Abstract
The role of robotic surgery in the curative-intent treatment of esophageal cancer patients is yet to be defined. To compare short-term outcomes between conventional minimally invasive (cMIE) and robot-assisted minimally invasive esophagectomy (RAMIE) in esophageal cancer patients. PubMed, Web of Science and Cochrane Library were systematically searched. The included studies compared short-term outcomes between cMIE and RAMIE. Individual risk of bias was calculated using the MINORS and RoB2 scales. There were no statistically significant differences between RAMIE and cMIE regarding conversion to open procedure, mean number of harvested lymph nodes in the mediastinum, abdomen and along the right recurrent laryngeal nerve (RLN), 30- and 90-day mortality rates, chyle leakage, RLN palsy as well as cardiac and infectious complication rates. Estimated blood loss (MD − 71.78 mL, p < 0.00001), total number of harvested lymph nodes (MD 2.18 nodes, p < 0.0001) and along the left RLN (MD 0.73 nodes, p = 0.03), pulmonary complications (RR 0.70, p = 0.001) and length of hospital stay (MD − 3.03 days, p < 0.0001) are outcomes that favored RAMIE. A significantly shorter operating time (MD 29.01 min, p = 0.004) and a lower rate of anastomotic leakage (RR 1.23, p = 0.0005) were seen in cMIE. RAMIE has indicated to be a safe and feasible alternative to cMIE, with a tendency towards superiority in blood loss, lymph node yield, pulmonary complications and length of hospital stay. There was significant heterogeneity among studies for some of the outcomes measured. Further studies are necessary to confirm these results and overcome current limitations.
Similar content being viewed by others
Introduction
Esophageal cancer is currently ranked seventh, globally, in terms of incidence, among cancer cases. It is also the sixth leading cause of cancer-related deaths, being responsible for 1 in every 18 cancer deaths in 2020. Histologically, the incidence of squamous cell carcinoma (SCC) has been in constant decline due to a change in economic gains, dietary regimes, and tobacco consumption—all major risk factors for esophageal cancer. Therefore, we are witnessing a shift in the histopathological subtypes, leading to an increase in the incidence of esophageal adenocarcinoma (AC) worldwide [1].
In spite of poor prognosis and long-term survival, the milestone of the disease’s primary management is currently set at radical esophagectomy and extended lymphadenectomy with previous eventual neoadjuvant chemoradiotherapy [2,3,4,5,6]. Its surgical approach underwent a paradigm change from open thoracotomy and laparotomy to conventional minimally invasive esophagectomy (cMIE), combining both thoracoscopy and laparoscopy [3]. This technique offers a decrease in total complication rates, wound infection rates, intraoperative blood loss, length of hospital stay, resulting in an improvement in both morbidity and mortality postoperatively [7,8,9,10,11]. Nonetheless, cMIE is not without limitations. Restricted movement of instruments, decreasing dexterity, prove to be technically complex and demanding, requiring a high number of patients to complete a surgeon’s learning curve [12, 13].
Robot-assisted minimally invasive esophagectomy (RAMIE) was first introduced in 2003 [14] and has since gradually proved to overcome some of cMIE’s limitations. Accounting for its articulated instruments, with 7 degrees of freedom, tremor filtering systems and improved magnification, precise dissection along narrower spaces is made simpler to surgeons [15, 16]. Whether these advantages represent better short- and long-term outcomes to esophageal cancer patients is still unclear.
Recently, several systematic reviews and meta-analyses reported RAMIE to be a safe and feasible alternative to cMIE in the treatment of esophageal cancer patients. RAMIE was associated with a tendency of longer operating time, less estimated blood loss and shorter length of hospital stay. RAMIE also yielded a larger number of lymph nodes and had lower rates of pulmonary complications when compared to cMIE [17,18,19,20,21,22,23,24].
Therefore, with the aim of analyzing RAMIE’s current contribution to the surgical approach of esophageal cancer patients, we conducted this systematic review and meta-analysis, assessing recent observational clinical studies (OCS) and randomized controlled trials (RCT), comparing short-term outcomes associated with RAMIE and cMIE. As robotic systems become more and more available worldwide and surgeons adhere to these, their technique upgrades over time. It becomes imperative to analyze the most recent data regarding this topic, as it is in constant evolution and rapidly changes as systems become more complex.
Materials and methods
This systematic review and meta-analysis followed the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) Statement guidelines and was prospectively registered on PROSPERO (Prospective Register of Systematic Reviews) under registration No. CRD42023466345.
Literature search
Two reviewers independently conducted a literature search on the following online databases: PubMed (MEDLINE), Web of Science and Cochrane Library. The search query terms that were chosen to be used on PubMed were: (((laparoscopic esophagectomy) OR (minimally invasive esophagectomy) OR (video assisted esophagectomy) OR (thoracoscopic esophagectomy) OR ((esophagectomy[MeSH Terms]) AND ((laparoscopy[MeSH Terms]) OR (minimally invasive surgical procedures[MeSH Terms]) OR (surgery, video assisted[MeSH Terms]) OR (surgery, thoracoscopic[MeSH Terms]) OR (surgery, video assisted thoracoscopic[MeSH Terms])))) OR ((robotic esophagectomy) OR ((esophagectomy[MeSH Terms]) AND (robotics[MeSH Terms])))) AND (esophagus neoplasms[MeSH Terms]). Under the Web of Science database, the following query was used: [ALL = (laparoscopic esophagectomy) OR ALL = (minimally invasive esophagectomy) OR ALL = (video assisted esophagectomy) OR ALL = (thoracoscopic esophagectomy) OR ALL = (robotic esophagectomy)] AND [ALL = (esophagus cancer) OR ALL = (esophagus neoplasms)]. On Cochrane Library, the search entry was: laparoscopic esophagectomy in All Text OR minimally invasive esophagectomy in All Text OR thoracoscopic esophagectomy in All Text OR robotic esophagectomy in All Text AND esophagus neoplasms in All Text. Previous systematic reviews and meta-analysis were also consulted to identify additional studies of interest.
Eligibility criteria
After the search results were exported, duplicated studies were detected and excluded. The resulting articles were assessed based on their titles and abstracts. Observational clinical studies and randomized controlled trials were deemed as relevant for inclusion when perioperative and short-term outcomes of robot-assisted and conventional minimally invasive curative-intent esophagectomies for resectable esophageal cancer patients were compared.
