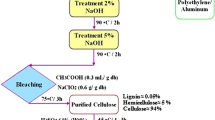
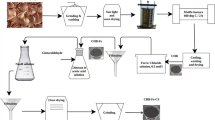
Abstract
One of the most important principles in the twenty-first century is to provide clean water, and the use and dimensions of advanced technologies in water supply are important. One of the most effective directing methods in water treatment is the adsorption method. Especially in recent years, with the re-evaluation of wastes on the agenda, instead of being left to rot, shellfish are re-evaluated by chemical or biological methods and new products are obtained. Chitin and its main derivative, chitosan, are the main products produced in this way. In order to increase the performance and efficiency of using chitosan in water treatment, it is necessary to mechanically strengthen and functionalize chitosan. In this study, it was aimed to prepare a graphene oxide (GO)-doped chitosan microcapsule modified with Fe3O4 nanoparticle. Chitosan-based microcapsules containing different ratios of GO@Fe3O4 were prepared. GO, GO@Fe3O4 and microcapsules (MC); Characterizations were carried out using methods such as SEM, FTIR, EDX mapping, XPS and TGA, and arsenic adsorption performance was investigated in real water samples. In line with the results obtained, it was observed that it showed a stable and high arsenic adsorption performance of 95%.








Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Akın I, Zor E (2016) Grafen Tabanlı Nanokompozit İçerikli Membran Hazırlama ve Karakterizasyonu. S.Ü.BAP-16401085
Banerjee SB, Iyer ES, Kashyap R (2003) Corporate environmentalism: antecedents and influence of industry type. J Mark 67(2):106–122. https://doi.org/10.2307/30040526
Bao X, Chen L (2012) Recent progress in distributed fiber optic sensors. Sensors 12(7):8601–8639. https://doi.org/10.3390/s120708601
Baran T (2020) Pd NPs@Fe3O4/chitosan/pumice hybrid beads: a highly active, magnetically retrievable and reusable nanocatalyst for cyanation of aryl halides. Carbohyd Polym 237:116105. https://doi.org/10.1016/j.carbpol.2020.116105
Baran T, Mentes A (2015) Cu(II) and Pd(II) complexes of water soluble O-carboxymethyl chitosan schiff bases: synthesis characterization. Int J Biol Macromol 79:542–554. https://doi.org/10.1016/j.ijbiomac.2015.05.021
Chandra V, Park J, Chun Y, Lee JW, Hwang C, Kim K (2010) Water-dispersible magnetite-reduced graphene oxide composites for arsenic removal. ACS Nano 4(7):3979–3986. https://doi.org/10.1021/nn1008897
Chen YW, Wang JL (2011) Preparation and characterization of magnetic chitosan nanoparticles and its application for Cu(II) removal. Chem Eng J 168:286–292. https://doi.org/10.1016/j.cej.2011.01.006
Chen YW, Wang JL (2012) The characteristics and mechanism of Co(II) removal from aqueous solution by a novel xanthate-modified magnetic chitosan. Nucl Eng Des 242:452–457. https://doi.org/10.1016/j.nucengdes.2011.11.004
Chen YW, Hu J, Wang JL (2012) Kinetics and thermodynamics of Cu(II) biosorption on to a novel magnetic chitosan composite bead. Environ Technol 33:2345–2351. https://doi.org/10.1080/09593330.2012.668940
Daniel RD, Sungjin P, Christopher W, Bielawski, Rodney SR (2010) The chemistry of graphene oxide. Chem Soc Rev 39:228–240. https://doi.org/10.1007/978-3-319-15500-5_3
Dung MD, Nga TTV, Lan NT, Thanh NK (2022) Adsorption behavior and mechanism of As(V) on magnetic Fe3O4− graphene oxide (GO) nanohybrid composite material. Anal Sci 38:427–436. https://doi.org/10.1007/s44211-022-00064-z
