Abstract
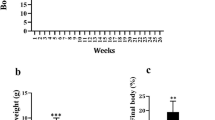
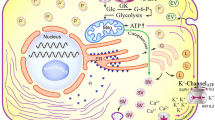
In the present study, the effects of β-escin, a central component of the horse chestnut (Aesculus hippocastanum L.) plant, were on oxidative stress, inflammation, and islet cells of Langerhans investigated in type 2 diabetes. In the experimental type 2 diabetes model, rats were first fed a high-fat diet (HFD) for ten weeks. Then, type 2 diabetes was induced by a double dose of 30 mg/kg streptozotocin injection at five-day intervals. The experimental animals were administered three different β-escin doses (5 mg/kg, 10 mg/kg, and 20 mg/kg) by gavage for four weeks after the type-2 diabetes was induced. Essential biochemical (insulin, glucose, ghrelin, cholesterol, triglyceride, and VLDL), inflammation (TNF-α, IFN-γ, IL-1α, IL-18, leptin, adiponectin), and oxidative stress (TAS, TOS, OSI) parameters were analyzed in the study samples. Furthermore, caspase three positive (cas3+) and insulin-positive (Ins+) cell rates were determined with immunohistochemical methods to identify the effects of β-escin on pancreatic tissue islet cells. The analysis revealed that administering various β-escin doses increased beta cell functions by inhibiting apoptosis in pancreatic islet cells and increasing insulin synthesis in rats with type 2 diabetes. Thus, it was concluded that β-escin exhibited antidiabetic activities since increased serum insulin levels and decreased blood glucose levels were observed. Furthermore, it was determined that inflammation was reduced with the decrease in glucotoxicity due to β-escin administration. The findings suggested that β-escin may be employed in treating diabetic complications.



Similar content being viewed by others
Data availability
Not applicable.
Change history
24 November 2022
A Correction to this paper has been published: https://doi.org/10.1007/s11756-022-01278-2
Abbreviations
- ANOVA:
-
Analysis of variance
- ATP:
-
Adenosine triphosphate
- Cas3+ :
-
Caspase 3 positive
- ELISA:
-
Enzyme-linked ımmunosorbent assay
- HFD:
-
High-Fat Diet
- HOMA-IR:
-
Homeostasis model assessment ınsulin resistant
- HOMA-β:
-
Homeostasis model assessment beta cell function
- IFN-γ:
-
Interferon gamma
- IL-10:
-
Interleukın-10
- IL-18:
-
Interleukın-18
- IL-1α:
-
Interleukın-1α
- IL-1β:
-
Interleukın -1β
- IL-6:
-
Interleukın-6
- Ins+ :
-
Insulin positive
- OSI:
-
Oxidative stres ındex
- RBP4:
-
Retinol binding protein-4
- STZ:
-
Streptozotocin
- TAS:
-
Total antioxidant status
- TNF-α:
-
Tumor necrosis factor-α
- TOS:
-
Total oxidant status
- VLDL:
-
Very low-density lipoprotein
References
Arnould T, Janssens D, Michiels C, Remacle J (1996) Effect of aescine on hypoxia-induced activation of human endothelial cells. Eur J Pharmacol 315(2):227–233. https://doi.org/10.1016/S0014-2999(96)00645-0
Avci G, Kucukkurt I, Kupeli Akkol E, Yeşilada E (2010) Effects of escin mixture from the seeds of Aesculus hippocastanum on obesity in mice fed a high fat diet. Pharm Biol 48(3):247–252. https://doi.org/10.3109/13880200903085466
Channar SP, Meghji KA, Baloch MS, Kashif, Abbas A et al (2019) The efficacy of aescin on diabetic nephropathy: A streptozotocin-induced diabetic rat model. Indo Am J Pharm Scı 06(05):10690–10694. https://doi.org/10.5281/zenodo.3228712
Domanski D, Zegrocka-Stendel O, Perzanowska A, Dutkiewicz M, Kowalewska M et al (2016) Molecular mechanism for cellular response to β-escin and ıts therapeutic ımplications. PLoS ONE 11(10):e0164365. https://doi.org/10.1371/journal.pone.0164365
Du Y, Song Y, Zhang L, Zhang M, Fu F (2016) Combined treatment with low dose prednisone and escin improves the anti-arthritic effect in experimental arthritis. Int Immunopharmacol 31:257–265. https://doi.org/10.1016/j.intimp.2016.01.006
Frick RW (2000) Three treatments for chronic venous insufficiency: escin, hydroxyethlrutoside, and Daflon. Angiology 51(3):197–205. https://doi.org/10.1177/000331970005100303
