Abstract
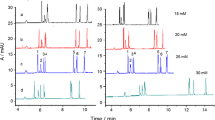
Virginiamycin antibiotic is used in Mexico for animal growth promotion but has been banned in Europe due to risk of resistance development. Giving that monitoring of antibiotics is critical, the objective of this work was to develop an analytical method based on capillary electrophoresis and liquid-liquid extraction, to quantify virginiamycin antibiotic in livestock wastes. Linearity, precision, bias and extraction parameters were evaluated following ISO/IEC 17025 procedures. Method and extraction validation were satisfactorily, with average values of absolute recovery AR = 86%, extraction efficiency EE = 87%, and matrix effects ME = -14%, RSD<15% for concentrations as low as 50 \(\upmu \)g/L.






Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author C.Díaz-Quiroz, upon reasonable request.
References
Aarestrup FM, Seyfarth AM, Emborg HD et al (2001) Effect of abolishment of the use of antimicrobial agents for growth promotion on occurrence of antimicrobial resistance in fecal enterococci from food animals in denmark. Antimicrob Agents Chemother 45(7):2054–9. https://doi.org/10.1128/aac.45.7.2054-2059.2001
Anadón A, Martínez-Larrañaga MR (1999) Residues of antimicrobial drugs and feed additives in animal products: regulatory aspects. Livest Prod Sci 59(2):183–198. https://doi.org/10.1016/S0301-6226(99)00026-3
Aranas AT, Guidote AM, Quirino JP (2009) Sweeping and new on-line sample preconcentration techniques in capillary electrophoresis. Anal Bioanal Chem 394(1):175–185. https://doi.org/10.1007/s00216-009-2646-7
Bellanger X, Guilloteau H, Bonot S et al (2014) Demonstrating plasmid-based horizontal gene transfer in complex environmental matrices: a practical approach for a critical review. Sci Total Environ 493:872–882. https://doi.org/10.1016/j.scitotenv.2014.06.070
Bengtsson-Palme J, Larsson DGJ (2015) Antibiotic resistance genes in the environment: prioritizing risks. Nat Rev Microbiol 13(6):396–396. https://doi.org/10.1038/nrmicro3399-c1
Butaye P, Devriese LA, Haesebrouck F (2003) Antimicrobial growth promoters used in animal feed: effects of less well known antibiotics on gram-positive bacteria. Clin Microbiol Rev 16(2):175–188. https://doi.org/10.1128/CMR.16.2.175-188.2003
Díaz-Quiroz C, Hernández-Chávez JF, Ulloa-Mercado G, et al (2020) Simultaneous extraction of antibiotic and estrogen from animal blood serum using aqueous two-phase systems as predictor of environmental impact. Environ Sci Pollut Res 27(23):28,536–28,544
Díaz-Quiroz CA, Francisco Hernández-Chávez J, Ulloa-Mercado G et al (2018) Simultaneous quantification of antibiotics in wastewater from pig farms by capillary electrophoresis. J Chromatogr B 1092:386–393. https://doi.org/10.1016/j.jchromb.2018.06.017
Flurer CL (2001) Analysis of antibiotics by capillary electrophoresis. Electrophoresis 22(19):4249–4261. https://doi.org/10.1002/1522-2683(200111)22:19<4249::AID-ELPS4249>3.0.CO;2-8
Goffic FL, Capmau ML, Bonnet D et al (1977) Plasmid-mediated pristinamycin resistance. pac iia: a new enzyme which modifies pristinamycin iia. J Antibiot (Tokyo) 30(8):665–9. https://doi.org/10.7164/antibiotics.30.665
Hammerum AM, Jensen LB, Aarestrup FM (1998) Detection of the sata gene and transferability of virginiamycin resistance in enterococcus faecium from food-animals. FEMS Microbiol Lett 168(1):145–151. https://doi.org/10.1111/j.1574-6968.1998.tb13267.x
Hernando MD, Mezcua M, Fernández-Alba AR, et al (2006) Environmental risk assessment of pharmaceutical residues in wastewater effluents, surface waters and sediments. Talanta 69(2 SPEC. ISS.):334–342. https://doi.org/10.1016/j.talanta.2005.09.037
