Abstract
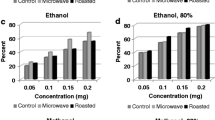
Eleven antioxidants including nine phenolic compounds (rutin, quercetin, hesperidin, hesperetin, naringin, naringenin, chlorogenic acid, caffeic acid, ferulic acid), vitamin E (α-tocopherol), and butylated hydroxytoluene (BHT) were selected to investigate their inhibitory effects on thermal oxidation of cholesterol in air and lard. The results indicated that the unoxidized cholesterol decreased with heating time whilst cholesterol oxidation products (COPs) increased with heating time. The major COPs produced were 7α-hydroxycholesterol, 7β-hydroxycholesterol, 5,6β-epoxycholesterol, 5,6α-epoxycholesterol, and 7-ketocholesterol. When cholesterol was heated in air for an hour, rutin, quercetin, chlorogenic acid, and caffeic acid showed a strong inhibitory effect. When cholesterol was heated in lard, caffeic acid, quercetin, and chlorogenic acid demonstrated inhibitory action during the initial 0.5 h (p < 0.05), with caffeic acid being the best inhibitor. Hesperetin, naringenin, caffeic acid, ferulic acid, vitamin E, and BHT could decrease the peroxide value during the initial 0.5 and 1 h (p < 0.05) in lard. It seemed that 200 ppm antioxidant could not obviously retard the long-term oxidation of cholesterol and lard under high temperature, but caffeic acid, quercetin, and chlorogenic acid displayed great untapped potential to prevent thermal oxidation of cholesterol 0.5 h at least. For the sake of health and flavor, fast stir-frying over a high flame is recommended. If baking or deep fat frying food in oil, it is best to limit cooking time to within 0.5 h.






Similar content being viewed by others
Abbreviations
- AAPH:
-
2,2-Azobis(2-amidinopropane) dihydrochloride
- ABTS:
-
2,2′-Azinobis(3-ethylbenzothiazoline-6-sulfonate)
- BHT:
-
Butylated hydroxytoluene
- COPs:
-
Cholesterol oxidation products
- DPPH:
-
2,2-Diphenyl-1-picrylhydrazyl radical
- FRAP:
-
Ferric reducing antioxidant power
- POV:
-
Peroxide value
- ORAC:
-
Oxygen radical absorbing capacity
- PCL:
-
Photochemiluminescence
- TBARS:
-
Thiobarbituric acid reactive substances
- TMS:
-
Trimethylsilyl
- TPTZ:
-
2,4,6-Tris(2-pyridyl)-s-triazine
References
Otaegui-Arrazola A, Menendez-Carreno M, Ansorena D, Astiasaran I (2010) Oxysterols: a world to explore. Food Chem Toxicol 48:3289–3303
Xu GH, Guan L, Sun JL, Chen ZY (2009) Oxidation of cholesterol and beta-sitosterol and prevention by natural antioxidants. J Agric Food Chem 57:9284–9292
Valenzuela A, Sanhueza J, Nieto S (2003) Cholesterol oxidation: health hazard and the role of antioxidants in prevention. Biol Chem 36:291–302
Boselli E, Rodriguez-Estrada MT, Ferioli F, Caboni MF, Lercker G (2010) Cholesterol photosensitised oxidation of horse meat slices stored under different packaging films. Meat Sci 85:500–505
Ansorena D, Barriuso B, Cardenia V, Astiasarán I, Lercker G, Rodriguez-Estrada MT (2013) Thermo-oxidation of cholesterol: effect of the unsaturation degree of the lipid matrix. Food Chem 141:2757–2764
Cardenia V, Rodriguez-Estrada MT, Boselli E, Lercker G (2013) Cholesterol photosensitized oxidation in food and biological systems. Biochimie 95:473–481
Savage GP, Dutta PC, Rodriguez-Estrada MT (2002) Cholesterol oxides: their occurrence and methods to prevent their generation in foods. Asia Pac J Clin Nutr 11:72–78
Sottero B, Gamba P, Gargiulo S, Leonarduzzi G, Poli G (2009) Cholesterol oxidation products and disease: an emerging topic of interest in medicinal chemistry. Curr Med Chem 16:685–705
Al Kanhal MA, Ahmad F, Al Othman AA, Zrif Z, Al Org S, Al Murshed KS (2002) Effect of pure and oxidized cholesterol-rich diets on some biochemical parameters in rats. Int J Food Sci Nutr 53:381–388
