Abstract
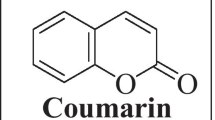
Natural compounds are very much important as they can cure diseases naturally without harmful effects from natural sources. Coumarin is also a natural compound and a large number of coumarins almost 1300 coumarins have been recognized from natural sources, mainly from green plants. Substitution on coumarins is responsible for their therapeutic, biochemical, and pharmacological applications. Coumarins are present in plants and can also be prepared in the laboratory. Coumarin, a drug is used in heat problems as it is an anti-coagulant i.e., it stops and helps to cure blood clots of blood vessels. This article is based upon the investigation of the absorption spectra of coumarin and its derivatives. The absorption spectra involve the study of UV–Visible spectroscopy and fluorescence spectroscopy. Solvatochromic shifts and the effects of the salts on fluorescence spectra and UV–Visible spectra will also be studied.















Similar content being viewed by others
References
Abu-Eittah RH, El-Tawil BAH (1985) The electronic absorption spectra of some coumarins. A molecular orbital treatment. Canadian J Chem 63(6):1173–1179
Ayyanger NR, Sirnivasan KV, Daniel T (1991) polycyclic compounds Part IV. Synthesis laser characteristics and dyeing behavior of 7-diethylamino-2H-I-benzopyran-2-ones. Dyes Pigment 16(3):197–204. https://doi.org/10.1016/0143-7208(91)85010-6
Azim SA, Al-Hazmy SM, Ebeid EM, El-Daly SA (2005) A new coumarin laser dye 3-(benzothiazol-2-yl)-7-hydroxycoumarin. Opt Laser Technol 37(3):245–249. https://doi.org/10.1016/j.optlastec.2004.04.003
Bandaranayake WM, Selliah SS, Sultanbawa MU, Games DE (1975) Xanthones and 4-phenylcoumarins of Mesua thwaitesii. Phytochemistry 14(1):265–269. https://doi.org/10.1016/0031-9422(75)85052-7
Basile A, Sorbo S, Spadaro V, Bruno M, Maggio A, Faraone N, Rosselli S (2009) Antimicrobial and antioxidant activities of coumarins from the roots of Ferulago campestris (Apiaceae). Molecules 14(3):939. https://doi.org/10.3390/molecules14030939
Bourgaud F, Hehn A, Larbat R, Doerper S, Gontier E, Kellner S, Matern U (2006) Biosynthesis of coumarins in plants: a major pathway still to be unravelled for cytochrome P450 enzymes. Phytochem Rev 5(2–3):293–308. https://doi.org/10.1007/s11101-006-9040-2
Chain EÁ (1958) Chemistry and biochemistry of antibiotics. Ann Rev Biochem 27(1):167–222. https://doi.org/10.1146/annurev.bi.27.070158.001123
Chiang CC, Cheng MJ, Peng CF, Huang HY, Chen IS (2010) A novel dimeric coumarin analog and antimycobacterial constituents from Fatoua pilosa. Chem Biodivers 7(7):1728–1736. https://doi.org/10.1002/cbdv.200900326
Christie RM (1994) Pigments, dyes and fluorescent brightening agents for plastics: an overview. Polym Int 34(4):351–361. https://doi.org/10.1002/pi.1994.210340401
Christie RM, Lui CH (2000) Studies of fluorescent dyes: part 2. An investigation of the synthesis and electronic spectral properties of substituted 3-(2′-benzimidazolyl) coumarins. Dyes Pigment 47(1–2):79–89. https://doi.org/10.1016/S0143-7208(00)00066-8
Dean FM, Parton B (1969) The structure and synthesis of (+)-obliquin. J Chem Soc C Org 4:526–531. https://doi.org/10.1039/j39690000526
Donovalová J, Cigáň M, Stankovičová H, Gašpar J, Danko M, Gáplovský A, Hrdlovič P (2012) Spectral properties of substituted coumarins in solution and polymer matrices. Molecules 17(3):3259–3276. https://doi.org/10.3390/molecules17033259
Drexhage KH (1973) Structure and properties of laser dyes. Dye Lasers. https://doi.org/10.1007/978-3-662-11579-4_4
Drexhage KH (1976) Fluorescence efficiency of laser dyes. J Res Natl Bureau Stand Sect A Phys Chem 80(3):421. https://doi.org/10.6028/jres.080A.044
Drlica K, Snyder M (1978) Superhelical Escherichia coli DNA: relaxation by coumermycin. J Mol Biol 120(2):145–154. https://doi.org/10.1016/0022-2836(78)90061-X
El-Sheikh M, Azim S, Abou Saif A, Okba E (2017) A spectroscopic and photochemical behavior of some coumarin derivatives. Delta J Sci 38:183
Fadda AA, Refat HM, Zaki ME (2000) Utility of sulphones in heterocyclic synthesis: synthesis of some pyridine, chromene and thiophene derivatives. Molecules 5(5):701–709. https://doi.org/10.3390/50500701
