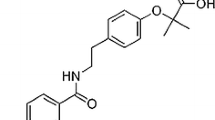
Abstract
[1-(2-isopropyl-5-methylphenoxy)-3-(yridine-3-ylmethylamino)propan-2-ol] is a novel oxipropanolamine derivative. It had been synthesized in a previous study with the idea that could be a prodrug and determined to show anti-α-glycosidase, antiacetylcholinesterase, anticarbonic anhydrase and antibacterial activities. However, to suggest it as a drug candidate, genotoxicity data related to it should be available. Thus, in this research, we aimed to evaluate its genotoxicity. We performed the chromosomal aberration and micronucleus tests in human peripheral lymphocytes. In all tests, lymphocyte cultures were treated with four concentrations (50, 25, 12.5, 6.25 μg/mL) of this derivative. According to our results, it significantly increased the chromosomal abnormalities at 25 and 50 μg/mL concentrations both 24 h and 48 h periods. Also, it significantly decreased the mitotic index at all concentrations. On the other hand, it significantly increased the micronucleus frequencies at 6.25, 12.5, and 25 μg/mL concentrations. In micronucleus test, no binucleate cells were detected at the highest concentration (50 μg/mL) and a total number of 4000 binucleate cells couldn’t be reached at the second-highest concentration (25 μg/mL). These results evaluated together, we can suggest that the test substance is cytotoxic and aneugenic at all used concentrations in human peripheral lymphocytes. Also, it’s clastogenic and genotoxic at high application concentrations.



Similar content being viewed by others
Abbreviations
- anti-AChE:
-
Anti-acetylcholinesterase
- anti-CA:
-
Anti-carbonic anhydrase
- BN:
-
Binucleated
- CA:
-
Chromosomal aberrations
- csb:
-
Chromosome breaks
- ctb:
-
Chromatid break
- cte:
-
Chromatid exchanges
- dc:
-
Dicentric chromosomes
- DMSO:
-
Dimethyl sulfoxide
- er:
-
Endoredublication
- f:
-
Fragment
- LC50:
-
50% lethal concentration
- μg/mL:
-
Microgram/ml
- MI:
-
Mitotic index
- MN:
-
Micronucleus
- NC:
-
Negative control
- PC:
-
Positive control
- SC:
-
Solvent control
- SCE:
-
Sister chromatid exchange
- scu:
-
Sister chromatid union
- SE:
-
Standard error
- TS:
-
Test substance
References
Araldi RP, Melo TCD, Mendes TB, Sa’Junior PL, Nozima BHN, Ito ET, Carvalho RF, Souza EB, Stocco R (2015) Using the comet and micronucleus assays genotoxicity studies: a review. Biomed Pharmacother 72:74–82. https://doi.org/10.1016/j.biopha.2015.04.004
Aruna N, Krishnamurthy NB (1986) Mutagenic evaluation of propranolol in somatic and germ cells of mice. Mutat Res Lett 173:207–210. https://doi.org/10.1016/0165-7992(86)90037-0
Aydın E, Türkez H (2014) In vitro cytotoxicity, genotoxicity and antioxidant potentials of thymol on human blood cells. J Essent Oil Res 26(2):133–140. https://doi.org/10.1080/10412905.2013.860411
Aydın S, Başaran AA, Başaran N (2005) The effects of thyme volatiles on the induction of DNA damage by the heterocyclic amine IQ and mitomycin C. Mutat Res/Genet Toxicol Environ Mutagen 581(1–2):43–53. https://doi.org/10.1016/j.mrgentox.2004.10.017
Azirak S, Rencuzogullari E (2008) The in vivo genotoxic effects of carvacrol and thymol in rat bone marrow cells. Environ Toxicol: An Int J 23(6):728–735. https://doi.org/10.1002/tox.20380