Estevinho BN, Rocha F, Santos L, Alves A (2013) Microencapsulation with chitosan by spray drying for industry applications: a review. Trends Food Sci Technol 31(2):138–155. https://doi.org/10.1016/j.tifs.2013.04.001
Geng Z, Lin Y, Yu X, Shen Q, Ma L, Li Z, Pan N, Wang X (2012) Highly efficient dye adsorption and removal: a functional hybrid of reduced graphene oxide–Fe3O4 nanoparticles as an easily regenerative adsorbent. J Mater Chem 22:3527–3535. https://doi.org/10.1039/c2jm15544c
Gomaa YA, El-Khordagui LK, Boraei NA, Darwish IA (2010) Chitosan microparticles incorporating a hydrophilic sunscreen agent. Carbohyd Polym 81(2):234–242. https://doi.org/10.1016/j.carbpol.2010.02.024
Hummers WS Jr, Offeman RE (1958) Preparation of graphitic oxide. J Am Chem Soc 80(6):1339. https://doi.org/10.1021/ja01539a017
Jin F, Lv W, Zhang C, Li Z, Su R, Qi W, Yang QH, He Z (2013) High-performance ultrafiltration membranes based on polyethersulfone−graphene oxide composites. RSC Adv 3:21394–21397. https://doi.org/10.1039/c3ra42908c
Karthika V, AlSalhi MS, Devanesan S, Gopinath K, Arumugam A, Govindarajan M (2020) Chitosan overlaid Fe3O4/rGO nanocomposite for targeted drug delivery imaging and biomedical applications. Sci Rep 10:18912
Kassaee MZ, Motamedi E, Majdi M (2011) Magnetic Fe3O4− graphene oxide/polystyrene: fabrication and characterization of a promising nanocomposite. Chem Eng J 172(1):540–549. https://doi.org/10.1016/j.cej.2011.05.093
Kuchlyan J, Kundu N, Banik D, Roy A, Sarkar N (2015) Spectroscopy and fluorescence lifetime imaging microscopy to probe the interaction of bovine serum albumin with graphene oxide. Langmuir 31:13793–13801
Liu M, Wen T, Wu X, Chen C, Hu J, Li J, Wang X (2013) Synthesis of porous Fe3O4 hollow microspheres/graphene oxide composite for Cr(VI) removal. Dalton Trans 42:14710. https://doi.org/10.1039/C3DT50955A
Marcano DC, Kosynkin DV, Berlin JM, Sinitskii A, Sun Z, Slesarev A, Alemany LB, Lu W, Tour JM (2010) Improved synthesis of graphene oxide. ACS Nano 4(8):4806–4814. https://doi.org/10.1021/nn1006368
Monllor P, Bonet MA, Cases F (2007) Characterization of the behaviour of flavour microcapsules in cotton fabrics. Eur Polym J 43:2481–2490. https://doi.org/10.1016/j.eurpolymj.2007.04.004
Ngah WSW, Endud CS, Mayanar R (2002) Removal of copper(II) ions from aqueous solution onto chitosan and cross-linked chitosan beads. React Funct Polym 50:181–190. https://doi.org/10.1016/S1381-5148(01)00113-4
Nor AZ, Yacou C, Motuzas J, Zhang X, Diniz da Costa JC (2014) Structural and functional investigation of graphene oxide–Fe3O4 nanocomposites for the heterogeneous fenton-like reaction. Sci Rep 4:4594. https://doi.org/10.1038/srep04594
Övez B, Yüksel M (2002) Parfümlerin çapraz bağlı mikrokapsüllerden yavaş salgılanmaları. Ekoloji Çevre Dergisi 11(43):26–29
Patel G, Patel S, Prajapati D, Mehta R (2010) RP-HPLC method for simultaneous estimation of amlodipine besylate and hydrochlorothiazide in combined dosage forms. Stamford J Pharm Sci 3(1):49–53. https://doi.org/10.3329/sjps.v3i1.6798
Rudall KM, Kenchington W (1973) The chitin system. Biol Rev 48(4):597–633. https://doi.org/10.1111/j.1469-185X.1973.tb01570.x
Sargın I, Kaya M, Arslan G, Baran T, Ceter T (2015) Preparation and characterisation of biodegradable pollen–chitosan microcapsules and its application in heavy metal removal. Biores Technol 177:1–7. https://doi.org/10.1016/j.biortech.2014.11.067