When available, full text articles were obtained and reviewed independently by two authors. Any discrepancies in selection were resolved by consensus. Studies were excluded according to the following criteria: (1) articles whose surgical technique did not include a robot-assisted thoracic phase esophagectomy; (2) comparison to a hybrid approach, instead of a conventional minimally invasive one; (3) studies that only assessed long-term outcomes; (4) absence of a comparison with a conventional minimally invasive technique. Indexed abstract posters and presentations, editorials, comments, and letters were also excluded.
Data extraction
Eligible studies were assessed for data extraction by two independent reviewers. The following information was gathered from each study: author, publication year, country, study design and period, occurrence of propensity score matching (PSM), sample size, surgical procedure, histopathological tumor characteristics, neoadjuvant therapy prevalence, demographic data (age and sex) and short-term outcomes. Short-term outcomes were chosen according to previous systematic reviews: surgical outcomes—operating time, estimated blood loss, conversion to open procedure rate, harvested lymph nodes [total, mediastinal, abdominal, and along the left and right recurrent laryngeal nerve (RLN)] and 30- and 90-day mortality rates; and postoperative outcomes—anastomotic leakage, chyle leakage, recurrent laryngeal nerve palsy, pulmonary, cardiac and infectious complications and length of hospital stay (Tables 1, 2).
Quality assessment
The risk of bias and study quality assessment was performed using the MINORS (Methodological Index for Non-Randomized Studies) scale [25] in the observational clinical studies eligible for analysis. As for randomized controlled trials, the Cochrane Risk of Bias 2 tool (RoB2) [26] was used. It was conducted by two authors, and any disagreements were solved through discussion. MINORS scoring system comprises 12 items, 8 methodological and 4 additional criteria applied to comparative studies. In regards to methodology, studies were scored 0 (not reported), 1 (reported but inadequate) or 2 (reported and adequate) points, according to the description of a clearly stated aim, inclusion of consecutive patients, involvement of a prospective collection of data, existence of endpoints appropriate to the aim of the study, performance of an unbiased assessment of the study endpoint, admittance of a follow-up period appropriate to the aim of the study, registration of a loss to follow-up below 5% and a prospective calculation of the study size. The 4 additional criteria applied to comparative studies used the same scoring system, assessing the existence of an adequate and contemporary control group, without confounding factors, and the presence of an adequate statistical analysis (Table 1 and Supplementary Table 1).
Statistical analysis
The meta-analysis was conducted using Review Manager (Version 5.4.1).
Results related to continuous outcomes were presented as mean differences (MD) with 95% confidence intervals (CI), by using the generic inverse variance method. Regarding dichotomous outcomes, results were presented as risk ratios (RR) with 95% CI, also using the generic inverse variance method. There were studies that reported their outcomes as median and range. In these cases, mean and standard deviation were estimated using a method described by Wan et al. [27]. Statistical significance was assessed using an alpha (α) level of 0.05. Heterogeneity was explored using the I2 measure and Cochran’s Q test, using a significance cutoff point of 0.10. We applied a random effects model due to the clinical heterogeneity of the included studies. PSM data were also analyzed to eliminate possible causes of heterogeneity. Publication bias among included studies was investigated by using funnel plots, provided as Supplementary File 1.
Results
Study selection
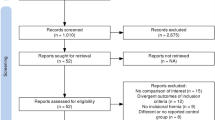
The conducted literature search of PubMed, Web of Science and Cochrane Library resulted in 4567 articles selected for review. Duplicated studies were excluded, resulting in 3003 records whose titles and abstracts were screened. Upon review, 72 full-text articles were assessed for eligibility. From these, 37 studies were excluded according to inclusion and exclusion criteria. Previous systematic reviews yielded 3 additional studies after manual screening of relevant references. Therefore, 35 full-text studies were included for quality assessment and analysis (Fig. 1). Demographic data and details from the included studies are presented in Table 1. OCS were more prevalent, with 34 results [28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61], and 1 RCT [62] was also included. A total of 18,187 participants were analyzed, of which 5205 underwent RAMIE, and 12,982 were operated under cMIE.
Quality assessment
All included studies revealed a fair methodological quality. The median score in the MINORS scale for OCS was 21, ranging from 19 to 22 points. As for the included RCT, the RoB2 tool calculated a low risk of bias judgement (Table 1 and Supplementary Table 1).
Surgical outcomes
Operating time
The conducted meta-analysis gathered 26 studies which reported each operating time. The test for overall effect concluded that cMIE has a significantly shorter operating time when compared to RAMIE (MD 29.01, p = 0.004 [95% CI 9.37, 48.66], I2 = 97%, p < 0.00001). Mean operating time was 395.72 min in the cMIE group and 424.91 min in the RAMIE group (Fig. 2).
Estimated blood loss
Estimated blood loss was assessed and measured in 25 studies. It has appeared to be significantly less in RAMIE when compared to cMIE (MD − 71.78, p < 0.00001 [95% CI − 96.24, − 47.33], I2 = 97%, p < 0.00001). Mean estimated blood loss was 209.68 mL in the RAMIE group and 387.18 mL in the cMIE group (Fig. 2).
Conversion to open procedure rate
This outcome was included in 18 studies and did not show any statistically significant difference between the two groups (RR 0.64, p = 0.14 [95% CI 0.36, 1.16], I2 = 80%, p < 0.00001). Conversion to open procedure had a rate of 4.87% (197/4047) in the RAMIE group and of 8.22% (857/10431) in the cMIE group.
Harvested lymph nodes, TOTAL
The total number of harvested lymph nodes during the procedures was recorded and presented in 25 studies. Mean total number of harvested lymph nodes in the RAMIE group was 28.89 and 26.61 in the cMIE group. Their difference was statistically significant, favoring RAMIE (MD 2.18, p < 0.0001 [95% CI 1.15, 3.21], I2 = 78%, p < 0.00001) (Fig. 2).
Harvested lymph nodes, MEDIASTINAL
The number of mediastinal harvested lymph nodes was measured in 13 studies. There was no statistically significant difference between the two groups (MD − 0.76, p = 0.46 [95% CI − 2.77, 1.25], I2 = 91%, p < 0.00001). Mean number of mediastinal harvested lymph nodes was 18.72 in the RAMIE group and 19.61 in the cMIE group.
Harvested lymph nodes, ABDOMINAL
This outcome was reported in 10 studies. The mean number of abdominal harvested lymph nodes was 9.38 in the RAMIE group and 8.85 in the cMIE group, showing no statistically significant difference between these groups (MD 0.13, p = 0.66 [95% CI − 0.47, 0.73], I2 = 58%, p = 0.01).
