Abstract
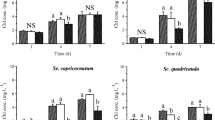
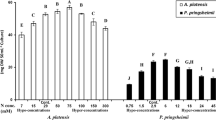
Uniconazole, as a plant growth retardant, can enhance stress tolerance in plants, possibly because of improved antioxidation defense mechanisms with higher activities of superoxide dismutase (SOD) and peroxidase (POD) enzymes that retard lipid peroxidation and membrane deterioration. These years much attention has been focused on the responses of antioxidant system in plants to uniconazole stress, but such studies on aquatic organism are very few. Moreover, no information is available on growth and antioxidant response in marine microalgae to uniconazole. In this paper, the growth and antioxidant responses of two marine microalgal species, Platymonas helgolandica and Pavlova viridis, at six uniconazole concentrations (0–15 mg L−1) were investigated. The results demonstrated that 3 mg L−1 uniconazole could increase significantly chlorophyll a and carbohydrate contents of P. helgolandica (P < 0.05). Higher concentrations (≥12 mg L−1) of uniconazole could inhibit significantly the growth, dry weight, chlorophyll-a and carbohydrate contents of P. helgolandica and P. viridis (P < 0.05). Uniconazole caused a significant increase in lipid peroxidation production (MDA) at higher concentrations (≥ 9 mg L−1). The activities of antioxidant enzymes, superoxide dismutase (SOD) and catalase (CAT) were enhanced remarkably at low concentrations of uniconazole. However, significant reduction of SOD and CAT activities was observed at higher concentrations of uniconazole.
Similar content being viewed by others
References
Allen, R. D., 1995. Dissection of oxidative stress tolerance using transgenic plants. Plant Physiology, 107: 1049–1054.
Campanella, L., Cubadda, F., Sammartino, M. P., and Saoncella, A., 2000. An algal biosensor for the monitoring of water toxicity in estuarine environments. Water Research, 25: 69–76.
Chen, D. Q., Li, Y. N., and Pen, C. L., 2000. The effect of S-3307 on growth characteristics and yield of soybean. Journal of Hubei Agricultural College, 20: 108–114.
Elisabetta, M., and Gioacchino, S., 2004. Copper-induced changes of non-protein thiols and antioxidant enzymes in the marine microalga Phaeodactylum tricornutum. Plant Science, 167: 289–296.
Halliwell, B., and Gutteridge, J. M. C., 1999. Free Radicals in Biology and Medicine. 3rd edition. Oxford University Press, New York, 936pp.
Heath, R. L., and Packer, L., 1968. Photoperoxidation in isolated chloroplasts I. Kinetics and stoichiometry of fatty acid peroxidation. Archives of Biochemistry and Biophysics, 125: 189–198.
Kang, M. C., Cha, S. H., Wijesinghe, W. A. J. P., Kang, S. M., Lee, S. H., Kim, E. A., Song, C. B., and Jeon, Y. J., 2013. Protective effect of marine algae phlorotannins against AAPH-induced oxidative stress in zebrafish embryo. Food Chemistry, 138: 950–955.
Kasai, F., Takamura, N., and Hatakeyama, S., 1993. Effects of smetryne on growth of various freshwater algal taxa. Environmental Pollution, 79: 77–83.
Li, M., Hu, C., Zhu, Q., Chen, L., Kong, Z., and Liu, Z., 2006. Copper and zinc induction of lipid peroxidation and effects on antioxidant enzyme activities in the microalga Pavlova viridis (Prymnesiophyceae). Chemosphere, 62: 565–572.
Li, Q. M., and Yang, W. Y., 2004. Effects of soaking seeds with uniconazole on several photosynthetic characters of maize seedlings. Plant Physiology Communications, 40: 31–33.
Meneely, J. P., Campbell, K., Greef, C., Lochhead, M. J., and Elliott, C. T., 2013. Development and validation of an ultrasensitive fluorescence planar waveguide biosensor for the detection of paralytic shellfish toxins in marine algae. Biosensors and Bioelectronics, 41: 691–697.
