Abstract
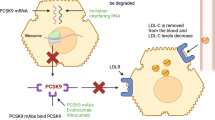
Statin-induced lowering of low-density lipoprotein cholesterol (LDL-C) reduces cardiovascular morbidity and mortality, but many patients do not adequately reduce their LDL-C levels. Monoclonal antibodies targeting PCKS9 are currently in the advanced phase of development. We aimed to investigate the efficacy and safety of PCSK9 inhibitors in patients at different cardiovascular risk in a systematic review. Studies were searched on MEDLINE and EMBASE until January 2016. Differences in the outcomes among groups were expressed as mean differences, or pooled odds ratio (OR) and corresponding 95% confidence interval (CI), which were calculated using a fixed-effects and a random-effects model. Statistical heterogeneity was evaluated using the I 2 statistic. 22 RCTs and 8833 patients were included. Six studies were performed in patients affected by homozygous or heterozygous familial hypercholesterolemia, or with increased cardiovascular risk, two in patients with statin intolerance, three in statin-naïve patients, and 10 in patients unable to achieve LDL-C target with statin therapy. PCSK9 inhibitors were associated with a statistically significant reduction of LDL-C (mean = −48.8%; 95% CI −54.1, −43.4; I 2 = 94%) compared to control groups, and with a statistically significant reduction in death for any cause (OR = 0.34; 95% CI 0.17, 0.69; I 2 = 0) and a favorable trend for cardiovascular events (OR = 0.79; 95% CI 0.61, 1.02; I 2 = 0%). PCSK9 inhibitors reduce LDL-C concentration in every group explored. A significant reduction in death by all cause was observed in the PCSK9 inhibitors groups, compared with control groups, even in the short time frame studied.



Similar content being viewed by others
References
Seidah NG, Benjannet S, Wickham L et al (2003) The secretory proprotein convertase neural apoptosis-regulated convertase 1 (NARC-1):liver regeneration and neural differentiation. Proc Natl Acad Sci USA 100:928–933
Abifadel M, Varret M, Rabés JP et al (2003) Mutation in PSCK9 cause autosomal dominant hypercholesterolemia. Nat Genet 34:154–156
Davignon J, Dubuc G, Seidah NG (2010) The influence of PCSK9 polymorphisms on serum low-density lipoprotein cholesterol and risk of atherosclerosis. Curr Atheroscler Rep 12:308–315
Cohen JC, Pertsemlidis A, Kotowski IK et al (2005) Low LDL cholesterol in individuals of African descent resulting from frequent nonsense mutations in PCSK9. Nat Genet 37:161–165
Cohen JC, Boerwinkle E, Mosley TH Jr et al (2006) Sequence variations in PCSK9, low LDL and protection against coronary heart disease. N Engl J Med 354:1264–1272
Benn M, Nordestgaard BG, Grande P et al (2010) PCSK9 R46L, low density lipoprotein cholesterol levels and risk of ischemic heart disease: 3 independent studies and meta analyses. J Am Coll Cardiol 55:2833–2842
Rashid S, Curtis DE, Garuti R et al (2005) Decreased plasma cholesterol and hypersensitivity to statins in mice lacking PCSK9. Proc Nat Acad Sci USA 102:5374–5379
Moher D, Liberati A, Tetzlaff J et al (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 151(264–269):W264
Higgins JP, Altman DG, Gotzsche PC et al (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. 343:d5928
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7:177–188
Higgins JP, Thompson SG, Deeks JJ et al (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560
Stein EA, Gipe D, Bergeron J, Gaudet D et al (2012) Effect of a monoclonal antibody to PCSK9, REGN727/SAR236553, to reduce low-density lipoprotein cholesterol in patients with heterozygous familial hypercholesterolaemia on stable statin dose with or without ezetimibe therapy: a phase 2 randomised controlled trial. Lancet 380:29–36