Harvested lymph nodes, LEFT RLN
Harvested lymph nodes along the left RLN were assessed in 12 studies. The mean yield of these nodes was 3.56 in the RAMIE group and 2.85 in the cMIE group, favoring RAMIE (MD 0.73, p = 0.03 [95% CI 0.06, 1.39], I2 = 93%, p < 0.00001) (Fig. 2).
Harvested lymph nodes, RIGHT RLN
As for the harvested lymph nodes along the right RLN, these were included in 9 articles, without evidence of a statistically significant difference between the two groups (MD 0.12, p = 0.53 [95% CI − 0.26, 0.51], I2 = 86%, p < 0.00001). Mean yield of these nodes was of 2.23 in the RAMIE group and of 2.12 in the cMIE group.
30-day mortality rate
Twenty studies reported their 30-day mortality rate for each procedure, being 1.63% (44/2707) in the RAMIE group and 1.87% (117/6244) in the cMIE group. There was no statistically significant difference between the two groups (RR 1.03, p = 0.88 [95% CI 0.73, 1.44], I2 = 0%, p = 0.53).
90-Day Mortality rate
This outcome was measured in 18 studies. The rate of 90-day mortality was 3.55% (106/2987) in the RAMIE group and 4.84% (336/6946) in the cMIE group, showing no statistically significant difference between the two groups (RR 0.95, p = 0.66 [95% CI 0.77, 1.18], I2 = 0%, p = 0.93).
Postoperative outcomes
Anastomotic leakage
Anastomotic leakage was measured and recorded in 28 studies. The rate of this complication was 12.47% (391/3136) in the RAMIE group and 11.43% (785/6866) in the cMIE group, favoring cMIE (RR 1.23, p = 0.0005 [95% CI 1.09, 1.38], I2 = 0%, p = 0.64) (Fig. 3).
Chyle leakage
Nineteen studies reported this outcome. There was no statistically significant difference between the two groups (RR 1.07, p = 0.74 [95% CI 0.72, 1.60], I2 = 13%, p = 0.30). The rate of chyle leakage was 2.82% (69/2443) in the RAMIE group and 3.84% (197/5135) in the cMIE group.
Recurrent laryngeal nerve palsy
RLN palsy was included in 23 studies. The rate of this event was 8.94% (237/2652) in the RAMIE group and 7.63% (423/5541) in the cMIE group, with no statistically significant difference between the two (RR 0.96, p = 0.62 [95% CI 0.82, 1.13], I2 = 7%, p = 0.36).
Pulmonary complications
Pulmonary complications were assessed in 29 studies. The rate of this complication was 20.13% (641/3185) in the RAMIE group and 22.20% (1547/6969) in the cMIE group. There was no statistically significant difference between the two groups (RR 0.89, p = 0.10 [95% CI 0.77, 1.02], I2 = 18%, p = 0.20) (Fig. 3).
Cardiac complications
Cardiac complications were included in 20 studies. The rate of this complication was 14.02% (365/2604) in the RAMIE group and 15.74% (823/5228) in the cMIE group. There was no statistically significant difference between the two groups (RR 1.01, p = 0.88 [95% CI 0.86, 1.19], I2 = 3%, p = 0.42).
Infectious complications
Seventeen studies reported infectious complications. The rate of this event was 2.53% (31/1223) in the RAMIE group and 3.18% (50/1572) in the cMIE group. These results showed no statistically significant difference between them (RR 0.97, p = 0.92 [95% CI 0.61, 1.56], I2 = 0%, p = 0.52).
Length of hospital stay
This outcome was included in 26 studies. The mean length of hospital stay was of 18.57 days in the RAMIE group and 33.11 days in the cMIE group. Meta-analysis favors RAMIE (MD − 3.03, p < 0.0001 [95% CI − 4.51, − 1.54], I2 = 96%, p < 0.00001) (Fig. 3).
Propensity Score Matching Analysis
Propensity Score Matching Analysis information is gathered under Table 3. It was not possible to conduct a meta-analysis for the 30-Day Mortality rate outcome, as only one event was recorded in the cMIE group, in one of the six included studies. RAMIE showed superiority over cMIE in estimated blood loss, total number of harvested lymph nodes along the left RLN, and over pulmonary complications. Regarding operating time, cMIE was able to be executed under a lower amount of time. The rest of the measured outcomes presented no statistically significant differences between the two groups.
Discussion
This systematic review and meta-analysis directly compares relevant short-term outcomes between robot-assisted and conventional minimally invasive esophagectomies in esophageal cancer patients. Previous meta-analysis [17,18,19,20,21,22,23,24] concluded that RAMIE is a superior surgical approach when compared to cMIE regarding blood loss, number of harvested lymph nodes, RLN palsy and pulmonary complications, while operating time is shorter in cMIE. Nonetheless, some of these results are also associated with a high percentage of heterogeneity among the studies, compromising their validity. With the publication of further data and literature, it became necessary to perform an updated meta-analysis to not only confirm the safety and feasibility of RAMIE, but also to establish new conclusions when compared to cMIE.
Our analysis concluded that both surgical approaches result in similar rates of conversion to open procedure, mean number of harvested lymph nodes in the mediastinum, abdomen and along the right RLN, 30- and 90-day mortality rates, chyle leakage, RLN palsy as well as cardiac and infectious complication rates.
A significantly shorter operating time and a lower rate of anastomotic leakage is seen in cMIE, while estimated blood loss, total number of harvested lymph nodes and along the left RLN, pulmonary complications and length of hospital stay are outcomes that favor RAMIE.
Surgical outcomes
Operating time
The mean operating time revealed to be around 29 min shorter in the cMIE group, when compared to RAMIE. This result is coherent with three previous meta-analysis [17, 18, 21]. There are two main reasons that explain a longer duration of surgical procedures with RAMIE.
Firstly, port setup, docking and repositioning of instruments and uninstallation of devices all account for extra time in an inexperienced surgical team. Secondly, most of the reviewed studies reported their data with surgeons that had not yet completed their learning curve in this specific type of surgery. Recent studies suggest that this learning curve is achieved in surgeons after 20–22 cases of successful RAMIE, with a previously vast experience in cMIE, attaining proficiency, lessening complications, and reducing operating time [59, 60].
