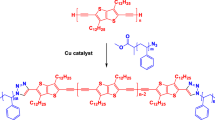
Abstract
A conjugated diblock copolymer of poly(3-hexylthiophene)-block-poly(2-(4,6-dichlorotriazin-2-yl]oxy)ethyl methacrylate) (P3HT-b-PDCTMA) has been obtained via the combination of Grignard metathesis method and organo-catalyzed atom transfer radical polymerization with a polymerization conversion of 73%. The synthesized diblock copolymer P3HT-b-PDCTMA exhibited an Mn of 17,500 g/mol, which is approximately close to the theoretical value, and with polydispersity index (Đ) of 1.42. The conjugated diblock copolymer P3HT-b-PDCTMA bears functional reactive triazine chloride groups that act as a platform to react with mercaptan or amine groups via efficient nucleophilic aromatic substitution reactions. The chemical structures and macromolecular characteristic properties of the “rod–coil” conjugated diblock copolymers have been characterized by FT-IR, 1H NMR spectroscopy and gel permeation chromatography. In addition, their optical properties and conformation have been investigated via UV–Vis spectroscopy.














Similar content being viewed by others
References
Allushi A, Jockusch S, Yilmaz G, Yagci Y (2016) Photoinitiated metal-free controlled/living radical polymerization using polynuclear aromatic hydrocarbons. Macromolecules 49(20):7785–7792. https://doi.org/10.1021/acs.macromol.6b01752
Altintas O, Tunca U (2011) Synthesis of terpolymers by click reactions. Chem Asian J 6:2584–2591. https://doi.org/10.1002/asia.201100138
Barner-Kowollik C, Du Prez FE, Espeel P, Hawker CJ, Junkers T, Schlaad H, Van Camp W (2011) “Clicking” polymers or just efficient linking: what is the difference? Angew Chem Int Ed 50(1):60–62. https://doi.org/10.1002/anie.201003707
Blotny G (2006) Recent applications of 2,4,6-trichloro-1,3,5-triazine and its derivatives in organic synthesis. Tetrahedron 62(41):9507–9522. https://doi.org/10.1016/j.tet.2006.07.039
Böckmann M, Schemme T, de Jong DH, Denz C, Heuer A, Doltsinis NL (2015) Structure of P3HT crystals, thin films, and solutions by UV/Vis spectral analysis. Phys Chem Chem Phys 17(43):28616–28625. https://doi.org/10.1039/C5CP03665H
Britze A, Möllmann V, Grundmeier G, Luftmann H, Kuckling D (2011) Synthesis of blockcopolymers P3HT-b-PS using a combination of grignard-metathesis and nitroxide-mediated radical polymerization. Macromol Chem Phys 212(7):679–690. https://doi.org/10.1002/macp.201000597
Chen Y-C, Lin Y-C, Hsieh H-C, Hsu L-C, Yang W-C, Isono T, Satoh T, Chen W-C (2021) Improving the performance of photonic transistor memory devices using conjugated block copolymers as a floating gate. J Mater Chem C 9(4):1259–1268. https://doi.org/10.1039/D0TC05326K
Choi H-N, Yang H-S, Chae J-H, Choi T-L, Lee I-H (2020) Synthesis of conjugated rod–coil block copolymers by RuPhos Pd-catalyzed Suzuki-Miyaura catalyst-transfer polycondensation: initiation from coil-type polymers. Macromolecules 53(13):5497–5503. https://doi.org/10.1021/acs.macromol.0c00949
Dadashi-Silab S, Doran S, Yagci Y (2016) Photoinduced electron transfer reactions for macromolecular syntheses. Chem Rev 116(17):10212–10275. https://doi.org/10.1021/acs.chemrev.5b00586
Dai C-A, Yen W-C, Lee Y-H, Ho C-C, Su W-F (2007) Facile synthesis of well-defined block copolymers containing regioregular poly(3-hexyl thiophene) via anionic macroinitiation method and their self-assembly behavior. J Am Chem Soc 129(36):11036–11038. https://doi.org/10.1021/ja0733991