Hirayama A, Honarpour N, Yoshida M et al (2014) Effects of evolocumab (AMG 145), a monoclonal antibody to PCSK9, in hypercholesterolemic, statin-treated Japanese patients at high cardiovascular risk—primary results from the phase 2 YUKAWA Study. Circ J 78:1073–1082
Raal FJ, Honarpour N, Blom DJ et al (2015) Inhibition of PCSK9 with evolocumab in homozygous familial hypercholesterolaemia (TESLA Part B): a randomised, double-blind, placebo-controlled trial. Lancet 385:341–350
Raal F, Scott R, Somaratne R et al (2012) Low-density lipoprotein cholesterol-lowering effects of AMG 145, a monoclonal antibody to proprotein convertase subtilisin/kexin type 9 serine protease in patients with heterozygous familial hypercholesterolemia: the reduction of LDL-C With PCSK9 Inhibition in heterozygous familial Hypercholesterolemia disorder (RUTHERFORD) randomized trial. Circulation 126:2408–2417
Raal FJ, Stein EA, Dufour R et al (2015) PCSK9 inhibition with evolocumab (AMG 145) in heterozygous familial hypercholesterolaemia (RUTHERFORD-2): a randomised, double-blind, placebo-controlled trial. Lancet 385:331–340
Koren MJ, Giugliano RP, Raal FJ et al (2014) Efficacy and safety of longer-term administration of evolocumab (AMG 145) in patients with hypercholesterolemia 52-week results from the open-label study of long-term evaluation against LDL-C (OSLER) randomized trial. Circulation 129:234–243
Sabatine MS, Giugliano RP, Wiviott SD et al (2015) Efficacy and safety of evolocumab in reducing lipids and cardiovascular events. N Eng J Med. 372:1500–1509
Farnier M, Jones P, Severance R et al (2016) Efficacy and safety of adding alirocumab to rosuvastatin versus adding ezetimibe or doubling the rosuvastatin dose in high cardiovascular-risk patients: the ODYSSEY OPTIONS II randomized trial. Atherosclerosis 244:138–146
Bays H, Gaudet D, Weiss R et al (2015) Alirocumab as add-on to atorvastatin versus other lipid treatment strategies: ODYSSEY OPTIONS I randomized trial. J Clin Endocrinol Metab 100:3140–3148
Roth EM, Taskinen MR, Ginsberg HN et al (2014) Monotherapy with the PCSK9 inhibitor alirocumab versus ezetimibe in patients with hypercholesterolemia: results of a 24 week, double-blind, randomized, phase 3 study. Int J Cardiol 176:55–61
Robinson JG, Farnier M, Krempf M et al (2015) Efficacy and safety of alirocumab in reducing lipids and cardiovascular events. N Eng J Med. 372:1489–1499
Cannon CP, Cariou B, Blom D et al (2015) Efficacy and safety of alirocumab in high cardiovascular risk patients with inadequately controlled hypercholesterolaemia on maximally tolerated doses of statins: the ODYSSEY COMBO II randomized controlled trial. Eur Heart J 36:1186–1194
Kereiakes DJ, Robinson JG, Cannon CP et al (2015) Efficacy and safety of the proprotein convertase subtilisin/kexin type 9 inhibitor alirocumab among high cardiovascular risk patients on maximally tolerated statin therapy: the ODYSSEY COMBO I study. Am Heart J 169:906–915
Koren MJ, Scott R, Kim JB et al (2012) Efficacy, safety, and tolerability of a monoclonal antibody to proprotein convertase subtilisin/kexin type 9 as monotherapy in patients with hypercholesterolaemia (MENDEL): a randomised, double-blind, placebo-controlled, phase 2 study. Lancet 380:1995–2006
Koren MJ, Lundqvist P, Bolognese M et al (2014) Anti-PCSK9 monotherapy for hypercholesterolemia: The MENDEL-2 randomized, controlled, phase III clinical trial of evolocumab. J Am Coll Cardiol 23:2531–2540
Giugliano RP, Desai NR, Kohli P et al (2012) Efficacy, safety, and tolerability of a monoclonal antibody to proprotein convertase subtilisin/kexin type 9 in combination with a statin in patients with hypercholesterolaemia (LAPLACE-TIMI 57): a randomised, placebo-controlled, dose-ranging, phase 2 study. Lancet 380:2007–2017
Robinson JG, Nedergaard BS, Rogers WJ et al (2014) Effect of evolocumab or ezetimibe added to moderate or high-intensity statin therapy on LDL-C lowering in patients with hypercholesterolemia The LAPLACE-2 randomized clinical trial. JAMA 311:1870–1882