Bazylak G, Nagels LJ (2003) A novel potentiometric approach for detection of beta-adrenergics and beta-adrenolytics in high-performance liquid chromatography. II Farmaco 58(8):591–603. https://doi.org/10.1016/S0014-827X(03)00096-X
Belato KK, de Oliveira JR, de Oliveira FS, de Oliveira LD, Camargo SEA (2018) Cytotoxicity and genotoxicity of thymol verified in murine macrophages (RAW 264.7) after antimicrobial analysis in Candida albicans, Staphylococcus aureus and Streptococcus mutans. J Funct Foods 40:455–460. https://doi.org/10.1016/j.jff.2017.11.035
Brambilla G, Martelli A (2006) Genotoxicity and carcinogenicity studies of antihypertensive agents. Mutat Res/Rev Mutat Res 612(2):115–149. https://doi.org/10.1016/j.mrrev.2005.12.002
Buyukleyla M, Rencuzogullari E (2009) The effects of thymol on sister chromatid exchange chromosome aberration and micronucleus in human lymphocytes. Ecotoxicol Environ Saf 72(3):943–947. https://doi.org/10.1016/j.ecoenv.2008.10.005
Carre MC, Youlassani A, Caubere P (1984) Synthesis of a novel series of (aryloxy) propanolamines: new selective. Beta 2-blocking agents. J Med Chem 27(6):792–799. https://doi.org/10.1021/jm00372a016
Cecchetti V, Fravolini A, Schiaffella F, Tabarrini O, Bruni G, Segre G (1993) o-chlorobenzenesulfonamidic derivatives of (aryloxy) propanolamines as. beta.-blocking/diuretic agents. J Med Chem 36(1):157–161. https://doi.org/10.1021/jm00053a020
Chandirasekar R, Kumar BL, Sasikala K, Jayakumar R, Suresh K, Venkatesan R, Rachel J, Kavitha H, Ganesh GH (2014) Assessment of genotoxic and molecular mechanisms of cancer risk in smoking and smokeless tobacco users. Mutat Rese/Gene Toxicol Environ Mutagen 767:21–27. https://doi.org/10.1016/j.mrgentox.2014.04.007
Crowther AF, Howe R, McLoughlin BJ, Mallion KB, Rao BS, Smith LH, Turner RW (1972) Beta.-Adrenergic blocking agents. 12. Heterocyclic compounds related to propranolol. J Med Chem 15(3):260–266. https://doi.org/10.1021/jm00273a013
Epel D (1963) The effects of carbon monoxide inhibition on ATP level and the rate of mitosis in the sea urchin egg. J Cell Biol 17(2):315–319. https://doi.org/10.1083/jcb.17.2.315
Fei C, Zhang J, Lin Y, Wang X, Zhang K, Zhang L, Zheng W, Wang M, Li T, Xiao Z, Xue F, Wang C (2015) Safety evaluation of a triazine compound nitromezuril by assessing bacterial reverse mutation, sperm abnormalities, micronucleus and chromosomal aberration. Regul Toxicol Pharmacol 71:585–589. https://doi.org/10.1016/j.yrtph.2015.01.011
Giweli A, Džamić AM, Soković M, Ristić MS, Marin PD (2012) Antimicrobial and antioxidant activities of essential oils of Satureja thymbra growing wild in Libya. Molecules 17(5):4836–4850. https://doi.org/10.3390/molecules17054836
Hagmar L, Bonassi S, Stromberg U, Brøgger A, Knudsen LE, Norppa H, Reuterwall C (1998) Chromosomal aberrations in lymphocytes predict human cancer: a report from the European Study Group on Cytogenetic Biomarkers and Health (ESCH). Cancer Res 58:4117–4121
Imming P, Sinning C, Meyer A (2006) Drugs, their targets and the nature and number of drug targets. Nat Rev Drug Discov 5:821–834. https://doi.org/10.1038/nrd2132
Jaafari A, Tilaoui M, Mouse HA, M’bark LA, Aboufatima R, Chait A, Lepoivre M, Zyad A (2012) Comparative study of the antitumor effect of natural monoterpenes: relationship to cell cycle analysis. Rev Brasi Farmaco 22(3):534–540. https://doi.org/10.1590/S0102-695X2012005000021