Okuda, T., Matsuda, Y., and Yamanaka, A., 1991. Abrupt increases in the level of hydrogen peroxide in leaves of winter wheat is caused by cold treatment. Plant Physiology, 97: 1265–1267.
Part, L., Botti, C., and Fichet, T., 2008. Effect of plant growth regulators on floral differentiation and seed production in jojoba (Simmondsia chinensis (Link) Schneider). Industrial Crops and Products, 27(1): 44–49.
Qiu, J., Wang, R. M., Yan, J. Z., and Hu, J., 2005. Seed film coating with uniconazole improves rape seedling growth in relation to physiological changes under waterlogging stress. Plant Growth Regulation, 47: 75–81.
Romanowska-Duda, Z., Tarczynska, M., and Zalewski, M., 2001. The control of cyanobacterial blooms by plant growth retardants (ancymidol, paclobutrazol, uniconazole). Water Science and Technology: Water Supply, 1: 247–250.
Sasaki, E., Ogura, T., Takei, K., Kojima, M., Kitahata, N., Sakakibara, H., Asami, T., and Shimada, Y., 2013. Uniconazole, a cytochrome P450 inhibitor, inhibits trans-zeatin biosynthesis in Arabidopsis. Phytochemistry, 87: 30–38.
Scandalios, J. G., 1993. Oxygen stress and superoxide dismutases. Plant Physioloy, 101: 7–12.
Van Dijk, H. F. G., and Roelofs, J. G. M., 1988. Effects of excessive ammonium deposition on the nutritional status and condition of pine needles. Physiologia Plantarum, 73: 494–501.
Wong, P. K., 2000. Effects of 2, 4-D, glyphosate and paraquat on growth, photosynthesis and chlorophyll a synthesis of Scenedesmus quadricauda Berb614. Chemosphere, 41: 177–182.
Wu, C. W., Sun, J. Q., Zhang, A. P., and Liu, W. P., 2013. Dissipation and enantioselective degradation of plant growth retardants paclobutrazol and uniconazole in open field, greenhouse, and laboratory soils. Environmental Science and Technology, 47: 843–849.
Yucesan, B., Turker, A. U., and Gurel, E., 2007. TDZ-induced high frequency plant regeneration through multiple shoot formation in witloof chicory (Cichorium intybus L.). Plant Cell, Tissue and Organ Culture, 99(3): 243–250.
Zhang, J., Cao, X. L., Yong, T. W., and Yang, W. Y., 2012. Seed treatment with uniconazole powder induced drought tolerance of soybean in relation to changes in photosynthesis and chlorophyll fluorescence. Research on Crops, 13(1): 147–154.
Zhang, M. C., Duan, L. S., Tian, X. L., He, Z. P., Li, J. M., Wang, B. M., and Li, Z. H., 2007. Uniconazole-induced tolerance of soybean to water deficit stress in relation to changes in photosynthesis, hormones and antioxidant system. Plant Physioloy, 164(6): 709–717.
Zhou, W. J., and Leul, M., 1998. Uniconazole-induced alleviation of freezing injury in relation to changes in hormonal balance, enzyme activities and lipid peroxidation in winter rape. Plant Growth Regulation, 26: 41–47.
Zhou, W. J., and Leul, M., 1999. Uniconazole-induced tolerance of rape plants to heat stress in relation to changes in hormonal levels, enzyme activities and lipid peroxidation. Plant Growth Regulation, 27: 99–104.
Zhou, W. J., and Ye, Q. F., 1996. Physiological and yield effects of uniconazole on winter rape (Brassica napus L.). Plant Growth Regulation, 15: 69–73.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mei, X., Zheng, K., Wang, L. et al. Studies on the effects on growth and antioxidant responses of two marine microalgal species to uniconazole. J. Ocean Univ. China 13, 877–882 (2014). https://doi.org/10.1007/s11802-014-2326-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11802-014-2326-x