Farooq S (2005) 555 Medicinal Plants. Field and Laboratory Manual. International Book Distributors
Fischer A, Cremer CS, Stelzer EH (1995) Fluorescence of coumarins and xanthenes after two-photon absorption with a pulsed titanium–sapphire laser. Appl Opt 34(12):1989–2003. https://doi.org/10.1364/AO.34.001989
Gellert M, O’Dea MH, Itoh T, Tomizawa JI (1976) Novobiocin and coumermycin inhibit DNA supercoiling catalyzed by DNA gyrase. Proceed Nat Acad Sci 73(12):4474–4478. https://doi.org/10.1073/pnas.73.12.4474
Goodwin RH, Pollock BM, Studies on roots. I, (1954) Properties and distribution of fluorescent constituents in Avena roots. Am J Bot 1:516–520. https://doi.org/10.2307/2438863
Gordo J, Avó J, Parola AJ, Lima JC, Pereira A, Branco PS (2011) Convenient synthesis of 3-vinyl and 3-styryl coumarins. Org Lett 13(19):5112–5115. https://doi.org/10.1021/ol201983
Grynkiewicz G, Poenie M, Tsien RY (1985) A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem 260(6):3440–3450. https://doi.org/10.1016/S0021-9258(19)83641-4
Hara K, Sato T, Katoh R, Furube A, Ohga Y, Shinpo A, Suga S, Sayama K, Sugihara H, Arakawa H (2003) Molecular design of coumarin dyes for efficient dye-sensitized solar cells. J Phys Chem B 107(2):597–606. https://doi.org/10.1021/jp026963x
Hara K, Wang ZS, Sato T, Furube A, Katoh R, Sugihara H, Dan-Oh Y, Kasada C, Shinpo A, Suga S (2005) Oligothiophene-containing coumarin dyes for efficient dye-sensitized solar cells. J Phys Chem B 109(32):15476–15482. https://doi.org/10.1021/jp0518557
Harborne JB (1982) the natural coumarins: occurrence, chemistry and biochemistry (book). Plant Cell Environ 5(6):435–436
Hon YS, Tseng TW, Cheng CY (2009) Electrocyclization of cis-dienals in organic synthesis: a new and versatile synthetic method for the preparation of aryl-and heteroaryl-fused coumarins. Chem Commun 37:5618–5620. https://doi.org/10.1039/B912887E
Horowitz RM, Gentili B (1960) Flavonoids of citrus IV Isolation of some aglycones from the lemon (Citrus limon). J Org Chem 25(12):2183–2187. https://doi.org/10.1021/jo01082a024
Hoult JR, Payá M (1996) Pharmacological and biochemical actions of simple coumarins: natural products with therapeutic potential. Gen Pharmacol Vasc Syst 27(4):713–722. https://doi.org/10.1016/0306-3623(95)02112-4
Hsu SF, Lee CC, Hwang SW, Chen HH, Chen CH, Hu AT (2005) Color-saturated and highly efficient top-emitting organic light-emitting devices. Thin Solid Film 478(1–2):271–4. https://doi.org/10.1016/j.tsf.2004.10.038, https://doi.org/10.1016/j.snb.2013.03.031, https://doi.org/10.1111/1365-3040.ep11611630
Hung LS, Chen CH (2002) recent progress of molecular organic electroluminescent materials and devices. Mater Sci Eng R Rep 39(5–6):143–222. https://doi.org/10.1016/S0927-796X(02)00093-1
Improta R, Barone V, Santoro F (2007) Ab initio calculations of absorption spectra of large molecules in solution: coumarin C153. Angew Chem Int Ed 46(3):405–408. https://doi.org/10.1002/anie.200602907
Iranshahi M, Askari M, Sahebkar A, Hadjipavlou LD (2009) Evaluation of antioxidant, anti-inflammatory and lipoxygenase inhibitory activities of the prenylated coumarin umbelliprenin. DARU J Pharmaceuti Sci 17(2):99–103
Irfan M, Iqbal J, Eliasson B, Ayub K, Rama UA, Khan S. U. D. J. J. O. M. S (2017) J Mol Struct 13(30):603
Jagtap AR, Satam VS, Rajule RN, Kanetkar VR (2009) The synthesis and characterization of novel coumarin dyes derived from 1, 4-diethyl-1, 2, 3, 4-tetrahydro-7-hydroxyquinoxalin-6-carboxaldehyde. Dyes Pigm 82(1):84–89. https://doi.org/10.1016/j.dyepig.2008.11.007
Jiang M, Paul N, Bieniek N, Buckup T, Hampp N, Motzkus M (2017) Photocleavage of coumarin dimers studied by femtosecond UV transient absorption spectroscopy. Phys Chem Chem Phys 19(6):4597–4606. https://doi.org/10.1039/C6CP08076F
Jones G, Jackson WR, Choi CY, Bergmark WR (1985) Solvent effects on emission yield and lifetime for coumarin laser dyes. Requirements for a rotatory decay mechanism. J Phys Chem 89(2):294–300. https://doi.org/10.1021/j100248a024
Joshi BS, Kamat VN, Saksena AK (1967) Structures of Clausenin and Clausenidin two new pyranocoumarins from the roots of Clausena heptaphylla wt. & Am. Tetrahedron 23(12):4785–4789. https://doi.org/10.1016/S0040-4020(01)92576-1