Upon examination of our forest plot, we conclude that three of the included studies demonstrate that the robotic surgery group operated in a significantly shorter timeframe [42, 57, 62]. This is due to a docking time of only around 5 min in each of these scenarios and a fully experienced surgical team, both in assistants that perform equally in RAMIE and cMIE and also surgeons that have completed their learning curve. Therefore, it has been proven that once surgeons attain their proficiency with the equipment, RAMIE offers advantages in the manipulation of instruments and camera control that enable increased efficiency in the operation [62].
Analysis of PSM data from studies that performed this procedure leads us to a similar conclusion. The mean difference was larger, achieving over 41 min, favoring cMIE. Again, one of these studies [42] had statistically significant shorter operating times favoring RAMIE, explained by the same reasons.
Estimated blood loss
Our analysis suggests that RAMIE is associated with a reduction in estimated blood lost during the surgical procedures. The difference of blood loss between the two approaches is approximately 72 mL, favoring RAMIE, whose clinical significance may be residual. Previous meta-analyses present the same conclusion [17,18,19]. This result accounts for a majority of included studies that show no statistically significant difference between the two groups, but with several outliers that describe major complications of episodical events in the cMIE group. Therefore, this conclusion is to be taken carefully, taking these factors into consideration and an elevated heterogeneity among studies.
Nonetheless, this difference may be explained due to RAMIE’s improved instrument dexterity navigating in the surgical field and its tremor filtering systems. Lymph node dissection is made easier, damaging less blood vessels along the procedure [19].
PSM data suggest an even smaller, but still significant difference. The mean difference between the two procedures is approximately 25 mL. Such a small amount of blood lost might not be clinically relevant to the patient’s morbidity perioperatively.
Conversion to open procedure rate
This outcome showed no statistically significant difference between RAMIE and cMIE. Three of the included studies had a higher rate of conversion to open procedure [40, 42, 43]. These were explained by all the advantages that the robotic system has to offer in attaining proficiency in the procedures. Aside from these studies, all others had comparable results, enhancing the need for further data on this topic.
Upon analysis of PSM information, the same conclusion is achieved. A wider population needs to be assessed in order to have more information regarding conversion.
Harvested lymph nodes, TOTAL, MEDIASTINAL, ABDOMINAL, LRLN, RRLN
Lymphadenectomy has become an essential element of the primary management of esophagectomies. Due to a narrow operative field, lymph node dissection is especially challenging along each of the RLNs, being the surgeon’s intent to harvest as many lymph nodes as possible, without damaging the RLN or adjacent structures. This procedure is important to both categorize the tumor according to the TNM classification, but also apply curative-intent treatment [19].
Harvested lymph nodes had statistically significant results in total numbers and in those that were yielded along the left RLN, both favoring the RAMIE group. Previous meta-analyses discuss similar findings [19, 23, 24]. Nonetheless, our results present high heterogeneity among studies, which leads to questions regarding the external validity of these findings.
The robotic group was favored in 8 of the 25 included studies when total lymph nodes dissected were recorded. The mean difference between the groups was of approximately two additional lymph nodes. Along the left RLN region, our search included 12 studies. Results also favored RAMIE, with five of these that individually favored the robotic approach. RAMIE yielded approximately one additional lymph node over cMIE. Lymph node count that were dissected in the mediastinum, abdomen, and along the right RLN did not show differences between the two groups. Improved anatomical dissection precision, featured in RAMIE, accounts for a larger number of lymph node yield in these regions. This is due to a magnified surgical field of view and improved imaging, with improved dexterity and flexibility of instruments and physiological tremor filtering systems. Regarding the left upper mediastinum, where the left RLN is dissected, the robotic approach is able to provide the surgeon with an upgraded vision and field of view, promoting the identification of the nerve. This, along with an improved posture and comfort when operating, enables the surgeon to have a greater ease of access to such a narrow and out of reach area like the left upper mediastinum.
A minimum of 15 lymph nodes necessary for accurate staging [63] was achieved in all studies, which reflects the quality of oncologic resection and staging. Whether these additional nodes using the RAMIE technique represent better long-term resection is still unclear. Clinically, a more relevant measure to be reported by authors would be the total number of positive lymph nodes harvested, as these directly influence the disease’s course of action and prognosis.
Patients that underwent neoadjuvant therapy before surgery often present with edema and fibrosis in the affected areas. In these patients, RAMIE performed particularly better than cMIE due to its advantageous features [62].
Studies that performed PSM showed similar results, further increasing evidence regarding RAMIE’s superiority in the dissection of lymph nodes during esophagectomies.
30- and 90-day mortality rates
Early mortality rates had no statistically significant differences between the two surgical approaches. The same conclusion was attained by previous meta-analyses [17, 18, 20,21,22,23,24]. 30-day mortality rates are below 2% and 90-day rates are below 5% in both groups. These two indicators reflect greatly on the quality of surgery performed on esophageal cancer patients and the occurrence of major complications after the procedure [64].
Postoperative outcomes
Anastomotic and chyle leakage
Anastomotic leakage is one of the most impactful complications that may occur after an esophagectomy, as it leads to high rates of morbidity and mortality in such patients. Several important factors are highlighted when analyzing predisposing conditions that lead to this complication. Preoperatively, diseases that compromise the perfusion of the anastomosis are congestive heart failure, coronary artery disease, diabetes, smoking, and hypertension. With regard specifically to the surgery, the anastomotic technique, the location of the anastomosis, the type of conduit and its location all contribute to the quality of the performed anastomosis and therefore influence its longevity [65].
In our study, cMIE had a statistically significant lower rate of anastomotic leakage. This is highly influenced by a single OCS that had a large study population from a national database [58]. One of their reported limitations is the lack of information regarding the anastomotic technique used in all procedures, which directly influences this outcome. Furthermore, this study did not exclude the surgical learning curve of minimally invasive operations, which is inherently accompanied by a higher rate of complications in such a critical phase of the surgery. If this study is removed from the analysis, results are no longer statistically significant, favoring no technique.
On the other hand, chyle leakage, another important factor in postoperative morbidity, did not favor any approach. Heterogeneity was low, and there was no selection bias upon funnel plot analysis.
Information from PSM studies corroborates these results, showing similar rates of anastomotic and chyle leakage.