Jyoti S, Siddique YH, Khan S, Naz F, Ali F (2015) Effect on micronucleus frequency and DNA damage in buccal epithelial cells of various factors among pan masala and gutkha chewers. Oral Sci Int 12:9–14. https://doi.org/10.1016/S1348-8643(14)00030-5
Kirkland D (2010) Evaluation of different cytotoxic and cytostatic measures for the in vitro micronucleus test (MNVit): summary of results in the collaborative trial. Mutat Res 702:139–147. https://doi.org/10.1016/j.mrgentox.2010.02.001
Kirsch-Volders M, Plas G, Elhajouji A, Lukamowicz M, Gonzalez L, Loock KV, Decordier I (2011) The in vitro MN assay in 2011: origin and fate, biological significance, protocols, high throughput methodologies and toxicological relevance. Arch Toxicol 85:873–899. https://doi.org/10.1007/s00204-011-0691-4
Kordali S, Cakir A, Ozer H, Cakmakci R, Kesdek M, Mete E (2008) Antifungal, phytotoxic and insecticidal properties of essential oil isolated from Turkish Origanum acutidens and its three components, carvacrol, thymol and p-cymene. Bioresource Technol 99(18):8788–8795. https://doi.org/10.1016/j.biortech.2008.04.048
Kumar D, Rawat DS (2013) Synthesis and antioxidant activity of thymol and carvacrol based Schiff bases. Bioorgan Med Chem Lett 23(3):641–645. https://doi.org/10.1016/j.bmcl.2012.12.001
Lambert RJW, Skandamis PN, Coote PJ, Nyshas GJ (2001) A study of the minimum inhibitory concentration and mode of action of oregano essential oil, thymol and carvacrol. J Appl Microbiol 91(3):453–462. https://doi.org/10.1046/j.1365-2672.2001.01428.x
LLana-Ruiz-Cabello M, Maisanaba S, Puerto M, Prieto AI, Pichardo S, Jos Á, Cameán AM (2014) Evaluation of the mutagenicity and genotoxic potential of carvacrol and thymol using the Ames Salmonella test and alkaline, Endo III-and FPG-modified comet assays with the human cell line Caco-2. Food Chem Toxicol 72:122–128. https://doi.org/10.1016/j.fct.2014.07.013
Machin PJ, Hurst DN, Bradshaw RM, Blaber LC, Burden DT, Melarange RA (1984) Beta 1 selective adrenoceptor antagonists. 3. 4-Azolyl linked phenoxypropanolamines. J Med Chem 27(4):503–509. https://doi.org/10.1021/jm00370a012
Maisanaba S, Prieto AI, Puerto M, Gutiérrez-Praena D, Demir E, Marcos R, Cameán AM (2015) In vitro genotoxicity testing of carvacrol and thymol using the micronucleus and mouse lymphoma assays, Mutat Res/Gene Toxicol Environ Mutagene. 784:37–44. https://doi.org/10.1016/j.mrgentox.2015.05.005
Martelli A, Allavena A, Sottofattori E, Brambilla G (1994) Low clastogenic activity in vivo of the N-nitroso derivatives of 5 β-adrenergic-blocking drugs proved to be potent genotoxins in vitro. Toxicol Lett 73(3):185–191. https://doi.org/10.1016/0378-4274(94)90057-4
Mauleón D, Pujol MD, Rosell G (1988) Beta-adrenergic antagonists: N-alkyl and N-amidoethyl (arylalkoxy) propanolamines related to propranolol. Eur J Med Chem 23(5):421–426. https://doi.org/10.1016/0223-5234(88)90138-9
Okine LKN, Ioannides C, Parke DV (1983) Studies on the possible mutagenicity of β-adrenergic blocker drugs. Toxicol Lett 16(3–4):167–174. https://doi.org/10.1016/0378-4274(83)90175-3
Pei RS, Zhou F, Ji BP, Xu J (2009) Evaluation of combined antibacterial effects of eugenol, cinnamaldehyde, thymol, and carvacrol against E. coli with an improved method. J Food Sci 74(7):379–383. https://doi.org/10.1111/j.1750-3841.2009.01287.x
