Abstract
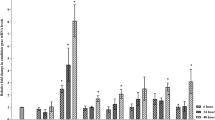
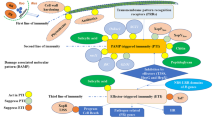
To evaluate the effectiveness of a germin-like protein (GLP) in legumes against the serious soil-borne pathogen Fusarium oxysporum f. sp. lentis, an Oryza sativa root-expressed GLP (OsRGLP1) was expressed in the model legume Medicago truncatula using the recombinant vector pCOsRGLP1. The transgene was highly expressed in M. truncatula transformed lines as assessed by RT-qPCR. Consistent with the active status of the transgene there was an elevated accumulation of H2O2 in transformed progeny. Enzymatic characterization of T1 transgenic progeny showed increased superoxide dismutase (SOD) activity. The additional SOD activity in transgenic lines was insensitive to potassium cyanide and sensitive to H2O2 indicating its resemblance to FeSOD. The effectiveness of the OsRGLP1 gene was tested by monitoring the root disease after infection of wild-type and transgenic lines. Wild-type plants were greatly affected by the pathogen infection showing a percent disease index value of 50 compared to 10–18 for the transgenic lines. The tolerance of the transgenic lines leads to recovery in fresh weight and pod production to an almost normal level. Analysis of defense-related genes downstream of hydrogen peroxide (H2O2) in transgenic plants showed induction of salicylic acid and jasmonate signaling pathways and increased expression of some pathogenesis-related-1 (PR-1) genes and a plant defensin gene. Overall, the findings suggest that OsRGLP1 provides protection against the fungal pathogen F. oxysporum that may involve the direct influence of H2O2 on signaling pathways leading to the activation of defense-related genes.






Similar content being viewed by others
References
Banerjee J, Maiti MK (2010) Functional role of rice germin-like protein1 in regulation of plant height and disease resistance. Biochem Biophys Res Commun 394(1):178–183
Barker DG, Bianchi S, Blondon F, Dattée Y, Duc G, Essad S et al (1990) Medicago truncatula, a model plant for studying the molecular genetics of the Rhizobium-legume symbiosis. Plant Mol Biol Report 8(1):40–49
Bundock P, Hooykaas PJ (1996) Integration of Agrobacterium tumefaciens T-DNA in the Saccharomyces cerevisiae genome by illegitimate recombination. Proc Natl Acad Sci 93(26):15272–15275
Chabaud M, de Carvalho-Niebel F, Barker D (2003) Efficient transformation of Medicago truncatula cv. Jemalong using the hypervirulent Agrobacterium tumefaciens strain AGL1. Plant Cell Rep 22(1):46–51
Chen X, Wang ML, Holbrook C, Culbreath A, Liang X, Brenneman T et al (2011) Identification and characterization of a multigene family encoding germin-like proteins in cultivated peanut (Arachis hypogaea L.). Plant Mol Biol Report 29(2):389–403
Cheng HY, Song SQ (2006) Species and organ diversity in the effects of hydrogen peroxide on superoxide dismutase activity in vitro. J Integ Plant Biol 48(6):672–678
Cheng Y, Zhang H, Yao J, Wang X, Xu J, Han Q et al (2012) Characterization of non-host resistance in broad bean to the wheat stripe rust pathogen. BMC Plant Biol 12(1):96
Christensen JH, Baucher M, O’Connell A, Van Montagu M, Boerjan W (2000) Control of lignin biosynthesis. In: Jain SM, Minocha S (eds) Molecular biology of woody plants, vol 1. Kluwer Academic Publishers, Dordrecht, pp 227–267
Christensen AB, Thordal-Christensen H, Zimmermann G, Gjetting T, Lyngkjær MF, Dudler R, Schweizer P (2004) The germinlike protein GLP4 exhibits superoxide dismutase activity and is an important component of quantitative resistance in wheat and barley. Mol Plant Microbe Interact 17(1):109–117