Recurrent laryngeal nerve palsy
Thoracic surgeries, specifically esophagectomies, imply heavy esophageal manipulation and traction, which may damage RLN bilaterally. Neuropathy caused by stretching, compression, thermal and ischemic injury is a common complication that brings significant morbidity. Paralysis of the RLN leads to pulmonary complications, hoarseness, longer length of hospital stay, and long-term recovery, which in turn, deteriorate postoperative quality of life [66, 67].
Despite more extended lymph node dissection in RLNs in the robotic procedure, the occurrence of RLN palsy was similar in both groups. This result is also in accordance with previous meta-analyses [18,19,20,21,22, 24]. Therefore, one may argue that with RAMIE’s improved manipulation and visualization features, it is possible to attain better lymph node yields without compromising RLN damage and paralysis. It is also possible that once surgeons complete their learning curves in the technique, the rate of this complication decreases. There is also a need for further studies and information that differentiate the extent of nerve injury, for further characterization. PSM data lead us to the same conclusion.
Pulmonary, cardiac and infectious complications
Overall complications greatly reflect both the immediate response to such a major surgery, but also the prognosis and long-term development of the disease [68]. It has been proven that these short-term complications directly influence tumor progression and long-term survival. Minimally invasive surgical approaches and multidisciplinary case management are successful in the prevention of such outcomes [69].
In our unmatched pool of data, there was a great tendency towards lower pulmonary complication rates in the RAMIE approach, with a low level of heterogeneity among studies. This may be explained by the recent advantages that RAMIE has brought. Improved magnification and dexterity in the surgical field provide a better visualization and manipulation of ligaments and fasciae [70]. Due to this, the avoidance of dissection of the vagus nerve and preservation of increased segments of pulmonary parenchyma are better achieved. The vagus nerve is responsible for the regulation of essential pulmonary functions and mechanisms. The cough reflex, mucous production, bronchus diameter and regulation of inflammation and edema are all regulated by the vagus nerve [71]. Therefore, vagotomy will decrease the action of these response mechanisms to esophagectomy’s aggression and inflammatory response [72], leading to an increase in the rate of pulmonary complications. RAMIE may be able to overcome this unintended result and reduce the incidence of pneumonia, acute respiratory distress syndrome and other complications. Cardiac and infectious complications had similar rates of occurrence in the two surgical techniques. Recent meta-analyses present similar results [17, 18, 20, 22,23,24].
Analysis of PSM information further increase evidence of RAMIE’s superiority over cMIE in regards to pulmonary complications. There was a reduction of approximately 30% in the incidence of these complications. Conclusions regarding cardiac and infectious complications remained unaltered.
Length of hospital stay
Lower rates of postoperative complications directly influence a patient’s length of hospital stay after an esophagectomy [73]. As our results suggest a lower incidence of pulmonary complications in RAMIE patients, it is expected that those will require a shorter period of hospitalization.
Our analysis favors RAMIE to provide statistically significant shorter lengths of hospital stay, when compared to cMIE. Nonetheless, these results are to be taken with caution, as heterogeneity is high, and three studies [28, 46, 54] showed discrepant results when compared to the rest of the included ones. The calculated mean length of hospital stay was approximately 19 days in the RAMIE group and 33 days in the cMIE group. These are greatly influenced by Asian cultural and non-clinical factors that extend hospitalization further [74].
Upon analysis of PSM information, this difference is greatly attenuated and no longer statistically significant. The RAMIE group presented with a mean length of hospital stay of 17 days, and the cMIE group with 19 days. Some of the previous outlier cases are no longer taken into consideration and baseline patient characteristics are more similar. Nonetheless, there is still a high tendency towards a shorter length of hospital stay associated with the RAMIE procedure, which should be analyzed considering the context.
Limitations
Our study acknowledges some limitations which may compromise the analysis of the attained results. First, all but one of the included studies are observational clinical studies, most of these being retrospective non-randomized comparative studies. Even though all studies present an assessed low risk of bias, patient selection bias is inherently present due to several cultural, clinical, and non-clinical factors of the preferred surgical approach. Furthermore, this systematic review includes studies that gather data from large national databases, from multiple institutions and different managing software. This results in a non-uniform way of coding and storing information, which may lead to unpredicted conclusions. Second, the wide majority of the reviewed studies represent the Asian population and local health policies and standards, which may not be generalizable globally. Third, heterogeneity was high in several measured outcomes, which may compromise the validity of the results. Varied reasons partially account for these differences, amongst which, surgical team’s and surgeons’ previous experience and position in the learning curve of the used surgical approach influence the most. Baseline demographics and clinical characteristics of patients in both groups also vastly impact heterogeneity. In fact, it was only possible to gather PSM information from 18 of the 35 included studies. Fourth, the analyzed studies included different surgical approaches to esophagectomy (McKeown and Ivor-Lewis), which may generate another degree of heterogeneity to the data.
Conclusion
In conclusion, our study reinforces RAMIE’s safety and feasibility in the curative treatment of esophageal cancer patients. Analyzing the collected information, RAMIE shows superiority over cMIE in reducing blood lost during surgery, increasing the amount of dissected lymph nodes, decreasing pulmonary complications, and shortening periods of hospitalization after such an extensive and challenging surgery. Nonetheless, there seems to be a tendency towards shorter operative times and lower rates of anastomotic leakage using cMIE. Conversion to open surgery, 30- and 90-day mortality rates, chyle leakage, RLN palsy and cardiac and infectious complications did not show statistically significant differences between the two approaches.
Upon review of these results, RAMIE has indicated to be non-inferior to cMIE, with prospects of superiority in some fields. With the consolidation of the robotic approach, new opportunities will arise for the implementation of systems that enhance the surgical experience, such as haptic features, with kinesthetic and tactile feedback. Integration of preoperative imaging exams in the surgical field of view is also a possibility, overlapping anatomical structures with auxiliary images, which in turn facilitates the identification of dissection layers, tumor borders, and others. With the improvement of the surgeon’s comfort and confidence in decision-making, patients will be provided with a better standard of care.
Further studies, especially RCTs, are still necessary to overcome the limitations presented, in order to achieve definite conclusions regarding RAMIE’s current position in the treatment of esophageal cancer patients.