Guardiola F, Garcia-Cruset S, Bou R, Codony R (2004) Artifactual oxidation of cholesterol during the analysis of cholesterol oxidation products: protective effect of antioxidants. J AOAC Int 87:493–498
Lee HW, Chien JT, Chen BH (2008) Inhibition of cholesterol oxidation in marinated foods as affected by antioxidants during heating. Food Chem 108:234–244
Shozen K, Ohshima T, Ushio H, Takiguchi A, Koizumi C (1997) Effects of antioxidants and packing on cholesterol oxidation in processed anchovy during storage. Lebensm Wiss Technol 30:2–8
Guardiola F, Codony R, Rafecas M, Grau A, Jordan A, Boatella J (1997) Oxysterol formation in spray-dried egg processed and stored under various conditions: prevention and relationship with other quality parameters. J Agric Food Chem 45:2229–2243
Prior RL, Wu X, Schaich K (2005) Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J Agric Food Chem 53:4290–4302
Alvarez-Parrilla E, de la Rosa LA, Amarowicz R, Shahidi F (2012) Protective effect of fresh and processed Jalapeño and Serrano peppers against food lipid and human LDL cholesterol oxidation. Food Chem 133:827–834
Mariutti L, Nogueira GC, Bragagnolo N (2008) Optimization and validation of analytical conditions for cholesterol and cholesterol oxides extraction in chicken meat using response surface methodology. J Agric Food Chem 56:2913–2918
Zunin P, Boggia R, Evangelisti F (2001) Identification and quantification of cholesterol oxidation products in canned tuna. J Am Oil Chem Soc 78:1037–1040
Osada K, Kodama T, Yamada K, Sugano M (1993) Oxidation of cholesterol by heating. J Agric Food Chem 41:1198–1202
Shantha NC, Decker EA (1994) Rapid, sensitive, iron-based spectrophophotometric methods for determination of peroxide values of food lipids. J AOAC Int 77:421–424
Gorinstein S, Haruenkit R, Park YS, Jung ST, Zachwieja Z, Jastrzebski Z, Katrich E, Trakhtenberg S, Belloso OM (2004) Bioactive compounds and antioxidant potential in fresh and dried Jaffa® sweeties, a new kind of citrus fruit. J Sci Food Agric 84:1459–1463
Benzie IF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Anal Biochem 239:70–76
Wang H, Cao GH, Prior RL (1996) Total antioxidant capacity of fruits. J Agric Food Chem 44:701–705
Barriuso B, Otaegui-Arrazola A, Menéndez-Carreño M, Astiasarán I, Ansorena D (2012) Sterols heating: degradation and formation of their ring-structure polar oxidation products. Food Chem 135:706–712
Chien JT, Wang HC, Chen BH (1998) Kinetic model of the cholesterol oxidation during heating. J Agric Food Chem 46:2572–2577
Bartosz G (2014) Antioxidant and prooxidant activity of food components. Food oxidants and antioxidants-chemical, biological, and functional properties. CRC, Boca Raton, pp 375–432
Valenzuela A, Sanhueza J, Nieto S (2002) Effect of synthetic antioxidants on cholesterol stability during the thermal-induced oxidation of a polyunsaturated vegetable oil. J Am Oil Chem Soc 79:325–328
Wong D, Wang MF (2013) Inhibitory activities of some vitamins on the formation of cholesterol oxidation products in beef patties. J Agric Food Chem 61:8471–8476
Kmiecik D, Korczak J, Rudzinska M, Kobus-Cisowska J, Gramza-Michalowska A, Hes M (2011) β-Sitosterol and campesterol stabilisation by natural and synthetic antioxidants during heating. Food Chem 128:937–942
Acknowledgments
Project supported by the National Natural Science Foundation of China (31201304), Postdoctoral Science Foundation of China (2013M541787), and Postdoctoral Science Foundation of Zhejiang Province (BSH1301025).
Author information
Authors and Affiliations
Corresponding authors
About this article
Cite this article
Xu, G., Liu, D., Zhao, G. et al. Effect of Eleven Antioxidants in Inhibiting Thermal Oxidation of Cholesterol. J Am Oil Chem Soc 93, 215–225 (2016). https://doi.org/10.1007/s11746-015-2757-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11746-015-2757-6