Dhindsa RS, Plumb-Dhindsa P, Thorpe TA (1981) Leaf senescence: correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J Exp Bot 32(1):93–101
Dunwell JM, Gibbings JG, Mahmood T, Naqvi SMS (2008) Germin and germin-like proteins: evolution, structure, and function. Crit Rev Plant Sci 27(5):342–375
Godfrey D, Able AJ, Dry IB (2007) Induction of a grapevine germin-like protein (VvGLP3) gene is closely linked to the site of Erysiphe necator infection: a possible role in defense? Mol Plant Microbe Interact 20(9):1112–1125
Hu X, Neill S, Cai W, Tang Z (2003) Hydrogen peroxide and jasmonic acid mediate oligogalacturonic acid-induced saponin accumulation in suspension-cultured cells of Panax ginseng. Physiol Plant 118(3):414–421
Knecht K, Seyffarth M, Desel C, Thurau T, Sherameti I, Lou B et al (2010) Expression of BvGLP-1 encoding a germin-like protein from sugar beet in Arabidopsis thaliana leads to resistance against phytopathogenic fungi. Mol Plant Microbe Interact 23(4):446–457
Lazo GR, Stein P, Ludwig RA (1991) A DNA transformation competent Arabidopsis genomic library in Agrobacterium. Nat Biotechnol 9(10):963–967
Le Henanff G, Heitz T, Mestre P, Mutterer J, Walter B, Chong J (2009) Characterization of Vitis vinifera NPR1 homologs involved in the regulation of pathogenesis-related gene expression. BMC Plant Biol 9:54
Lee SH, Shon YG, Kim CY, Chun HJ, Cheong YH, Kim ZH et al (1999) Variations in the morphology of rice plants regenerated from protoplasts using different culture procedures. Plant Cell Tiss Org 57(3):179–187
Legrand M, Kauffmann S, Geoffroy P, Fritig B (1987) Biological function of pathogenesis-related proteins: four tobacco pathogenesis-related proteins are chitinases. Proc Nat Acad Sci USA 84(19):6750–6754
Leon J, Lawton MA, Raskin I (1995) Hydrogen peroxide stimulates salicylic acid biosynthesis in tobacco. Plant Physiol 108(4):1673–1678
Levine A, Tenhaken R, Dixon R, Lamb C (1994) H2O2 from the oxidative burst orchestrates the plant hypersensitive disease resistance response. Cell 79(4):583–593
Liang H, Maynard CA, Allen RD, Powell WA (2001) Increased Septoria musiva resistance in transgenic hybrid poplar leaves expressing a wheat oxalate oxidase gene. Plant Mol Biol 45(6):619–629
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408
Murphy TM, Huerta AJ (1990) Hydrogen peroxide formation in cultured rose cells in response to UV-C radiation. Physiol Plant 78(2):247–253
Nolan KE, Irwanto RR, Rose RJ (2003) Auxin up-regulates MtSERK1 expression in both Medicago truncatula root-forming and embryogenic cultures. Plant Physiol 133(1):218–230
Nolan KE, SongY Liao S, Saeed NA, Zhang X, Rose RJ (2014) An unusual abscisic acid and gibberellic acid synergism increases somatic embryogenesis, facilitates its genetic analysis and improves transformation in Medicago truncatula. PLoS One 9:e99908
Park CJ, An JM, Shin YC, Kim KJ, Lee BJ, Paek KH (2004) Molecular characterization of pepper germin-like protein as the novel PR-16 family of pathogenesis-related proteins isolated during the resistance response to viral and bacterial infection. Planta 219(5):797–806
Piffanelli P, Zhou F, Casais C, Orme J, Jarosch B, Schaffrath U et al (2002) The barley MLO modulator of defense and cell death is responsive to biotic and abiotic stress stimuli. Plant Physiol 129(3):1076–1085
Ramírez-Suero M, Khanshour A, Martinez Y, Rickauer M (2010) A study on the susceptibility of the model legume plant Medicago truncatula to the soil-borne pathogen Fusarium oxysporum. Eur J Plant Pathol 126(4):517–530
Richards E, Reichardt M, Rogers S (1997) Preparation of plant DNA using CTAB Short protocols in molecular biology 3:2.10–2.11