Tran VS, Ngo HH, Guo W, Nguyen TH, Luong TML, Nguyen XH, Phan TLA, Le VT, Nguyen MP, Nguyen MK (2023) New chitosan-biochar composite derived from agricultural waste for removing sulfamethoxazole antibiotics in water. Biores Technol 385:129384. https://doi.org/10.1016/j.biortech.2023.129384
Travlou NA, Kyzas GZ, Lazaridis NK, Deliyanni EA (2013) Functionalization of graphite oxide with magnetic chitosan for the preparation of a nanocomposite dye adsorbent. Langmuir 29:1657–1668. https://doi.org/10.1021/la304696y
Tung VC, Chen L, Allen MJ, Wassei JK, Nelson K, Kaner RB, Yang Y (2009) Low-temperature solution processing of graphene-carbon nanotube hybrid materials for high-performance transparent conductors. Nano Lett 9(5):1949–1955. https://doi.org/10.1021/nl9001525
Van Hoa N, TrungKhong T, Hoang Quyen TT, Si TT (2016) One-step facile synthesis of mesoporous graphene/Fe3O4/chitosan nanocomposite and its adsorption capacity for a textile dye. J Water Process Eng 9:170–178
Wang J, Chen C (2014) Chitosan-based biosorbents: modification and application for biosorption of heavy metals and radionuclides. Biores Technol 160:129–141. https://doi.org/10.1016/j.biortech.2013.12.110
Wang J, Zhuang S (2022) Chitosan-based materials: preparation modification and application. J Clean Prod 355:131825. https://doi.org/10.1016/j.jclepro.2022.131825
Wang W, Tang B, Ju B, Gao Z, Xiu J, Zhang S (2017) Fe3O4− functionalized graphene nanosheet embedded phase change material composites: efficient magnetic-and sunlight driven energy conversion and storage. J Mater Chem A 5(3):958–968. https://doi.org/10.1039/C6TA07144A
Zhang G, Ren Z, Zhang X, Chen J (2013) Nanostructured iron(III)-copper(II) binary oxide: a novel adsorbent for enhanced arsenic removal from aqueous solutions. Water Res 47(12):4022–4031. https://doi.org/10.1016/j.watres.2012.11.059
Zheng N, Qiao L, Liu J, Lu J, Li W, Li C, Liu Q, Xue Y, Zhang Q (2019) Microcapsules of multilayered shell structure synthesized via one-part strategy and their application in self-healing coatings. Compos Commun 12:26–32. https://doi.org/10.1016/j.coco.2018.12.006
Zheng N, Liu J, Wang Y, Li C, Zhang Q (2021) Preparation of chitosan-reduced graphene oxide (CS-RGO) microcapsules and its application in UV/moisture-induced self-healing coatings. Prog Org Coat 151:106055. https://doi.org/10.1016/j.porgcoat.2020.106055
Zheng N, Liu J, Wang G, Yao P, Dang L, Liu Z, Lu J, J Li W (2023) Robust UV/moisture dual curable PDMS-microcapsule-silica functional material for self-healing, antifouling, and antibacterial applications. Nano Res 16:7810–7819. https://doi.org/10.1007/s12274-023-5563-8
Zotzmann J, Feldbusch E, Kruppke I, Aibibu D, Regenspurg S (2023) Removal of lead and copper ions from geothermal brine at various temperatures and salinities using chemically crosslinked chitosan. Appl Geochem 155:105733. https://doi.org/10.1016/j.apgeochem.2023.105733
Acknowledgements
I would like to thank Necmettin Erbakan University Scientific Research Projects Coordinator for their support.
Funding
This research was funded by the Scientific Research Projects Co-ordinatorship of Necmettin Erbakan University. (Project number: 191715001).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
I have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Akin, İ. Preparation of chitosan microcapsules containing modified graphene oxide and investigation of water treatment performance. Chem. Pap. 78, 2005–2013 (2024). https://doi.org/10.1007/s11696-023-03222-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-023-03222-3