Furnes MW, Zhao CM, Chen D (2009) development of obesity is associated with ıncreased calories per meal rather than per day. a study of high-fat diet-ınduced obesity in young rats. Obes Surg 19(10):1430–1438. https://doi.org/10.1007/s11695-009-9863-1
Galic S, Oakhill JS, Steinberg GR (2010) Adipose tissue as an endocrine organ. Mol Cell Endocrinol 316(2):129–139. https://doi.org/10.1016/j.mce.2009.08.018
Geisler R, Dargel C, Hellweg T (2019) The biosurfactant β-aescin: a review on the physico-chemical properties and ıts ınteraction with lipid model membranes and Langmuir monolayers. Molecules 25(1):117. https://doi.org/10.3390/molecules25010117
Guillaume M, Padioleau F (1994) Veinotonic effect, vascular protection, antiinflammatory and free radical scavenging properties of horse chestnut extract. Arzneimittelforschung 44(1):25–35
Hazman O, Aksoy L, Büyükben A (2016) Effects of crocin on experimental obesity and type-2 diabetes. Turk J Med Sci 46(5):1593–1602. https://doi.org/10.3906/sag-1506-108
Hazman O, Celik S (2014) Effects of oral anti-diabetic agent sitagliptin on total antioxidant and oxidant status in rats with type 2 diabetes mellitus. J Appl Biol Sci 8(1):31–37. Retrieved from https://www.jabsonline.org/index.php/jabs/article/view/372. Accessed 14 Nov 2022.
Hazman O, Ovalı S (2015) Investigation of the anti-ınflammatory effects of safranal on high-fat diet and multiple low-dose streptozotocin ınduced type 2 diabetes rat model. Inflammation 38(3):1012–1019. https://doi.org/10.1007/s10753-014-0065-1
Hu JN, Zhu XM, Han LK, Saito M, Sun YS et al (2008) Anti-obesity effects of escins extracted from the seeds of Aesculus turbinata blume (Hippocastanaceae). Chem Pharm Bull (Tokyo) 56(1):12–16. https://doi.org/10.1248/cpb.56.12
Karabağ-Coban F, Bulduk İ, Liman R, Ciğerci İ, İnce S, Hazman Ö (2016) Oleuropein alleviates malathion-induced oxidative stress and DNA damage in rats. Toxicol Environ Chem 98(1):101–108. https://doi.org/10.1080/02772248.2015.1110156
Karabag-Coban F, Hazman O, Fatih Bozkurt M, Ince S (2017) Antioxidant status and anti-inflammatory effects of oleuropein in streptozotocin-induced diabetic nephropathy in rats. Eur J Med Plants 18(2):1–10. https://doi.org/10.9734/EJMP/2017/31953
Kucukkurt I, Ince S, Keles H, Küpeli Akkol E, Avci G et al (2010) Beneficial effects of Aesculus hippocastanum L. seed extract on the body’s own antioxidant defense system on subacute administration. J Ethnopharmacol 129(1):18–22. https://doi.org/10.1055/s-2007-967118
Li M, Lu C, Zhang L, Zhang J, Du Y et al (2015) Oral administration of escin ınhibits acute ınflammation and reduces ıntestinal mucosal ınjury in animal models. Evid Based Complement Alternat Med 503617. https://doi.org/10.1155/2015/503617
Matsuda H, Murakami T, Li Y, Yamahara J, Yoshikawa M (1998) Mode of action of escins Ia and IIa and E,Z-senegin II on glucose absorption in gastrointestinal tract. Bioorg Med Chem 6(7):1019–1023. https://doi.org/10.1016/s0968-0896(98)00054-6
Matthhews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF et al (1985) Homeostasis model assessment: insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28(7):412–419. https://doi.org/10.1007/BF00280883
Montopoli M, Froldi G, Comelli MC, Prosdocimi M, Caparrotta L (2007) Aescin protection of human vascular endothelial cells exposed to cobalt chloride mimicked hypoxia and inflammatory stimuli. Planta Med 73(3):285–288. https://doi.org/10.1055/s-2007-967118
Oschmann R, Biber A, Lang F, Stumpf H, Kunz K (1996) Pharmacokinetic of beta-escin after administration of various Aesculus extract containing formulations. Die Pharmazie (51):577–581
Shaikh S, Malik F, Uqaili AA, Kazi N, Rahim F et al (2020) Effect of aescin on β-cell physiology in fructose fed rat model. Ann Pakistan Inst Med Sci 16(1):15–19. https://doi.org/10.48036/apims.v16i1.265