Daripa S, Khawas K, Sharma A, Kumar A, Pal B, Das S, Jit S, Kuila BK (2020) Simple and direct synthetic route to a rod–coil conjugated block copolymer from either a rod or coil block using a single bifunctional initiator: a solvent dependent self-assembly and field effect mobility study. ACS Appl Polym Mater 2(3):1283–1293. https://doi.org/10.1021/acsapm.9b01158
Daripa S, Khawas K, Behere RP, Verma R, Kuila BK (2021) Efficient moisture-induced energy harvesting from water-soluble conjugated block copolymer-functionalized reduced graphene oxide. ACS Omega 6(11):7257–7265. https://doi.org/10.1021/acsomega.0c03717
de Cuendias A, Le Hellaye M, Lecommandoux S, Cloutet E, Cramail H (2005) Synthesis and self-assembly of polythiophene-based rod–coil and coil–rod–coil block copolymers. J Mater Chem 15(32):3264–3267. https://doi.org/10.1039/B508490C
Dumele O, Chen J, Passarelli JV, Stupp SI (2020) Supramolecular energy materials. Adv Mater 32(17):1907247. https://doi.org/10.1002/adma.201907247
Ferretti AM, Diterlizzi M, Porzio W, Giovanella U, Ganzer L, Virgili T, Vohra V, Arias E, Moggio I, Scavia G, Destri S, Zappia S (2022) Rod–coil block copolymer: fullerene blend water-processable nanoparticles: how molecular structure addresses morphology and efficiency in NP-OPVs. Nanomaterials 12(1):84
Grancharov G, Coulembier O, Surin M, Lazzaroni R, Dubois P (2010) Stereocomplexed materials based on poly(3-hexylthiophene)-b-poly(lactide) block copolymers: synthesis by organic catalysis, thermal properties, and microscopic morphology. Macromolecules 43(21):8957–8964. https://doi.org/10.1021/ma1012336
Grate JW, Mo K-F, Daily MD (2016) Triazine-based sequence-defined polymers with side-chain diversity and backbone-backbone interaction motifs. Angew Int Ed Chem 55(12):3925–3930. https://doi.org/10.1002/anie.201509864
Hsu L-C, Isono T, Lin Y-C, Kobayashi S, Chiang Y-C, Jiang D-H, Hung C-C, Ercan E, Yang W-C, Hsieh H-C, Tajima K, Satoh T, Chen W-C (2021) Stretchable OFET memories: tuning the morphology and the charge-trapping ability of conjugated block copolymers through soft segment branching. ACS Appl Mater Interfaces 13(2):2932–2943. https://doi.org/10.1021/acsami.0c18820
Hufnagel M, Fischer M, Thurn-Albrecht T, Thelakkat M (2015) Donor–acceptor block copolymers carrying pendant PC71BM fullerenes with an ordered nanoscale morphology. Polym Chem 6(5):813–826. https://doi.org/10.1039/C4PY01357C
Inagaki S, Higashihara T (2022) Synthesis of an ABC triblock copolymer by a bilateral click reaction using α, ω-bifunctionalized poly(3-hexylthiophene) as an inner segment. Polym Chem 13(24):3613–3618. https://doi.org/10.1039/D2PY00370H
Inomata H, Goushi K, Masuko T, Konno T, Imai T, Sasabe H, Brown JJ, Adachi C (2004) High-efficiency organic electrophosphorescent diodes using 1,3,5-triazine electron transport materials. Chem Mater 16(7):1285–1291. https://doi.org/10.1021/cm034689t
Jaymand M, Hatamzadeh M, Omidi Y (2015) Modification of polythiophene by the incorporation of processable polymeric chains: recent progress in synthesis and applications. Prog Polym Sci 47:26–69. https://doi.org/10.1016/j.progpolymsci.2014.11.004
Kang S, Kim G-H, Park S-J (2022) Conjugated block copolymers for functional nanostructures. Acc Chem Res 55(16):2224–2234. https://doi.org/10.1021/acs.accounts.2c00244
Kim JY, Lee K, Coates NE, Moses D, Nguyen T-Q, Dante M, Heeger AJ (2007) Efficient tandem polymer solar cells fabricated by all-solution processing. Science (new York, NY) 317(5835):222–225. https://doi.org/10.1126/science.1141711
Kim Y, Kim HJ, Kim J-S, Yun H, Park H, Han J, Kim BJ (2018) Modulating regioregularity of poly(3-hexylthiophene)-based amphiphilic block copolymers to control solution assembly from nanowires to micelles. Chem Mater 30(21):7912–7921. https://doi.org/10.1021/acs.chemmater.8b03510
