Abstract
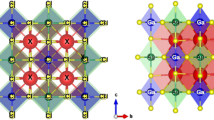
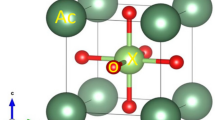
This work aims at synthesizing by reaction in the solid state orthophosphate type compounds usable in different applications (electrochemical performances, laser materials, magnetic materials, ionic conductors, potential in optics, catalysts, sensors, ceramics, etc.). The optimal operating conditions have been determined. The characterization of the materials obtained was carried out from a structural point of view by X-ray diffraction, Raman, and infrared spectroscopies, and morphologically, by scanning electron microscopy coupled with analysis by energy dispersive X-ray spectroscopy and transmission electron microscopy. The Rietveld procedure was used for the refinement investigation, and the findings reveal that the observed and calculated patterns are in excellent agreement. In the complete composition range (0 ≤ x ≤ 2), the Ba2 − xPbxSr(PO4)2 compounds crystallize in the hexagonal system at space group R \(\overline{3 }\) m with multiple units per crystal lattice Z = 3 and produce a solid solution. In solid solutions, the a and c lattice parameters varied, with a reduction in the a and c parameters proportional to the radius of the corresponding replaced ions. The M(1) and M(2) positions in the lacunar anionic orthophosphate structure were shown to be preferentially occupied by Pb2+/Ba2+/Sr2+ ions in solid solutions.
The vibrational spectra of this solid solution series of crystalline orthophosphate are assigned by comparison with other orthophosphate materials and interpreted based on factor group analysis in space group R \(\overline{3 }\) m (D3d5). Raman and IR spectroscopy studies give results justifying the crystal symmetry already demonstrated by X-ray diffraction.











Similar content being viewed by others
References
Amaterz E, Bouddouch A, Chennah A et al (2020) Heat treatment effect on the structure and morphology of strontium monoacid orthophosphate thin films. Materials Today: Proceedings 22:45–47. https://doi.org/10.1016/j.matpr.2019.08.070
Azdouz M (2018) Synthesis and structural characterization of Ba2Pb(PO4)2 and BaPb2(PO4)2 and study of thermal behavior of BaPb2(PO4)2 at high temperature, 17
Baaziz MB, Azdouz M, Azrour M et al (2019) Elaboration, vibrational study and thermal behavior of Lacunar Apatites NaPb3−xCdxCa(PO4)3 (0 ≤ x ≤ 1). J Struct Chem 60:1285–1298. https://doi.org/10.1134/S0022476619080080
Baaziz MB, Azdouz M, Azrour M et al (2018) Elaboration, Rietveld refinements and vibrational spectroscopic studies of a new lacunar apatite series: NaPb3–x CaxCd(PO4)3 (0 ≤ x ≤ 1). J Chem Res, pp 564–571. https://doi.org/10.3184/174751918X15404020197095
Babu Ballipalli C, Rajavaram R, Naresh V et al (2021) Synthesis and photoluminescent characteristics of Sm3+-doped Ba3(PO4)2 phosphor hierarchical architectures. Mater Sci Eng B 264:114979. https://doi.org/10.1016/j.mseb.2020.114979
Badri A, Hidouri M, Wattiaux A et al (2014) Crystal structure, IR and Mössbauer spectroscopy and magnetic properties of KZnFe(PO4)2 related to the zeolite-ABW-like compounds. Mater Res Bull 55:61–66. https://doi.org/10.1016/j.materresbull.2014.04.009
Boultif A, Louër D (1991) Indexing of powder diffraction patterns for low-symmetry lattices by the successive dichotomy method. J Appl Crystallogr 24:987–993. https://doi.org/10.1107/S0021889891006441
Cao R, Yu X, Sun X et al (2014) Near-infrared emission Ba3(PO4)2:Mn5+ phosphor and potential application in vivo fluorescence imaging. Spectrochim Acta Part A Mol Biomol Spectrosc 128:671–673. https://doi.org/10.1016/j.saa.2014.02.081
Eliassi MD, Zhao W, Tan WF (2014) Effect of carbonate and phosphate ratios on the transformation of calcium orthophosphates. Mater Res Bull 55:114–120. https://doi.org/10.1016/j.materresbull.2014.03.037
Enneffati M, Louati B, Guidara K et al (2018) Crystal structure characterization and AC electrical conduction behavior of sodium cadmium orthophosphate. J Mater Sci: Mater Electron 29:171–179. https://doi.org/10.1007/s10854-017-7901-7
Ji H, Huang Z, Xia Z et al (2015) Discovery of new solid solution phosphors via cation substitution-dependent phase transition in M 3 (PO 4) 2: Eu 2+ (M = Ca/Sr/Ba) Quasi-Binary sets. J Phys Chem C 119:2038–2045. https://doi.org/10.1021/jp509743r
Kazumasa S, Masayasu T (1990) The crystal structure refinements of the strontium and barium orthophosphates. Mineral J 15:141–146. https://doi.org/10.2465/minerj.15.141
Liang H, Tao Y, Zeng Q et al (2003) The optical spectroscopic properties of rare earth-activated barium orthophosphate in VUV-Vis range. Mater Res Bull 38:797–805. https://doi.org/10.1016/S0025-5408(03)00059-X
Manoun B, Popovic L, Waal DD, Verryn SMC (2003) Rietveld refinements of a new solid solution Ba(3–x)Srx(PO4)2 (0≤x≤3). Powder Diffr 18:122–127. https://doi.org/10.1154/1.1572487
