Abstract
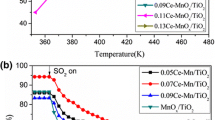
The catalytic oxidation of hydrogen sulfide (H2S) to elemental sulfur was studied over CeO2-TiO2 catalysts. The synthesized catalysts were characterized by various techniques such as X-ray diffraction, BET, X-ray photoelectron spectroscopy (XPS), temperature-programmed desorption of ammonia, and scanning electron microscopy (SEM). Catalytic performance studies of the CeO2-TiO2 catalysts showed that H2S was successfully converted to elemental sulfur without considerable emission of sulfur dioxide. CeO2-TiO2 catalysts with Ce/Ti=1/5 and 1/3 exhibited the highest H2S conversion, possibly due to the uniform dispersion of metal oxides, high surface area, and high amount of acid sites.
Similar content being viewed by others
References
B. M. Khudenko, G. M. Gitman and T. E. P. Wechsler, J. Environ. Eng., 119, 1233 (1993).
J. S. Eow, Environ. Prog. Sustain., 21, 143 (2002).
J. A. Lagasm J. Borsboom and P. H. Brben, Oil Gas J., 10, 68 (1988).
R. Ketter and N. Liermann, Oil Gas J., 11, 63 (1982).
R. Kettner, T. Lubcke and N. Liermann, Eur. Patent, 0078690 (1983).
P. F. M. T. van Nisselrooy and J. A. Lagas, Catal. Today, 16, 263 (1993).
J. Borsboom, B. G. Goar, G. Heijkoop and J. A. Lagas, Sulphur, 220, 44 (1992).
X. Zhang, Y. Tang, S. Qu, J. Da and Z. Hao, ACS Catal., 5, 1053 (2015).
M. Y. Shin, Ch. M. Nam, D. W. Park and J. S. Chung, Appl. Catal. A., 211, 213 (2001).
K. T. Li and Z. H. Chi, Appl. Catal. B., 31, 173 (2001).
K. T. Li and Z. H. Chi, Appl. Catal. A., 206, 197 (2001).
D. W. Park, B. K. Park, D. K. Park and H. C. Woo, Appl. Catal. A., 223, 215 (2002).
B. G. Kim, W. D. Ju, I. Kim, H. C. Woo and D. W. Park, Solid State Ion., 172, 135 (2004).
M. I. Kim, D. W. Park, S. W. Park, X. Yang, J. S. Choi and D. J. Suh, Catal. Today, 111 212 (2006).
K. T. Li and C. H. Huang, Ind. Eng. Chem. Res., 45, 7096 (2006).
K. V. Bineesh, D. R. Cho, S. Y. Kim, B. R. Jermy and D. W. Park, Catal. Commun., 9, 2040 (2008).
K. V. Bineesh, D. K. Kim, M. I. Kim and D. W. Park, Appl. Clay Sci., 53, 204 (2011).
K. V. Bineesh, D. K. Kim, M. I. Kim, M. Selvaraj and D. W. Park, Dalton Trans., 40, 3938 (2011).
K. V. Bineesh, D. K. Kim, D. W. Kim, H. J. Cho and D. W. Park, Energy Environ. Sci., 3, 302 (2010).
K. V. Bineesh, M. I. Kim, M. S. Park, K. Y. Lee and D. W. Park, Catal. Today, 175, 183 (2011).
K. V. Bineesh, M. I. Kim, G. H. Lee, M. Selvaraj, K. Hyun and D. W. Park, J. Ind. Eng. Chem., 18, 1845 (2012).
M. I. Kim, G. H. Lee, D. W. Kim, D. H. Kang and D. W. Park, Korean J. Chem. Eng., 31, 2162 (2104).
M. D. Soriano, P. Concepcion, P. Botella and J. M. Lopez Nieto, Top. Catal., 54, 729 (2011).
K. I. Hadjiivanov and D. G. Klissurski, Chem. Soc. Rev., 25, 61 (1996).
M. R. Hoffmann, S. T. Martin, W. Y. Choi and D. W. Bahnemann, Chem. Rev., 95, 69 (1995).
M. Flytzani-Stephanopoulos, T. L. Zhu and Y. Li, Catal. Today, 62, 145 (2000).
S. Watamabe, X. Ma and C. Song, J. Phys. Chem. E., 113, 14249 (2009).
Z. M. Shi, W. G. Yu and X. Bayar, Scr. Mater., 50, 885 (2004).
T. Lopez, F. Rojas, R. Alexander-Katz, F. Galindo, A. Balankin and A. Buljan, J. Solid State Chem., 177, 1873 (2004).
S. X. Yang, W. P. Zhu, Z. P. Jiang, Z. X. Chen and J. B. Wang, Appl. Surf. Sci., 252, 8499 (2006).
M. F. Luo, J. Chen, L. S. Chen, J. Q. Lu, Z. C. Feng and C. Li, Chem. Mater., 13, 197 (2001).
K. Josef Antony Raj and B. Viswanathan, Indian J. Chem. A., 49, 401 (2010).
B. Jiang, Z. Wu and Y. Liu, J. Hazard. Mater., 145, 1249 (2009).
X. Gao, Y. Jiang, Y. Zhong, Z. Luo and K. Cen, J. Hazard. Mater., 174, 734 (2010).
S. Yang, W. Zhu, Z. Jiang, Z. Chen and J. Wang, Appl. Surf. Sci., 252, 8499 (2006).
P. Burroughs, A. Hammett, A. F. Orchard and G. Thornton, J. Chem. Soc., Dalton Trans., 17, 1686 (1976).
A. A. Tsyganenko, D. V. Pozdnyakov and V. N. Filimonov, J. Mol. Struct., 29, 299 (1975).
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is dedicated to Prof. Seung-Il Woo on the occasion of his retirement from KAIST.
Rights and permissions
About this article
Cite this article
Kang, DH., Kim, MI. & Park, DW. Selective oxidation of H2S to sulfur over CeO2-TiO2 catalyst. Korean J. Chem. Eng. 33, 838–843 (2016). https://doi.org/10.1007/s11814-015-0240-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-015-0240-6