Data availability
No datasets were generated or analyzed during the current study.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249
Borggreve AS, Kingma BF, Domrachev SA, Koshkin MA, Ruurda JP, van Hillegersberg R et al (2018) Surgical treatment of esophageal cancer in the era of multimodality management. Ann N Y Acad Sci 1434(1):192–209
Iriarte F, Su S, Petrov RV, Bakhos CT, Abbas AE (2021) Surgical management of early esophageal cancer. Surg Clin N Am 101(3):427–441
Miyata H, Yamasaki M, Kurokawa Y, Takiguchi S, Nakajima K, Fujiwara Y et al (2011) Multimodal treatment for resectable esophageal cancer. Gen Thorac Cardiovasc Surg 59(7):461–466
Paul S, Altorki N (2014) Outcomes in the management of esophageal cancer. J Surg Oncol 110(5):599–610
Vaghjiani RG, Molena D (2017) Surgical management of esophageal cancer. Chin Clin Oncol 6(5):47
Akhtar NM, Chen D, Zhao Y, Dane D, Xue Y, Wang W et al (2020) Postoperative short-term outcomes of minimally invasive versus open esophagectomy for patients with esophageal cancer: An updated systematic review and meta-analysis. Thorac Cancer 11(6):1465–1475
Guo W, Ma X, Yang S, Zhu X, Qin W, Xiang J et al (2016) Combined thoracoscopic-laparoscopic esophagectomy versus open esophagectomy: a meta-analysis of outcomes. Surg Endosc 30(9):3873–3881
Muller-Stich BP, Probst P, Nienhuser H, Fazeli S, Senft J, Kalkum E et al (2021) Meta-analysis of randomized controlled trials and individual patient data comparing minimally invasive with open oesophagectomy for cancer. Br J Surg 108(9):1026–1033
Siaw-Acheampong K, Kamarajah SK, Gujjuri R, Bundred JR, Singh P, Griffiths EA (2020) Minimally invasive techniques for transthoracic oesophagectomy for oesophageal cancer: systematic review and network meta-analysis. BJS Open 4(5):787–803
Wang K, Zhong J, Liu Q, Lin P, Fu J (2022) A propensity score-matched analysis of thoracolaparoscopic vs open McKeown’s esophagectomy. Ann Thorac Surg 113(2):473–481
Bao T, Li KK, Liu B, Zhao XL, Wang YJ, Guo W (2022) Learning curve and associated prognosis of minimally invasive McKeown esophagectomy. Ann Thorac Surg 114(3):933–939
van Workum F, Stenstra M, Berkelmans GHK, Slaman AE, van Berge Henegouwen MI, Gisbertz SS et al (2019) Learning curve and associated morbidity of minimally invasive esophagectomy: a retrospective multicenter study. Ann Surg 269(1):88–94
Kernstine KH, DeArmond DT, Karimi M, Van Natta TL, Campos JH, Yoder MR, Everett JE (2004) The robotic, 2-stage, 3-field esophagolymphadenectomy. J Thorac Cardiovasc Surg 127(6):1847–1849
Egberts JH, Stein H, Aselmann H, Hendricks A, Becker T (2017) Fully robotic da Vinci Ivor-Lewis esophagectomy in four-arm technique-problems and solutions. Dis Esophagus 30(12):1–9
van der Sluis PC, Tagkalos E, Hadzijusufovic E, Babic B, Uzun E, van Hillegersberg R et al (2021) Robot-assisted minimally invasive esophagectomy with intrathoracic anastomosis (Ivor Lewis): promising results in 100 consecutive patients (the European experience). J Gastrointest Surg 25(1):1–8
Angeramo CA, Bras Harriott C, Casas MA, Schlottmann F (2021) Minimally invasive Ivor Lewis esophagectomy: robot-assisted versus laparoscopic-thoracoscopic technique. Syst Rev Meta-Anal Surg 170(6):1692–1701
Huang Y, Zhao YL, Song JD (2021) Early outcomes with robot-assisted vs. minimally invasive esophagectomy for esophageal cancer: a systematic review and meta-analysis of matched studies. Eur Rev Med Pharmacol Sci 25(24):7887–7897
Li XK, Xu Y, Zhou H, Cong ZZ, Wu WJ, Qiang Y, Shen Y (2021) Does robot-assisted minimally invasive oesophagectomy have superiority over thoraco-laparoscopic minimally invasive oesophagectomy in lymph node dissection? Dis Esophagus 34(2):doaa050
Magouliotis DE, Zotos PA, Fergadi MP, Koukousaki D, Zacharoulis D, Diamantis A et al (2022) Meta-analysis of robot-assisted versus video-assisted McKeown esophagectomy for esophageal cancer. Updates Surg 74(5):1501–1510
Manigrasso M, Vertaldi S, Marello A, Antoniou SA, Francis NK, DePalma GD, Milone M (2021) Robotic esophagectomy. A systematic review with meta-analysis of clinical outcomes. J Pers Med. 11(7):640
Mederos MA, de Virgilio MJ, Shenoy R, Ye L, Toste PA, Mak SS et al (2021) Comparison of clinical outcomes of robot-assisted, video-assisted, and open esophagectomy for esophageal cancer: a systematic review and meta-analysis. JAMA Netw Open 4(11):e2129228
Zhang Y, Dong D, Cao Y, Huang M, Li J, Zhang J et al (2023) Robotic versus conventional minimally invasive esophagectomy for esophageal cancer: a meta-analysis. Ann Surg 278(1):39–50
Zhou J, Xu J, Chen L, Hu J, Shu Y (2022) McKeown esophagectomy: robot-assisted versus conventional minimally invasive technique-systematic review and meta-analysis. Dis Esophagus 35(10):doac011
Slim K, Nini E, Forestier D, Kwiatkowski F, Panis Y, Chipponi J (2003) Methodological index for non-randomized studies (minors): development and validation of a new instrument. ANZ J Surg 73(9):712–716
Sterne JAC, Savovic J, Page MJ, Elbers RG, Blencowe NS, Boutron I et al (2019) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366:l4898
Wan X, Wang W, Liu J, Tong T (2014) Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol 14:135
Suda K, Ishida Y, Kawamura Y, Inaba K, Kanaya S, Teramukai S et al (2012) Robot-assisted thoracoscopic lymphadenectomy along the left recurrent laryngeal nerve for esophageal squamous cell carcinoma in the prone position: technical report and short-term outcomes. World J Surg 36(7):1608–1616
Weksler B, Sharma P, Moudgill N, Chojnacki KA, Rosato EL (2012) Robot-assisted minimally invasive esophagectomy is equivalent to thoracoscopic minimally invasive esophagectomy. Dis Esophagus 25(5):403–409