Kim P, Kim EY, Han SG, Lee J, Kang S, Park C, Cho K, Moon HC, Kim JK (2022) Enhanced vertical hole mobility through end-on chain orientation of poly(3-hexylthiophene)-based diblock copolymers by microphase separation. Macromolecules 55(14):6160–6166. https://doi.org/10.1021/acs.macromol.2c00791
Kubo T, Figg CA, Swartz JL, Brooks WLA, Sumerlin BS (2016) Multifunctional homopolymers: postpolymerization modification via sequential nucleophilic aromatic substitution. Macromolecules 49(6):2077–2084. https://doi.org/10.1021/acs.macromol.6b00181
Kubo T, Bentz KC, Powell KC, Figg CA, Swartz JL, Tansky M, Chauhan A, Savin DA, Sumerlin BS (2017) Modular and rapid access to amphiphilic homopolymers via successive chemoselective post-polymerization modification. Polym Chem 8(39):6028–6032. https://doi.org/10.1039/C7PY01585B
Laiho A, Nguyen HT, Sinno H, Engquist I, Berggren M, Dubois P, Coulembier O, Crispin X (2013) Amphiphilic poly(3-hexylthiophene)-based semiconducting copolymers for printing of polyelectrolyte-gated organic field-effect transistors. Macromolecules 46(11):4548–4557. https://doi.org/10.1021/ma400527z
Lim J, Simanek EE (2012) Triazine dendrimers as drug delivery systems: from synthesis to therapy. Adv Drug Deliv Rev 64(9):826–835. https://doi.org/10.1016/j.addr.2012.03.008
Liu C-L, Lin C-H, Kuo C-C, Lin S-T, Chen W-C (2011) Conjugated rod–coil block copolymers: synthesis, morphology, photophysical properties, and stimuli-responsive applications. Prog Polym Sci 36(5):603–637. https://doi.org/10.1016/j.progpolymsci.2010.07.008
Lin J-C, Lee W-Y, Kuo C-C, Li C, Mezzenga R, Chen W-C (2012) Synthesis, morphology, and field-effect transistor characteristics of new crystalline–crystalline diblock copolymers of poly(3-hexylthiophene-block-steryl acrylate). J Polym Sci A Polym Chem 50(4):686–695. https://doi.org/10.1002/pola.25078
Liu W, Wang Q-Q, Wang Y, Huang Z-T, Wang D-X (2014) Designed self-assemblies based on cooperative noncovalent interactions including anion-π, lone-pair electron–π and hydrogen bonding. RSC Adv 4(18):9339–9342. https://doi.org/10.1039/C3RA47748G
Liu X, Zhang L, Cheng Z, Zhu X (2016) Metal-free photoinduced electron transfer–atom transfer radical polymerization (PET–ATRP) via a visible light organic photocatalyst. Polym Chem 7(3):689–700. https://doi.org/10.1039/C5PY01765C
Loewe RS, Ewbank PC, Liu J, Zhai L, McCullough RD (2001) Regioregular, head-to-tail coupled poly(3-alkylthiophenes) made easy by the GRIM method: investigation of the reaction and the origin of regioselectivity. Macromolecules 34(13):4324–4333. https://doi.org/10.1021/ma001677+
Lohwasser RH, Thelakkat M (2012) Synthesis of amphiphilic rod–coil P3HT-b-P4VP carrying a long conjugated block using NMRP and click chemistry. Macromolecules 45(7):3070–3077. https://doi.org/10.1021/ma2024733
Lu S, Fan Q-L, Chua S-J, Huang W (2003) Synthesis of conjugated−ionic block copolymers by controlled radical polymerization. Macromolecules 36(2):304–310. https://doi.org/10.1021/ma020408b
Lu S, Liu T, Ke L, Ma D-G, Chua S-J, Huang W (2005) Polyfluorene-based light-emitting rod–coil block copolymers. Macromolecules 38(20):8494–8502. https://doi.org/10.1021/ma050267o
Maity N, Ghosh R, Nandi AK (2018) Optoelectronic properties of self-assembled nanostructures of polymer functionalized polythiophene and graphene. Langmuir 34(26):7585–7597. https://doi.org/10.1021/acs.langmuir.7b04387
McCullough RD (1998) The chemistry of conducting polythiophenes. Adv Mater 10(2):93–116. https://doi.org/10.1002/(SICI)1521-4095(199801)10:2%3c93::AID-ADMA93%3e3.0.CO;2-F
