Abstract
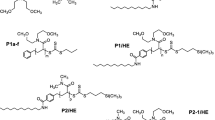
A series of methyl acrylate-acrylic acid amphiphilic triblock copolymers (PMA-PAA-PMA) were prepared by solution polymerization using S,S′-bis (α,α-dimethy1acetic acid) trithiocarbonate (BDAT) as a reversible addition fragmentation chain transfer (RAFT) agent and methyl acrylate (MA) as the first monomer. The triblock copolymers and their common MA homopolymer precursors were characterized in terms of their compositions, molecular weights and behavior at the air–water interface using 1H-NMR spectroscopy, thermogravimetric analysis, gel permeation chromatography, surface tension, transmission electron microscopy (TEM) and dynamic light scattering respectively. The results indicated that PMA-PAA-PMA was successfully synthesized through RAFT polymerization. The polydispersity index (PDI) decreased when the molar ratio [n(MA)/n(AA)] increased, the lowest PDI was obtained at 5.23 wt% RAFT and the molecular weights were consistent with the theoretical value as the RAFT agent percentage varied. The polymer neutralized by sodium hydroxide solution shows a low critical micelle concentration (CMC), which was <10−2 mol L−1 in water. The A min values increased and showed a maximum with decreased AA chain length. TEM showed that the neutralized polymer formed a special vesicle structure with large pore structure which led to a low CMC and surface tension of water.










Similar content being viewed by others
References
Alexandridis P, Lindman B (2000) Amphiphilic block copolymers: self-assembly and applications. Elsevier
Burguiere C, Pascual S, Bui C, Vairon JP, Charleux B et al. (2001) Block copolymers of poly(styrene) and poly(acrylic acid) of various molar masses, topologies, and compositions prepared via controlled/living radical polymerization. Application as stabilizers in emulsion polymerization. Macromolecules 34:4439–4450
Gaillard N, Guyot A, Claverie J (2003) Block copolymers of acrylic acid and butyl acrylate prepared by reversible addition-fragmentation chain transfer polymerization: synthesis, characterization, and use in emulsion polymerization. J Polym Sci Part A Polym Chem 41:684–698
Rodríguez-Hernández J, Lecommandoux S (2005) Reversible inside-out micellization of pH-responsive and water-soluble vesicles based on polypeptide diblock copolymers. J Am Chem Soc 127(7):2026–2027
Colombani O, Ruppel M, Schubert F, Zettl H, Pergushov DV, Mueller AHE (2007) Synthesis of poly(n-butyl acrylate)-block-poly(acrylic acid) diblock copolymers by ATRP and their micellization in water. Macromolecules 40:4338–4350
Discher BM, Won YY, Ege DS et al (1999) Polymersomes: tough vesicles made from diblock copolymers. Science 284(5417):1143–1146
Cheng GL, Boker A, Zhang MF, Krausch G, Muller AHE (2001) Amphiphilic cylindrical core-shell brushes via a “grafting from” process using ATRP. Macromolecules 34:6883–6888
Hawker CJ, Bosman AW, Harth E (2001) New polymer synthesis by nitroxide mediated living radical polymerizations. Chem Rev 101:3661–3688
Rizzardo E, Chiefari J, Chong BYK et al (1999) Tailored polymers by free radical processes. Macromol Symp 143(1):291–307
Colombani O, Ruppel M, Schubert F et al (2007) Synthesis of poly (n-butyl acrylate)-block-poly (acrylic acid) diblock copolymers by ATRP and their micellization in water. Macromolecules 40(12):4338–4350
Hoogenboom R, Schubert US, Van Camp W, Du Prez FE (2005) RAFT polymerization of 1-ethoxyethyl acrylate: a novel route toward near-monodisperse poly(acrylic acid) and derived block copolymer structures. Macromolecules 38:7653–7659
Zhou J, Zhang L, Ma J (2013) Fluorinated polyacrylate emulsifier-free emulsion mediated by poly(acrylic acid)-b-poly(hexafluorobutyl acrylate) trithiocarbonate via ab initio RAFT emulsion polymerization. Chem Eng J 223:8–17
Perrier S, Barner-Kowollik C, Quinn JF, Vana P, Davis TP (2002) Origin of inhibition effects in the reversible addition fragmentation chain transfer (RAFT) polymerization of methyl acrylate. Macromolecules 35:8300–8306