Park S, Hwang Y, Lee HJ, Park IK, Kim YT, Kang CH (2016) Comparison of robot-assisted esophagectomy and thoracoscopic esophagectomy in esophageal squamous cell carcinoma. J Thorac Dis 8(10):2853–2861
Chao YK, Hsieh MJ, Liu YH, Liu HP (2018) Lymph node evaluation in robot-assisted versus video-assisted thoracoscopic esophagectomy for esophageal squamous cell carcinoma: a propensity-matched analysis. World J Surg 42(2):590–598
He H, Wu Q, Wang Z, Zhang Y, Chen N, Fu J, Zhang G (2018) Short-term outcomes of robot-assisted minimally invasive esophagectomy for esophageal cancer: a propensity score matched analysis. J Cardiothorac Surg 13(1):52
Chen J, Liu Q, Zhang X, Yang H, Tan Z, Lin Y, Fu J (2019) Comparisons of short-term outcomes between robot-assisted and thoraco-laparoscopic esophagectomy with extended two-field lymph node dissection for resectable thoracic esophageal squamous cell carcinoma. J Thorac Dis 11(9):3874–3880
Deng HY, Luo J, Li SX, Li G, Alai G, Wang Y et al (2019) Does robot-assisted minimally invasive esophagectomy really have the advantage of lymphadenectomy over video-assisted minimally invasive esophagectomy in treating esophageal squamous cell carcinoma? A propensity score-matched analysis based on short-term outcomes. Dis Esophagus 32(7):110
Grimminger PP, Tagkalos E, Hadzijusufovic E, Corvinus F, Babic B, Lang H (2019) Change from hybrid to fully minimally invasive and robotic esophagectomy is possible without compromises. Thorac Cardiovasc Surg 67(7):589–596
Motoyama S, Sato Y, Wakita A, Kawakita Y, Nagaki Y, Imai K, Minamiya Y (2019) Extensive lymph node dissection around the left laryngeal nerve achieved with robot-assisted thoracoscopic esophagectomy. Anticancer Res 39(3):1337–1342
Zhang Y, Han Y, Gan Q, Xiang J, Jin R, Chen K et al (2019) Early outcomes of robot-assisted versus thoracoscopic-assisted Ivor Lewis esophagectomy for esophageal cancer: a propensity score-matched study. Ann Surg Oncol 26(5):1284–1291
Chao YK, Wen YW, Chuang WY, Cerfolio RJ (2020) Transition from video-assisted thoracoscopic to robotic esophagectomy: a single surgeon’s experience. Dis Esophagus 33(2):doz033
Gong L, Jiang H, Yue J, Duan X, Tang P, Ren P et al (2020) Comparison of the short-term outcomes of robot-assisted minimally invasive, video-assisted minimally invasive, and open esophagectomy. J Thorac Dis 12(3):916–924
Meredith K, Blinn P, Maramara T, Takahashi C, Huston J, Shridhar R (2020) Comparative outcomes of minimally invasive and robotic-assisted esophagectomy. Surg Endosc 34(2):814–820
Tagkalos E, Goense L, Hoppe-Lotichius M, Ruurda JP, Babic B, Hadzijusufovic E et al (2020) Robot-assisted minimally invasive esophagectomy (RAMIE) compared to conventional minimally invasive esophagectomy (MIE) for esophageal cancer: a propensity-matched analysis. Dis Esophagus 33(4):doz060
Yang Y, Zhang X, Li B, Hua R, Yang Y, He Y et al (2020) Short- and mid-term outcomes of robotic versus thoraco-laparoscopic McKeown esophagectomy for squamous cell esophageal cancer: a propensity score-matched study. Dis Esophagus 33(6):doz080
Ali AM, Bachman KC, Worrell SG, Gray KE, Perry Y, Linden PA, Towe CW (2021) Robotic minimally invasive esophagectomy provides superior surgical resection. Surg Endosc 35(11):6329–6334
Duan X, Yue J, Chen C, Gong L, Ma Z, Shang X et al (2021) Lymph node dissection around left recurrent laryngeal nerve: robot-assisted vs. video-assisted McKeown esophagectomy for esophageal squamous cell carcinoma. Surg Endosc 35(11):6108–6116
Ninomiya I, Okamoto K, Yamaguchi T, Saito H, Terai S, Moriyama H et al (2021) Optimization of robot-assisted thoracoscopic esophagectomy in the lateral decubitus position. Esophagus 18(3):482–488
Oshikiri T, Goto H, Horikawa M, Urakawa N, Hasegawa H, Kanaji S et al (2021) Incidence of recurrent laryngeal nerve palsy in robot-assisted versus conventional minimally invasive McKeown esophagectomy in prone position: a propensity score-matched study. Ann Surg Oncol 28(12):7249–7257
Shirakawa Y, Noma K, Kunitomo T, Hashimoto M, Maeda N, Tanabe S et al (2021) Initial introduction of robot-assisted, minimally invasive esophagectomy using the microanatomy-based concept in the upper mediastinum. Surg Endosc 35(12):6568–6576
Tsunoda S, Obama K, Hisamori S, Nishigori T, Okamura R, Maekawa H, Sakai Y (2021) Lower incidence of postoperative pulmonary complications following robot-assisted minimally invasive esophagectomy for esophageal cancer: propensity score-matched comparison to conventional minimally invasive esophagectomy. Ann Surg Oncol 28(2):639–647
Balasubramanian S, Chittawadagi B, Misra S, Ramakrishnan P, Chinnusamy P (2022) Propensity matched analysis of short term oncological and perioperative outcomes following robotic and thoracolaparoscopic esophagectomy for carcinoma esophagus- the first Indian experience. J Robot Surg 16(1):97–105
Chouliaras K, Attwood K, Brady M, Takahashi H, Peng JS, Yendamuri S et al (2022) Robotic versus thoraco-laparoscopic minimally invasive Ivor Lewis esophagectomy, a matched-pair single-center cohort analysis. Dis Esophagus 36(1):doac037
Dezube AR, Kucukak S, De Leon LE, Kostopanagiotou K, Jaklitsch MT, Wee JO (2022) Risk of chyle leak after robotic versus video-assisted thoracoscopic esophagectomy. Surg Endosc 36(2):1332–1338
Fujita T, Sato K, Ozaki A, Akutsu T, Fujiwara H, Kojima T, Daiko H (2022) Propensity-matched analysis of the short-term outcome of robot-assisted minimally invasive esophagectomy versus conventional thoracoscopic esophagectomy in thoracic esophageal cancer. World J Surg 46(8):1926–1933