Miyake GM, Theriot JC (2014) Perylene as an organic photocatalyst for the radical polymerization of functionalized vinyl monomers through oxidative quenching with alkyl bromides and visible light. Macromolecules 47(23):8255–8261. https://doi.org/10.1021/ma502044f
Miyakoshi R, Yokoyama A, Yokozawa T (2005) Catalyst-transfer polycondensation mechanism of Ni-catalyzed chain-growth polymerization leading to well-defined poly(3-hexylthiophene). J Am Chem Soc 127(49):17542–17547. https://doi.org/10.1021/ja0556880
Nardo D, Akers CM, Cheung NE, Isom CM, Spaude JT, Pack DW, Venditto VJ (2021) Cyanuric chloride as the basis for compositionally diverse lipids. RSC Adv 11(40):24752–24761. https://doi.org/10.1039/D1RA02425F
Nega AD, Pefkianakis EK, Vougioukalakis GC, Glynos E, Sakellariou G (2016) Synthesis of P3HT-b-PS donor-acceptor diblock copolymer carrying pendant fullerenes at precise positions along the PS block. Eur Polym J 83:148–160. https://doi.org/10.1016/j.eurpolymj.2016.08.014
Nguyen TA, Nguyen TT, Nguyen L-TT, Van Le T, Nguyen HT (2016) Synthesis and optical investigation of amphiphilic diblock copolymers containing regioregular poly(3-hexylthiophene) via post-polymerization modification. Synth Met 217:172–184. https://doi.org/10.1016/j.synthmet.2016.03.035
Nguyen TH, Nguyen L-TT, Nguyen VQ, Ngoc Tan Phan L, Zhang G, Yokozawa T, Thuy Thi Phung D, Tran Nguyen H (2018) Synthesis of poly(3-hexylthiophene) based rod–coil conjugated block copolymers via photoinduced metal-free atom transfer radical polymerization. Polym Chem 9(18):2484–2493. https://doi.org/10.1039/C8PY00361K
Nguyen D-T, Sharma S, Chen S-A, Komarov PV, Ivanov VA, Khokhlov AR (2021) Polymer–quantum dot composite hybrid solar cells with a bi-continuous network morphology using the block copolymer poly(3-hexylthiophene)-b-polystyrene or its blend with poly(3-hexylthiophene) as a donor. Mater Adv 2(3):1016–1023. https://doi.org/10.1039/D0MA00770F
Ogita K, Yomogita N, Otep S, Michinobu T (2022) Facile access to coil–rod–coil-type block copolymers by CuAAC-based macromolecular clicking. Polym J. https://doi.org/10.1038/s41428-022-00714-z
Otep S, Tseng Y-C, Yomogita N, Chang J-F, Chueh C-C, Michinobu T (2022) Coil–rod–coil triblock copolymers synthesized by macromolecular clicking and their compatibilizer effects in all-polymer solar cells. J Mater Chem C 10(1):346–359. https://doi.org/10.1039/D1TC04948H
Ouhib F, Desbief S, Lazzaroni R, De Winter J, Gerbaux P, Jérôme C, Detrembleur C (2012) Thermally induced coupling of poly(thiophene)-based block copolymers prepared by grignard metathesis polymerization: a straightforward route toward highly regioregular multiblock conjugated copolymers. Macromolecules 45(17):6796–6806. https://doi.org/10.1021/ma3009405
Pan X, Lamson M, Yan J, Matyjaszewski K (2015) Photoinduced metal-free atom transfer radical polymerization of acrylonitrile. ACS Macro Lett 4(2):192–196. https://doi.org/10.1021/mz500834g
Park S-J, Kang S-G, Fryd M, Saven JG, Park S-J (2010) Highly tunable photoluminescent properties of amphiphilic conjugated block copolymers. J Am Chem Soc 132(29):9931–9933. https://doi.org/10.1021/ja1004569
Park M, Kang S, Nam C, Narasimha K, Lee WB, Park S-J (2022) Magnetic field-induced self-assembly of conjugated block copolymers and nanoparticles at the air-water interface. ACS Appl Mater Interfaces 14(6):8266–8273. https://doi.org/10.1021/acsami.1c22535
Pathiranage TMSK, Ma Z, Udamulle Gedara CM, Pan X, Lee Y, Gomez ED, Biewer MC, Matyjaszewski K, Stefan MC (2021) Improved self-assembly of P3HT with pyrene-functionalized methacrylates. ACS Omega 6(41):27325–27334. https://doi.org/10.1021/acsomega.1c04176
