of buckwheat in different development stage. In: Advances in buckwheat research: proceedings of the 10th International Symposium on buckwheat. Yangling, Shaanxi, China, pp 417‒421
Yarosh NP, Golenkovsky KP, Krotov AS (1967) Tartary buckwheat is a rich source of biologically active flavonoids. Plant Resour 1:40–47
Ylstra B, Muskens M, van Tunen AT (1996) Flavonoids are not essential for fertilization in Arabidopsis thaliana. Plant Mol Biol 32:1155–1167. https://doi.org/10.1007/BF00041399
Zagoskina NV, Kazantseva VV, Fesenko AN, Shirokova AV (2018) Accumulation of phenolic compounds at the initial steps of ontogenesis of Fagopyrum esculentum plants that differ in their ploidy levels. Biol Bull 45:171–178
Zaprometov MN (1993) Phenolic compounds: distribution, metabolism and function in plants. Science, Moscow
Zaprometov MN (1996) Phenolic compounds and their role in plant life. Science, Moscow
Zhanayeva TA (1988) Catabolism of phenolic compounds in connection with their functions in some higher plants. Abstract dis d-ra biol sciences, Novosibirsk
Acknowledgements
This paper was supported by Complex program of fundamental research of the FEB RAS "Far East" in 2018–2020 Project no. 18-5-025.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Borovaya, S.A., Klykov, A.G. Some aspects of flavonoid biosynthesis and accumulation in buckwheat plants. Plant Biotechnol Rep 14, 213–225 (2020). https://doi.org/10.1007/s11816-020-00614-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11816-020-00614-9