Toy AA, Vana P, Davis TP, Barner-Kowollik C (2004) Reversible addition fragmentation chain transfer (RAFT) polymerization of methyl acrylate: detailed structural investigation via coupled size exclusion chromatography-electrospray ionization mass spectrometry (SEC-ESI-MS). Macromolecules 37:744–751
Gaillard N, Guyot A, Claverie J (2003) Block copolymers of acrylic acid and butyl acrylate prepared by reversible addition-fragmentation chain transfer polymerization: synthesis, characterization, and use in emulsion polymerization. J Polym Sci Part A Polym Chem 41:684–698
Zhang S, Chen J, Taha M (2009) Synthesis of monodisperse styrene/methyl methacrylate/acrylic acid latex using surfactant-free emulsion copolymerization in air. J Appl Polym Sci 114:1598–1605
Zhang MF, Breiner T, Mori H, Muller AHE (2003) Amphiphilic cylindrical brushes with poly(acrylic acid) core and poly(n-butyl acrylate) shell and narrow length distribution. Polymer 44:1449–1458
Hautekeer JP, Varshney SK, Fayt R, Jacobs C, Jerome R, Teyssie P (1990) Anionic-polymerization of acrylic-monomers. 5. Synthesis, characterization, and modification of polystyrene poly(tert-butyl acrylate) diblock and triblock copolymers. Macromolecules 23:3893–3898
Li G, Shi L, An Y, Zhang W, Ma R (2006) Double-responsive core-shell-corona micelles from self-assembly of diblock copolymer of poly(t-butyl acrylate-co-acrylic acid)-b-poly (N-isopropylacrylamide). Polymer 47:4581–4587
Murayama S, Kuroda S, Osawa Z (1993) Hydrophobic and hydrophilic interpenetrating polymer networks (ipns) composed of polystyrene and poly(2-hydroxyethyl methacrylate). 2. Gradient composition in the ipns synthesized by photopolymerization. Polymer 34:3893–3898
Schoonbrood HAS, Unzue MJ, Beck OJ, Asua JM, Goni AM, Sherrington DC (1997) Reactive surfactants in heterophase polymerization. 7. Emulsion copolymerization mechanism involving three anionic polymerizable surfactants (surfmers) with styrene-butyl acrylate-acrylic acid. Macromolecules 30:6024–6033
Colombani O, Ruppel M, Burkhardt M, Drechsler M, Schumacher M et al (2007) Structure of micelles of poly(n-butyl acrylate)-block-poly (acrylic acid) diblock copolymers in aqueous solution. Macromolecules 40:4351–4362
Valette L, Massardier V, Pascault JP, Magny B (2002) Synthesis and photopolymerization of acrylic acrylate copolymers. J Appl Polym Sci 86:753–763
Goto A, Sato K, Tsujii Y et al (2001) Mechanism and kinetics of RAFT-based living radical polymerizations of styrene and methyl methacrylate. Macromolecules 34:402–408
Rosen MJ, Kunjappu JT (2012) Surfactants and interfacial phenomena. John Wiley & Sons
Alami E, Beinert G, Marie P, Zana R (1993) Alkanediyl-alpha, omega-bis (dimethylalkylammonium bromide) surfactants. 3. Behavior at the air–water-interface. Langmuir 9:1465–1467
Song LD, Rosen MJ (1996) Surface properties, micellization, and premicellar aggregation of gemini surfactants with rigid and flexible spacers. Langmuir 12:1149–1153
Lai JT, Filla D, Shea R (2002) Functional polymers from novel carboxyl-terminated trithiocarbonates as highly efficient RAFT agents. Macromolecules 35:6754–6756
Tanford C (1972) Micelle shape and size. J Phys Chem 76:3020–3024
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (21472235, 21464015, U1403192), the “One Hundred Talents” and “One Thousand Talents” Program (Y32H291501) of China and Returned overseas Young Talents Program (year 2014), Ministry of Human Resources and Social Security of the People’s Republic of China.
Author information
Authors and Affiliations
Corresponding authors
About this article
Cite this article
Yu, E., Zhang, L., Zhang, Y. et al. Synthesis of Gemini-Like Methyl Acrylate-Acrylic Acid-Methyl Acrylate Triblock Copolymers Surfactants by RAFT Polymerization in Solution and Investigation of their Behavior at the Air–Water Interface. J Surfact Deterg 18, 729–738 (2015). https://doi.org/10.1007/s11743-015-1708-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11743-015-1708-4