Peng J, Han Y (2020) Recent advances in conjugated polythiophene-based rod–rod block copolymers: from morphology control to optoelectronic applications. Giant 4:100039. https://doi.org/10.1016/j.giant.2020.100039
Pennetta C, Bono N, Ponti F, Bellucci MC, Viani F, Candiani G, Volonterio A (2021) Multifunctional neomycin-triazine-based cationic lipids for gene delivery with antibacterial properties. Bioconjug Chem 32(4):690–701. https://doi.org/10.1021/acs.bioconjchem.0c00616
Qi R, Qi W, Zhang Y, Liu B, Wang J, Li H, Yuan H, Xie S (2022) Fabrication of multilayered two-dimensional micelles and fibers by controlled self-assembly of rod–coil block copolymers. Polymers 14(19):4125
Rothmann MM, Haneder S, Da Como E, Lennartz C, Schildknecht C, Strohriegl P (2010) Donor-substituted 1,3,5-triazines as host materials for blue phosphorescent organic light-emitting diodes. Chem Mater 22(7):2403–2410. https://doi.org/10.1021/cm9033879
Segalman RA, McCulloch B, Kirmayer S, Urban JJ (2009) Block copolymers for organic optoelectronics. Macromolecules 42(23):9205–9216. https://doi.org/10.1021/ma901350w
Seo D, Park J, Shin TJ, Yoo PJ, Park J, Kwak K (2015) Bathochromic shift in absorption spectra of conjugated polymer nanoparticles with displacement along backbones. Macromol Res 23(6):574–577. https://doi.org/10.1007/s13233-015-3078-1
Shanmugam S, Boyer C (2016) Organic photocatalysts for cleaner polymer synthesis. Science (new York, NY) 352(6289):1053–1054. https://doi.org/10.1126/science.aaf7465
Sharma A, El-Faham A, de la Torre BG, Albericio F (2018) Exploring the orthogonal chemoselectivity of 2,4,6-trichloro-1,3,5-triazine (TCT) as a trifunctional linker with different nucleophiles: rules of the game. Front Chem 6:345. https://doi.org/10.3389/fchem.2018.00516
Shi B, Shen D, Li W, Wang G (2022) Self-assembly of copolymers containing crystallizable blocks: strategies and applications. Macromol Rapid Commun 43(14):2200071. https://doi.org/10.1002/marc.202200071
Stefan MC, Bhatt MP, Sista P, Magurudeniya HD (2012) Grignard metathesis (GRIM) polymerization for the synthesis of conjugated block copolymers containing regioregular poly(3-hexylthiophene). Polym Chem 3(7):1693–1701. https://doi.org/10.1039/C1PY00453K
Su Y-A, Maebayashi N, Fujita H, Lin Y-C, Chen C-I, Chen W-C, Michinobu T, Chueh C-C, Higashihara T (2020) Development of block copolymers with poly(3-hexylthiophene) segments as compatibilizers in non-fullerene organic solar cells. ACS Appl Mater Interfaces 12(10):12083–12092. https://doi.org/10.1021/acsami.9b22531
Thompson BC, Fréchet JMJ (2008) Polymer-fullerene composite solar cells. Angew Chem Int Ed 47(1):58–77. https://doi.org/10.1002/anie.200702506
Tsuchiya K, Ando K, Shimomura T, Ogino K (2016) Synthesis and characterization of poly(3-hexylthiophene)-block-poly(dimethylsiloxane) for photovoltaic application. Polymer 92:125–132. https://doi.org/10.1016/j.polymer.2016.03.092
Tu T-H, Chan Y-T (2020) Synthesis of terpyridine end-modified polystyrenes through ATRP for facile construction of metallo-supramolecular P3HT-b-PS diblock copolymers. Polymers 12(12):2842
Vineberg JG, Zuniga ES, Kamath A, Chen Y-J, Seitz JD, Ojima I (2014) Design, synthesis, and biological evaluations of tumor-targeting dual-warhead conjugates for a taxoid-camptothecin combination chemotherapy. J Med Chem 57(13):5777–5791. https://doi.org/10.1021/jm500631u