Rietz S, Bernsdorff FE, Cai D (2012) Members of the germin-like protein family in Brassica napus are candidates for the initiation of an oxidative burst that impedes pathogenesis of Sclerotinia sclerotiorum. J Exp Bot 63(15):5507–5519
Rose RJ (2008) Medicago truncatula as a model for understanding plant interactions with other organisms, plant development and stress biology: past, present and future. Funct Plant Biol 35(4):253–264
Rose RJ, Nolan KE, Bicego L (1999) The development of the highly regenerable seed line Jemalong 2HA for transformation of Medicago truncatula implications for regenerability via somatic embryogenesis. J Plant Physiol 155(6):788–791
Scandalios JG (1993) Oxygen stress and superoxide dismutases. Plant Physiol 101(1):7
Song Y, Nolan KE, Rose RJ (2013) Stable transformation of Medicago truncatula cv. Jemalong for gene analysis using Agrobacterium tumefaciens. In: Rose RJ (ed) Legume genomics, methods and protocols, methods in molecular biology series 1069. Humana Press, Springer, New York, pp 203–214
Stoilova T, Chavdarov P (2006) Evaluation of lentil germplasm for disease resistance to Fusarium wilt (Fusarium oxysporum f. sp. lentis). J Central Eur Agric 7(1):121–126
Thordal-Christensen H, Zhang Z, Wei Y, Collinge DB (1997) Subcellular localization of H2O2 in plants. H2O2 accumulation in papillae and hypersensitive response during the barley powdery mildew interaction. Plant J 11(6):1187–1194
Tran PT, Choi H, Kim SB, Lee HA, Choi D, Kim KH (2014) A simple method for screening of plant NBS-LRR genes that confer a hypersensitive response to plant viruses and its application for screening candidate pepper genes against Pepper mottle virus. J Virol Methods 201:57–64
Turner JG, Ellis C, Devoto A (2002) The jasmonate signal pathway. Plant Cell 14(suppl 1):S153–S164
Van Loon LC, Rep M, Pieterse CMJ (2006) Significance of inducible defense-related proteins in infected plants. Annu Rev Phytopathol 44:135–162
Vaucheret H, Fagard M (2001) Transcriptional gene silencing in plants: targets, inducers and regulators. Trends Genet 17(1):29–35
Yasmin T, Mahmood T, Hyder MZ, Akbar S, Naqvi SMS (2008) Cloning, sequencing and in silico analysis of germin-like protein gene 1 promoter from Oryza sativa L. ssp. indica. Pak J Bot 40:1627–1634
Yasmin T, Mumtaz A, Mahmood T, Hyder MZ, Naqvi SMS (2015) A germin-like protein gene of rice increased superoxide dismutase activity in transformed tobacco. Biol Plant 59(3):456–462
Young ND, Debellé F, Oldroyd GE, Geurts R, Cannon SB, Udvardi MK, Benedito VA, Mayer KF, Gouzy J, Schoof H, Van de Peer Y et al (2011) The Medicago genome provides insight into the evolution of rhizobial symbioses. Nature 480(7378):520–524
Zimmermann G, Baumlein H, Mock HP, Himmelbach A, Schweizer P (2006) The multigene family encoding germin-like proteins of barley. Regulation and function in Basal host resistance. Plant Physiol 142(1):181–192
Acknowledgments
This work was financially supported by Higher Education Commission under Indigenous PhD 5000 Fellowship Program (No. 17-5-4(Ps4-279) and International Research Scholarship Initiative (No. IRSIP-21-BMS-47). The authors are grateful to Center for Application of Molecular Biology to International Agriculture, Australia for the generous gift of pCAMBIA vectors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by E. Kuzniak-Gebarowska.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sultana, T., Deeba, F., Naz, F. et al. Expression of a rice GLP in Medicago truncatula exerting pleiotropic effects on resistance against Fusarium oxysporum through enhancing FeSOD-like activity. Acta Physiol Plant 38, 255 (2016). https://doi.org/10.1007/s11738-016-2273-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-016-2273-9