Magnusson B (2014) The fitness for purpose of analytical methods: a laboratory guide to method validation and related topics (2014)
Martinez JL (2009) Environmental pollution by antibiotics and by antibiotic resistance determinants. Environ Pollut 157(11):2893–2902. https://doi.org/10.1016/j.envpol.2009.05.051
Martinez JL (2009) The role of natural environments in the evolution of resistance traits in pathogenic bacteria. Proc R Soc B Biol Sci 276(1667):2521–2530. https://doi.org/10.1098/rspb.2009.0320
Palmer J, Munro NJ, Landers JP (1999) A universal concept for stacking neutral analytes in micellar capillary electrophoresis. Anal Chem 71(9):1679–1687. https://doi.org/10.1021/ac981302a
Sandegren L (2014) Selection of antibiotic resistance at very low antibiotic concentrations. Upsala J Med Sci 119(2):103–107. https://doi.org/10.3109/03009734.2014.904457
SENASICA (2016) Lista de compuestos sujetos a monitoreo [policy brief]. https://www.gob.mx/cms/uploads/attachment/file/128437/2-Listadodecompuestossujetosamonitoreo.pdf
Terabe S, Otsuka K, Ichikawa K et al (1984) Electrokinetic separations with micellar solutions and open-tubular capillaries. Anal Chem 56(1):111–113. https://doi.org/10.1021/ac00265a031
Tobback K, Li YM, Pizarro NA et al (1999) Micellar electrokinetic capillary chromatography of macrolide antibiotics: separation of tylosin, erythromycin and their related substances. J Chromatogr A 857(1–2):313–320
Tu̇ma P, Jaček M, Sommerová B, et al (2022) Monitoring of amoxicilline and ceftazidime in the microdialysate of diabetic foot and serum by capillary electrophoresis with contactless conductivity detection. Electrophoresis 43(11):1129–1139. https://doi.org/10.1002/elps.202100366
Ventola CL (2015) The antibiotic resistance crisis: part 1: causes and threats. Pharm Therapeutic 40(4):277
Wegener HC (2003) Antibiotics in animal feed and their role in resistance development. Curr Opin Microbiol 6(5):439–445. https://doi.org/10.1016/j.mib.2003.09.009
Wright GD (2010) Antibiotic resistance in the environment: a link to the clinic? Curr Opin Microbiol 13(5):589–594. https://doi.org/10.1016/j.mib.2010.08.005
Wuethrich A, Haddad PR, Quirino JP (2016) Field-enhanced sample injection micelle-to-solvent stacking capillary zone electrophoresis-electrospray ionization mass spectrometry of antibiotics in seawater after solid-phase extraction. Electrophoresis 37(9):1139–1142. https://doi.org/10.1002/elps.201500448
Funding
We appreciate the financing support of Instituto Tecnológico de Sonora through the program PROFAPI-2021-0084; F. Robles would like to acknowledge the scholarship 1002055 from CONACYT.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Juan Francisco Hernández-Chávez and Jesús Fernando Robles-Castro. The first draft of the manuscript was written by Carlos Abraham Díaz-Quiroz. Gabriela Ulloa-Mercado and Carlos Díaz contributed to original concept, funding, supplied reagent and manuscript revisions and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Consent to participate
Not applicable
Consent to publish
Not applicable
Ethical approval
Not applicable
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hernández-Chávez, J.F., Robles-Castro, J.F., Díaz-Quiroz, C.A. et al. Development of a simple and sensitive method for the determination of virginiamycin M1 antibiotic by capillary electrophoresis. Chem. Pap. 76, 7419–7427 (2022). https://doi.org/10.1007/s11696-022-02412-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-022-02412-9