Sirtori CR (2000) Aescin: pharmacology, pharmacokinetics and therapeutic profile. Pharmacol Res 44(3):183–193. https://doi.org/10.1006/phrs.2001.0847
Suryavanshi SV, Kulkarni YA (2021) Attenuation of cardiac autonomic neuropathy by escin in diabetic rats. Pharmacology 106(3–4):211–217. https://doi.org/10.1159/000509730
Suryavanshi SV, Kulkarni YA (2020) Escin alleviates peripheral neuropathy in streptozotocin induced diabetes in rats. Life Sci 254:117777. https://doi.org/10.1016/j.lfs.2020.117777
Tabata H, Hirayama J, Sowa R, Furuta H, Negoro T, Sanke T, Nanjo K (1992) Islet amyloid polypeptide (IAPP/amylin) causes insulin resistance in perfused rat hindlimb muscle. Diabetes Res Clin Pract 15(1):57–61. https://doi.org/10.1016/0168-8227(92)90068-3
Tokuyama T, Yagui K, Yamaguchi T, Huang CI, Kuramoto N, Shimada F, Miyazaki J, Horie H, Saito Y, Makino H, Kanatsuka A (1997) Expression of human islet amyloid polypeptide/amylin impairs insulin secretion in mouse pancreatic beta cells. Metab Clin Exp 46(9):1044–1051. https://doi.org/10.1016/s0026-0495(97)90276-6
Tokuyama Y, Kanatsuka A, Ohsawa H, Yamaguchi T, Makino H, Yoshida S, Nagase H, Inoue S (1991) Hypersecretion of islet amyloid polypeptide from pancreatic islets of ventromedial hypothalamic-lesioned rats and obese Zucker rats. Endocrinology 128(6):2739–2744. https://doi.org/10.1210/endo-128-6-2739
Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL (2003) Obesity is associated with macrophage accumulation in adipose tissue. J Clin Investig 112(12):1796–1808. https://doi.org/10.1172/JCI19246
Woods SC, Seeley RJ, Rushing PA, Seeley RJ, Rushing PA et al (2003) A controlled high-fat diet ınduces an obese syndrome in rats. J Nutr 133(4):1081–1087. https://doi.org/10.1093/jn/133.4.1081
Yoshikawa M, Harada E, Murakami T, Matsuda H, Wariishi N, Yamahara J, Murakami N, Kitagawa I, Escins-Ia (1994) Ib, IIa, IIb, and IIIa, bioactive triterpene oligoglycosides from the seeds of Aesculus hippocastanum L.: their inhibitory effects on ethanol absorption and hypoglycemic activity on glucose tolerance test. Chem Pharm Bull 42(6):1357–1359. https://doi.org/10.1248/cpb.42.1357
Zhang M, Lv XY, Li J, Xu ZG, Chen L (2008) The characterization of high-fat diet and multiple low-dose streptozotocin ınduced type 2 diabetes rat model. Exp Diabetes Res 704045:1–9. https://doi.org/10.1155/2008/704045
Zhang Z, Cao G, Sha L, Liu M (2015) The efficacy of sodium aescinate on cutaneous wound healing in diabetic rats. Inflammation 38(5):1942–1948. https://doi.org/10.1007/s10753-015-0174-5
Acknowledgements
This work was supported by Afyon Kocatepe University Scientific Research Projects Coordination Unit [Grant No. 17.FENED.11], Afyonkarahisar, Turkey. We thank Afyon Kocatepe University Scientific Research Projects Coordination Unit for supporting our work. In addition, some parameters (TAS, TOS and OSI levels) related to oxidative stress in the study were presented in a congress called “Uluslararası 29 Ekim Bilimsel Araştırmalar Sempozyumu”.
Author information
Authors and Affiliations
Contributions
Ömer Hazman; Methodology, Investigation, Writing-Reviewing and Editing; Zehra Betül Kumral; Applications to experimental animals and Biochemical analysis, Mehmet Savrık, Bobur Sındarov; Investigation, Writing-Original draft preparation and Biochemical analysis; Mehmet Fatih Bozkurt: Pathological analysis, Visualization, Data curation, Ahmet Büyükben: Writing and Editing.
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hazman, Ö., Bozkurt, M.F., Kumral, Z.B. et al. The effects of β-escin on inflammation, oxidative stress and Langerhans islet cells in high-fat diet and streptozotocin injection induced experimental type-2 diabetes model. Biologia 78, 549–561 (2023). https://doi.org/10.1007/s11756-022-01266-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-022-01266-6