Kulkarni A, Mulchandani JG, Sadat MS, Shetty N, Shetty S, Kumar MP, Kudari A (2022) Robot-assisted versus video-assisted thoraco-laparoscopic McKeown’s esophagectomy for esophageal cancer: a propensity score-matched analysis of minimally invasive approaches. J Robot Surg 16(6):1289–1297
Morimoto Y, Kawakubo H, Ishikawa A, Matsuda S, Hijikata N, Ando M et al (2022) Short-term outcomes of robot-assisted minimally invasive esophagectomy with extended lymphadenectomy for esophageal cancer compared with video-assisted minimally invasive esophagectomy: a single-center retrospective study. Asian J Endosc Surg 15(2):270–278
Trung LV, Loc NVV, Tien TPD, Ai BD, Lam TLQ, Vuong NL (2022) Robot-assisted versus thoraco-laparoscopic McKeown esophagectomy for esophageal cancer: a propensity score-matched study. J Gastrointest Surg 26(5):1093–1096
van der Sluis PC, Babic B, Uzun E, Tagkalos E, Berlth F, Hadzijusufovic E et al (2022) Robot-assisted and conventional minimally invasive esophagectomy are associated with better postoperative results compared to hybrid and open transthoracic esophagectomy. Eur J Surg Oncol 48(4):776–782
Jiang H, Guo X, Sun Y, Hua R, Li B, Li Z (2023) Robot-assisted versus thoracolaparoscopic oesophagectomy for locally advanced oesophageal squamous cell carcinoma after neoadjuvant chemoradiotherapy. Eur J Surg Oncol 49(4):832–837
Khaitan PG, Vekstein AM, Thibault D, Kosinski A, Hartwig MG, Block M et al (2023) robotic esophagectomy trends and early surgical outcomes: the US experience. Ann Thorac Surg 115(3):710–717
Narendra A, Barbour A (2023) Introducing robotic oesophagectomy into an Australian practice: an assessment of the early procedural outcomes and learning curve. ANZ J Surg 93(5):1300–1305
Sun HB, Jiang D, Liu XB, Xing WQ, Liu SL, Chen PN et al (2023) Perioperative outcomes and learning curve of robot-assisted McKeown esophagectomy. J Gastrointest Surg 27(1):17–26
Turner KM, Delman AM, Johnson K, Patel SH, Wilson GC, Shah SA, Van Haren RM (2023) Robotic-assisted minimally invasive esophagectomy: postoperative outcomes in a nationwide cohort. J Surg Res 283:152–160
Yang Y, Li B, Yi J, Hua R, Chen H, Tan L et al (2022) Robot-assisted versus conventional minimally invasive esophagectomy for resectable esophageal squamous cell carcinoma: early results of a multicenter randomized controlled trial: the RAMIE trial. Ann Surg 275(4):646–653
Merkow RP, Bilimoria KY, Chow WB, Merkow JS, Weyant MJ, Ko CY, Bentrem DJ (2012) Variation in lymph node examination after esophagectomy for cancer in the United States. Arch Surg 147(6):505–511
Talsma AK, Lingsma HF, Steyerberg EW, Wijnhoven BP, Van Lanschot JJ (2014) The 30-day versus in-hospital and 90-day mortality after esophagectomy as indicators for quality of care. Ann Surg 260(2):267–273
Kassis ES, Kosinski AS, Ross P Jr, Koppes KE, Donahue JM, Daniel VC (2013) Predictors of anastomotic leak after esophagectomy: an analysis of the society of thoracic surgeons general thoracic database. Ann Thorac Surg 96(6):1919–1926
Myssiorek D (2004) Recurrent laryngeal nerve paralysis: anatomy and etiology. Otolaryngol Clin N Am 37(1):25–44 (v)
Scholtemeijer MG, Seesing MFJ, Brenkman HJF, Janssen LM, van Hillegersberg R, Ruurda JP (2017) Recurrent laryngeal nerve injury after esophagectomy for esophageal cancer: incidence, management, and impact on short- and long-term outcomes. J Thorac Dis 9(Suppl 8):S868–S878
Booka E, Takeuchi H, Suda K, Fukuda K, Nakamura R, Wada N et al (2018) Meta-analysis of the impact of postoperative complications on survival after oesophagectomy for cancer. BJS Open 2(5):276–284
Booka E, Kikuchi H, Hiramatsu Y, Takeuchi H (2021) The impact of infectious complications after esophagectomy for esophageal cancer on cancer prognosis and treatment strategy. J Clin Med 10(19):4614
Weijs TJ, Ruurda JP, Luyer MDP, Cuesta MA, van Hillegersberg R, Bleys RLAW (2017) New insights into the surgical anatomy of the esophagus. J Thorac Dis 9(S8):S675–S680
Mazzone SB, Canning BJ (2013) Autonomic neural control of the airways. Handb Clin Neurol 117:215–228
D’Journo XB, Michelet P, Marin V, Diesnis I, Blayac D, Doddoli C et al (2010) An early inflammatory response to oesophagectomy predicts the occurrence of pulmonary complications. Eur J Cardiothorac Surg 37(5):1144–1151
Park KU, Rubinfeld I, Hodari A, Hammoud Z (2016) Prolonged length of stay after esophageal resection: identifying drivers of increased length of stay using the NSQIP database. J Am Coll Surg 223(2):286–290
Tiessen J, Kambara H, Sakai T, Kato K, Yamauchi K, McMillan C (2013) What causes international variations in length of stay: a comparative analysis for two inpatient conditions in Japanese and Canadian hospitals. Health Serv Manag Res 26(2–3):86–94
Funding
Open access funding provided by FCT|FCCN (b-on). The authors declare that no funds, grants or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
R. Perry: conceptualization, data curation, formal analysis, investigation, validation, visualization, writing—original draft, writing—review and editing. J. P. Barbosa: data curation, formal analysis, investigation, resources, supervision, validation, visualization, writing—original draft, writing—review and editing. I. Perry: data curation, investigation, validation, writing—original draft, writing—review and editing. J. Barbosa: conceptualization, investigation, supervision, visualization, writing—review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Perry, R., Barbosa, J.P., Perry, I. et al. Short-term outcomes of robot-assisted versus conventional minimally invasive esophagectomy for esophageal cancer: a systematic review and meta-analysis of 18,187 patients. J Robotic Surg 18, 125 (2024). https://doi.org/10.1007/s11701-024-01880-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11701-024-01880-3