Robbiano L, Martelli A, Allavena A, Mazzei M, Gazzaniga GM, Brambilla G (1991) Formation of the N-nitroso derivatives of six β-adrenergic-blocking agents and their genotoxic effects in rat and human hepatocytes. Cancer Res 51(9):2273–2279
Rokade YB, Sayyed RZ (2009) Naphthalene derivatives: a new range of antimicrobials with high therapeutic value. Rasayan J Chem 2(4): 972–980. RJC-499
Sabitha G, Arundhathi K, Sudhakar K, Sastry BS, Yadav JS (2010) A novel three-component one-pot reaction involving β-naphthol, aldehydes, and urea promoted by TMSCl/NaI. J Heterocycl Chem 47(2): 272–275. http://doi.org/123456789/7866
Sahoo CR, Paidesetty SK, Padhy RN (2021) The recent development of thymol derivative as a promising pharmacological scaffold. Drug Develop Res. https://doi.org/10.1002/ddr.21848
Sahu PK, Sahu PK, Thavaselvam D, Alafeefy AM, Agarwal DD (2015) Synthesis and evaluation of antimicrobial activity of 2-aminobenzothiazolomethyl naphthol derivatives. Med Chem Res 24(2):725–736. https://doi.org/10.1007/s00044-014-1150-6
Sedigh-Ardekani M, Saadat I, Saadat M (2013) Propranolol induced chromosomal aberrations in Chinese hamster ovary cell line. Mol Biol Res Commun 2(1–2):11–18. https://doi.org/10.22099/mbrc.2013.1460
Şen S, Beceren A, Aksoy H (2018) The importance of genotoxicity tests in new drug development process. J Hum Sci 15(3):1634–1649. https://doi.org/10.14687/jhs.v15i3.5400
Shettigar NB, Das S, Rao NB, Rao SB (2015) Thymol, a monoterpene phenolic derivative of cymene, abrogates mercury-induced oxidative stress resultant cytotoxicity and genotoxicity in hepatocarcinoma cells. EnvironToxicol 30(8):968–980. https://doi.org/10.1002/tox.21971
Tunca H, Berber AA, Çanakçi K, Tuna M, Yildiz SZ, Aksoy H (2017) Synthesis, characterization, and determination of genotoxic effect of a novel dimeric 8-hydroxyquinoline Cd (II) SCN complex. Drug Chem Toxicol 40(3):300–308. https://doi.org/10.1080/01480545.2016.1223094
Wechsler JB, Hsu CL, Bryce PJ (2014) IgE-mediated mast cell responses are inhibited by thymol-mediated, activation-induced cell death in skin inflammation. J Allergy Clin Immun 133(6):1735–1743. https://doi.org/10.1016/j.jaci.2013.12.024
Zengin M, Genc H, Taslimi P, Kestane A, Guclu E, Ogutlu A, Karabay O, Gulçin İ (2018) Novel thymol bearing oxypropanolamine derivatives as potent some metabolic enzyme inhibitors–their antidiabetic, anticholinergic and antibacterial potentials. Bioorg Chem 81:119–126. https://doi.org/10.1016/j.bioorg.2018.08.003
Acknowledgments
This research was approved by the Non-Invasive Research Ethics Committee of Sakarya University, Faculty of Medicine (03/01/2019-E.87). In addition, this study is performed according to the Declaration of Helsinki and an informed written consent form was obtained from donors who donated blood.
Funding
This study was supported by Sakarya University Scientific Research Projects Commission Presidency, Sakarya, Turkey (Project No: 2019-7-24-40).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declared no potential conflicts of interest concerning for to the research, authorship, and/or publication of this article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Meredova, G., Yıldız, E., Şen, S. et al. Genotoxicity of a novel thymol bearing oxipropanolamine derivative in human peripheral lymphocytes. Biologia 77, 559–567 (2022). https://doi.org/10.1007/s11756-021-00965-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-021-00965-w