Manthiram A, Goodenough JB (1989) Lithium insertion into Fe2 (SO4) 3 frameworks. J Power Sources 26:403–408. https://doi.org/10.1016/0378-7753(89)80153-3
Mathur A, Halappa P, Shivakumara C (2018) Synthesis and characterization of Sm3+ activated La1− xGdxPO4 phosphors for white LEDs applications. J Mater Sci Mater Electron 29:19951–19964
Megaw H (1973) Crystal structures: a working approach. WB Saunders Co., Toronto
Padhi AK, Nanjundaswamy KS, Masquelier C, Goodenough JB (1997) Mapping of transition metal redox energies in phosphates with NASICON structure by Lithium intercalation. J Electrochem Soc 144:2581–2586. https://doi.org/10.1149/1.1837868
Pelczarska AJ, Stefańska D, Watras A et al (2022) Structural and luminescence behavior of nanocrystalline orthophosphate KMeY(PO4)2: Eu3+ (Me = Ca, Sr) synthesized by hydrothermal method. Materials 15:1850. https://doi.org/10.3390/ma15051850
Poort SHM, Reijnhoudt HM, Van der Kuip HOT, Blasse G (1996) Luminescence of Eu2+ in silicate host lattices with alkaline earth ions in a row. J Alloy Compd 241:75–81. https://doi.org/10.1016/0925-8388(96)02324-9
Popovic L, Manoun B, de Waal D (2002) Crystal chemistry, vibrational spectra and factor group analysis of Ba(3–x)Srx(PO4)2 (0≤x≤3) solid solution series. J Alloy Compd 343:82–89. https://doi.org/10.1016/S0925-8388(02)00138-X
Qin C, Huang Y, Shi L et al (2009) Thermal stability of luminescence of NaCaPO 4 : Eu 2+ phosphor for white-light-emitting diodes. J Phys D Appl Phys 42:185105. https://doi.org/10.1088/0022-3727/42/18/185105
Redden MJ, Buerger MJ (1969) Note on the symmetry and cell of calcium orthovanadate. Z Kristallogr 129:459–460. https://doi.org/10.1524/zkri.1969.129.5-6.459
Rodriguez-Carvajal E JC (1990) Collected Abstracts of Powder Diffraction Meeting. Toulouse, France, pp 127–128
Roisnel T, Rodríquez-Carvajal J (2001) WinPLOTR: A Windows Tool for Powder Diffraction Pattern Analysis. Mater Sci Forum 378–381:118–123. https://doi.org/10.4028/www.scientific.net/MSF.378-381.118
Salje E, Devarajan V (1981) Potts model and phase transition in lead phosphate Pb3(PO4)2. J Phys c Solid State Phys 14:L1029–L1035. https://doi.org/10.1088/0022-3719/14/33/001
Shannon RD (1976) Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr a Crystal Phys Diffraction Theor General Crystallogr 32:751–767. https://doi.org/10.1107/S0567739476001551
Tāle I, Kūlis P, Kronghauz V (1979) Recombination luminescence mechanisms in Ba3(PO4)2. J Lumin 20:343–347. https://doi.org/10.1016/0022-2313(79)90003-6
Tarte P, Thelen J (1972) Spectre vibrationnel des composéu type Ba3 (XO4) 2 et Sr3 (XO4) 2 (Xv= P, As, V, Cr, Mn). Spectrochim Acta, Part A 28:5–14
Tong M, Liang Y, Yan P et al (2015a) Synthesis and luminescence properties of a bluish-green emitting phosphor Ba3(PO4)2: Ce3+, Tb3+. Opt Laser Technol 75:221–228. https://doi.org/10.1016/j.optlastec.2015.07.007
Tong M, Zhao J, Liang Y et al (2015b) Synthesis and characterization of micro/nano-structured BaHPO 4 /Ba 3 (PO 4) 2 /Ba 5 (PO 4) 3 OH phases and their luminescence. RSC Adv 5:27517–27525. https://doi.org/10.1039/C5RA01420D
Yi L, Zhou L, Gong F et al (2010) Preparation of SrZn2(PO4)2:Eu2+, Mn2+ phosphor and its photoluminescent properties. Mater Sci Eng B 172:132–135. https://doi.org/10.1016/j.mseb.2010.04.035
Yokota H, Matsumoto S, Salje EKH, Uesu Y (2019) Polar nature of domain boundaries in purely ferroelastic P b 3 ( P O 4 ) 2 investigated by second harmonic generation microscopy. Phys Rev B 100:024101. https://doi.org/10.1103/PhysRevB.100.024101
Yu R, Noh HM, Moon BK et al (2013) Synthesis and luminescence properties of a novel red-emitting phosphor Ba3La(PO4)3:Eu3+ for solid-state lighting. J Alloy Compd 576:236–241. https://doi.org/10.1016/j.jallcom.2013.04.150
Zachariasen WH (1948) The crystal structure of the normal orthophosphates of barium and strontium. Acta Cryst 1:263–265. https://doi.org/10.1107/S0365110X48000697
Zhai S, Lin C-C, Xue W (2014) Raman spectra of Sr3(PO4)2 and Ba3(PO4)2 orthophosphates at various temperatures. Vib Spectrosc 70:6–11. https://doi.org/10.1016/j.vibspec.2013.10.002
Zhu L, Huang Z, Molokeev MS et al (2016) Influence of cation substitution on the crystal structure and luminescent properties in apatite structural Ba4.97−Sr (PO4)3Cl:0.03Eu2+ phosphors. Chem Phys Lett 658:248–253. https://doi.org/10.1016/j.cplett.2016.06.061
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ben Baaziz, M., Ghibate, R., Merzougui, A. et al. Synthesis, structural characterizations and vibrational spectroscopy studies of Ba2 −xPbxSr(PO4)2; (0 ≤ x ≤ 2). Chem. Pap. 77, 2905–2915 (2023). https://doi.org/10.1007/s11696-023-02674-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-023-02674-x