Wagner D, Hoffmann ST, Heinemeyer U, Münster I, Köhler A, Strohriegl P (2013) Triazine based bipolar host materials for blue phosphorescent OLEDs. Chem Mater 25(18):3758–3765. https://doi.org/10.1021/cm4023216
Wang D-X, Wang M-X (2013) Anion−π interactions: generality, binding strength, and structure. J Am Chem Soc 135(2):892–897. https://doi.org/10.1021/ja310834w
Wang G, Swensen J, Moses D, Heeger AJ (2003) Increased mobility from regioregular poly(3-hexylthiophene) field-effect transistors. J Appl Phys 93(10):6137–6141. https://doi.org/10.1063/1.1568526
Wang X, Zhao C, Li Y, Lin Z, Xu H (2020) A facile and highly efficient route to amphiphilic star-like rod–coil block copolymer via a combination of atom transfer radical polymerization with thiolene click chemistry. Macromol Rapid Commun 41(5):1900540. https://doi.org/10.1002/marc.201900540
Wei Q, Nishizawa T, Tajima K, Hashimoto K (2008) Self-organized buffer layers in organic solar cells. Adv Mater 20(11):2211–2216. https://doi.org/10.1002/adma.200792876
Wu W-N, Tu T-H, Pai C-H, Cheng K-H, Tung S-H, Chan Y-T, Liu C-L (2022) Metallo-supramolecular rod–coil block copolymer thin films for stretchable organic field effect transistor application. Macromolecules. https://doi.org/10.1021/acs.macromol.2c00957
Xu L, Xu X-H, Liu N, Zou H, Wu Z-Q (2018) A facile synthetic route to multifunctional poly(3-hexylthiophene)-b-poly(phenyl isocyanide) copolymers: from aggregation-induced emission to controlled helicity. Macromolecules 51(19):7546–7555. https://doi.org/10.1021/acs.macromol.8b01478
Yang Z, Fu K, Yu J, Shi X, Zhou P, Cheng Z (2018) Facile preparation of nanoporous C60/P3HT thin films from PLA-b-C60-b-P3HT triblock copolymers. Appl Surf Sci 458:70–76. https://doi.org/10.1016/j.apsusc.2018.07.076
Zacharie B, Abbott SD, Bienvenu J-F, Cameron AD, Cloutier J, Duceppe J-S, Ezzitouni A, Fortin D, Houde K, Lauzon C, Moreau N, Perron V, Wilb N, Asselin M, Doucet A, Fafard M-È, Gaudreau D, Grouix B, Sarra-Bournet F, St-Amant N, Gagnon L, Penney CL (2010) 2,4,6-Trisubstituted triazines as protein a mimetics for the treatment of autoimmune diseases. J Med Chem 53(3):1138–1145. https://doi.org/10.1021/jm901403r
Zhang X, Lian X, Liu L, Zhang J, Zhao H (2008) Synthesis of comb copolymers with pendant chromophore groups based on RAFT polymerization and click chemistry and formation of electron donor–acceptor supramolecules. Macromolecules 41(21):7863–7869. https://doi.org/10.1021/ma801405j
Zhang Y, Shaikh H, Sneyd AJ, Tian J, Xiao J, Blackburn A, Rao A, Friend RH, Manners I (2021) efficient energy funneling in spatially tailored segmented conjugated block copolymer nanofiber-quantum dot or rod conjugates. J Am Chem Soc 143(18):7032–7041. https://doi.org/10.1021/jacs.1c01571
Zhang J, Li S, Yin Y, Xiang L, Xu F, Mai Y (2022) One-dimensional helical nanostructures from the hierarchical self-assembly of an achiral “rod–coil” alternating copolymer. Macromol Rapid Commun 43(21):2200437. https://doi.org/10.1002/marc.202200437
Acknowledgements
This research is funded by Vietnam National University Ho Chi Minh City (VNU-HCM) under Grant Number C2021-20-17 and NVTX2022: TX2022-20a-1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
I certify that there is no actual or potential conflict of interest in relation to this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bui, T.T., Nguyen, T.H., Tran, H.L. et al. Synthesis of rod–coil conjugated diblock copolymers, poly(3-hexylthiophene)-block-poly(2-(4,6-dichlorotriazin-2-yl]oxy)ethyl methacrylate) and click chemistry. Chem. Pap. 77, 4439–4456 (2023). https://doi.org/10.1007/s11696-023-02793-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-023-02793-5