Abstract
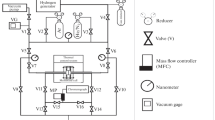
We conducted the WGS reaction on a catalytic membrane reactor consisting of a WGS catalyst bed, Pt/CeO2 and thin, defect-free, Pd-Cu alloy membranes. The presence of CO and other gases with H2 reduced the H2 permeation through the membrane by more than 50% and the effect of the other gases on the permeation reduction decreased in the following order: CO>CO2>N2. In a catalytic membrane reactor with helium sweep gas, the CO conversion was improved by about 65% compared with the catalyst without any membrane, and the CH4 formed from an undesirable side reaction was significantly reduced. Although the H2 permeation was severely reduced by surface phenomena such as blocking of available H2 dissociation sites by CO, CO2 and steam, the CO conversion was notably improved by the membrane presence. Moreover, the CO conversion was maintained at 98% even after 60 h of reaction and our Pd-Cu-Ni alloy membrane withstood the exposure of CO and the other gases. However, for separation of pure H2, a newly designed, catalyst-membrane system is required with better sealing and the ability to withstand the high operating pressure that drives the H2 permeation.
Similar content being viewed by others
References
X. Wang and R. J. Gorte, Appl. Cat. A: Gen., 247, 157 (2003).
T. Shido and Y. Iwasawa, J. Catal., 141, 71 (1993).
O. Goerke, P. Pfeifer and K. Schubert, Appl. Catal. A: Gen., 263, 11 (2004).
G. Jacobs, U. M. Graham, E. Chenu, P. M. Patterson, A. Dozier and B. H. Davis, J. Catal., 229, 499 (2005).
R. J. Gorte and S. Zhao, Catal. Today, 104, 18 (2005).
G. Barbieri, A. Brunetti, G. Tricoli and E. Drioli, J. Power Sources, 182, 160 (2008).
S. Battersby, M. C. Duke, S. Liu, V. Rudolph and J.C. Diniz da Costa, J. Memb. Sci., 316, 46 (2008).
T. A. Peters, M. Stange, H. Klette and R. Bredesen, J. Power Sources, 316, 119 (2008).
A. Brunetti, A. Caravella, G. Barbieri and E. Drioli, J. Memb. Sci., 306, 329 (2007).
O.U. Iyoha, H 2 production in palladium and palladium-copper membrane reactors at 1,173 K in the presence of H 2 S, Ph.D thesis, University of Pittsburgh (2007).
S.K. Ryi, J. S. Park, S. H. Kim, S.H. Cho, D.W. Kim and K.Y. Um, Sep. Purif. Technol., 50, 82 (2006).
T. Takeguchi, S. Manabe, R. Kikuchi, K. Eguchi, T. Kanazawa, S. Matsumoto and W. Ueda, Appl. Catal. A: Gen., 293, 91 (2005).
S.K. Ryi, J. S. Park, S. H. Kim, D.W. Kim and K. I. Cho, J. Memb. Sci., 318, 346 (2008).
S.K. Ryi, J. S. Park, S.H. Kim, S. C. Hong and D.W. Kim, Desalination, 200, 213 (2006).
S.K. Ryi, J. S. Park, S.H. Kim, S. C. Hong and D.W. Kim, Desalination, 200, 219 (2006).
S.K. Ryi, J. S. Park, S.H. Kim, S. C. Hong and D.W. Kim, Desalination, 200, 216 (2006).
S. K. Ryi, J. S. Park, S. H. Kim, D.W. Kim and J.W. Moon, J. Memb. Sci., 306, 261 (2007).
J.K. Ali, E. J. Newsom and D.W. T. Rippin, Chem. Eng. Sci., 49, 2129 (1994).
H. Amandusson, L.G. Ekedahl and H. Dannetun, Appl. Surf. Sci., 153, 259 (2000).
K. Hou and R. Hughes, J. Memb. Sci., 206, 119 (2002).
A. Unemoto, A. Kaimai, K. Sato, T. Otake, K. Yashiro, J. Mizusaki, T. Kawada, T. Tsuneki, Y. Shirasaki and I. Yasuda, Int. J. Hydro. Energy, 32, 2881 (2007).
F.C. Gielens, R. J. J. Knibbeler, P. F. J. Duysinx, H.D. Tong, M.A.G. Vorstman and J. T. F. Keurentjes, J. Memb. Sci., 279, 176 (2006).
S. Tosti, A. Adrover, A. Basile, V. Camilli, G. Chiappetta and V. Violante, Int. J. Hydro. Energy, 28, 105 (2003).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hwang, KR., Ihm, SK. & Park, Js. A catalytic membrane reactor for water-gas shift reaction. Korean J. Chem. Eng. 27, 816–821 (2010). https://doi.org/10.1007/s11814-010-0133-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-010-0133-7