Sullivan D, Olsson AG, Scott R et al (2012) Effect of a monoclonal antibody to PCSK9 on low-density lipoprotein cholesterol levels in statin-intolerant patients the GAUSS randomized trial. JAMA 308:2497–2506
Stroes E, Colquhoun D, Sullivan D et al (2014) Anti-PCSK9 antibody effectively lowers cholesterol in patients with statin intolerance the GAUSS-2 randomized, placebo-controlled phase 3 trial of evolocumab. J Am Coll Cardiol 23:2541–2548
Blom DJ, Hala T, Bolognese M et al (2014) A 52-week placebo-controlled trial of evolocumab in hyperlipidemia. N Eng J Med 370:1809–1819
Ballantyne CM, Neutel J, Cropp A et al (2015) Results of bococizumab, a monoclonal antibody against proprotein convertase subtilisin/kexin type 9, from a randomized, placebo controlled, dose ranging study in statin-treated subject with hypercholesterolemia. Am J Cardiol 115:1212–1221
Roth EM, McKenney JM, Hanotin C et al (2012) Atorvastatin with or without an antibody to PCSK9 in Primary Hypercholesterolemia. N Eng J Med 367:1891–1900
Peng W, Peng W, Qian Z et al (2016) Therapeutic efficacy of PCSK9 monoclonal antibodies in statin-nonresponsive patients with hypercholesterolemia and dyslipidemia: a systematic review and meta-analysis. Atherosclerosis 252:50–60
Sattar N, Preiss D, Robinson GJ et al (2016) Lipid-lowering efficacy of PCSK9 inhibitor evolocumab (AMG145) in patients with type 2 diabetes: a meta-analysis of individual patient data. Lancet Diabetes Endocrinol 4:403–410
Zhang XL, Zhu QQ, Zhu L et al (2015) Safety and efficacy of anti-PCSK9 antibodies: a meta-analysis of 25 randomized, controlled trials. BMC Med 23(13):123
Li C, Lin L, Zhang W et al (2015) Efficiency and safety of proprotein convertase subtilisin/kexin 9 monoclonal antibody on hypercholesterolemia: a meta-analysis of 20 randomized controlled trials. J Am Heart Assoc 4:e001937
Navarese EP, Kolodziejczak M, Shulze V et al (2015) Effects of proprotein convertase subtilisin/kexin type 9 antibodies in adults with hypercholesterolemia: a systematic review and a meta-analysis. Ann Intern Med 2015(163):40–51
Stein EA, Giugliano RP, Koren MJ et al (2014) Efficacy and safety of evolocumab (AMG145), a fully human monoclonal antibody to PCSK9, in hyperlipidaemic patients on various background lipid therapies: pooled analysis of 1359 patients in four phase 2 trials. Eur Heart J 35:2249–2259
ClinicalTrial.gov NCT02207634 clinicaltrials.gov/ct2/show/NCT02207634
Ridker PM, Tardif JC, Amarenco P et al (2017) Lipid-reduction variability and antidrug-antibody formation with bococizumab. N Eng J Med 376:1517–1526
Moja L, Pecoraro V, Ciccolallo L, Dall’Olmo L, Virgili G, Garattini S (2014) Flaws, in animal studies exploring statins and impact on meta-analysis. Eur J Clin Invest 44:597–612
Friedrich JO, Adhikari NK, Beyene J (2008) The ratio of means method as an alternative to mean differences for analyzing continuous outcome variables in meta-analysis: a simulation study. BMC Med Res Methodol 8:32
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Giugliano reports grants and personal fees from Amgen, personal fees from Sanofi, personal fees from Regeneron, grants and personal fees from Merck, personal fees from Pfizer, personal fees from Bristol Myers Squibb, outside the submitted work. Nothing to declare from the other authors.
Statement of human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
None
Funding
No organization funded our paper.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Squizzato, A., Suter, M.B., Nerone, M. et al. PCSK9 inhibitors for treating dyslipidemia in patients at different cardiovascular risk: a systematic review and a meta-analysis. Intern Emerg Med 12, 1043–1053 (2017). https://doi.org/10.1007/s11739-017-1708-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11739-017-1708-7