Kim TK, Lee DN, Kim HJ (2008) Highly selective fluorescent sensor for homocysteine and cysteine. Tetrahedron Lett 49(33):4879–4881. https://doi.org/10.1016/j.tetlet.2008.06.003
Kobayashi T (1978) Picosecond kinetics of 4-methylumbelliferone fluorescence observed with mode-locked laser and streak camera. J Phys Chem 82(21):2277–2281. https://doi.org/10.1021/j100510a004
Komatsu K, Urano Y, Kojima H, Nagano T (2007) Development of an iminocoumarin-based zinc sensor suitable for ratiometric fluorescence imaging of neuronal zinc. J Am Chem Soc 129(44):13447–13454. https://doi.org/10.1021/ja072432g
Kostova I (2005) Synthetic and natural coumarins as cytotoxic agents. Curr Med Chem 5(1):29–46. https://doi.org/10.2174/1568011053352550
Kostova I, Bhatia S, Grigorov P, Balkansky S, Parmar SV, Prasad KA, Saso L (2011) Coumarins as antioxidants. Curr Med Chem 18(25):3929–3951. https://doi.org/10.2174/092986711803414395
Kumar P (2018) Fundamentals and Techniques of Biophysics and Molecular biology. Pathfinder Publication unit of PAPL
Lacy A, O’Kennedy R (2004) Studies on coumarins and coumarin-related compounds to determine their therapeutic role in the treatment of cancer. Curr Pharm Des 10(30):3797–3811. https://doi.org/10.2174/1381612043382693
Lee KS, Kim TK, Lee JH, Kim HJ, Hong JI (2008) Fluorescence turn-on probe for homocysteine and cysteine in water. Chem Commun 46:6173–6175. https://doi.org/10.1039/B814581D
Lin W, Yuan L, Cao Z, Feng Y, Long L (2009) A sensitive and selective fluorescent thiol probe in water based on the conjugate 1,4-addition of thiols to α,β-unsaturated ketones. Chem A Eur J 15(20):5096–5103. https://doi.org/10.1002/chem.200802751
Lin W, Yuan L, Cao Z, Feng Y, Song J (2010) Through-bond energy transfer cassettes with minimal spectral overlap between the donor emission and acceptor absorption: Coumarin–rhodamine dyads with large pseudo-stokes shifts and emission shifts. Angew Chem Int Ed 49(2):375–379. https://doi.org/10.1002/anie.200904515
Maity D, Karthigeyan D, Kundu TK, Govindaraju T (2013) FRET-based rational strategy for ratiometric detection of Cu2+ and live cell imaging. Sens Actuators B Chem 176:831–837. https://doi.org/10.1016/j.snb.2012.09.071
Masrani KV, Rama HS, Bafna SL (1974) Ultraviolet absorption spectra: some substituted coumarins. J Appl Chem Biotech 24(6):331–341. https://doi.org/10.1002/jctb.2720240604
Mattoo BN (1956) Absorption and fluorescence spectra of coumarins. Trans Faraday Soc 52:1184–1194. https://doi.org/10.1039/TF9565201184
Mead JA, Smith JN, Williams RT (1958) Studies in detoxication. 72. The metabolism of coumarin and of o-coumaric acid. Biochem J 68(1):67–74. https://doi.org/10.1042/bj0680061
Minta A, Kao JP, Tsien RY (1989) Fluorescent indicators for cytosolic calcium based on rhodamine and fluorescein chromophores. J Biol Chem 264(14):8171–8178. https://doi.org/10.1016/S0021-9258(18)83165-9
Moylan CR (1994) Molecular hyperpolarizabilities of coumarin dyes. J Phys Chem 98(51):13513–13516. https://doi.org/10.1021/j100102a014
Nedumpara RJ, Thomas KJ, Jayasree VK, Girijavallabhan CP, Nampoori VP, Radhakrishnan P (2007) Study of solvent effect in laser emission from coumarin 540 dye solution. Appl Opt 46(21):4786–4792. https://doi.org/10.1364/AO.46.004786
Newman RA, Chen W, Madden TL (1998) Pharmaceutical properties of related calanolide compounds with activity against human immunodeficiency virus. J Pharm Sci 87(9):1077–1080. https://doi.org/10.1021/js980122d
Nikhil B, Shikha B, Anil P, Prakash N (2012) Diverse pharmacological activities of 3-substituted coumarins: A review. Int Res J Pharm 3(7):24–29
Nirupama JM, Khanapurmath NI, Chougala LS, Kulkarni MV, Kadadevarmath JS (2019) Effect of stereo electronic factors of coumarin derivatives during their interaction with TiO2 nanoparticles. J Mol Liq 291:111–266
Pagona G, Economopoulos SP, Tsikalas GK, Katerinopoulos HE, Tagmatarchis N (2010) Fullerene–coumarin dyad as a selective metal receptor: synthesis, photophysical properties, electrochemistry and ion binding studies. Chem A Eur J 16(39):11969–11976. https://doi.org/10.1002/chem.201001665
Patil AD, Freyer AJ, Eggleston DS, Haltiwanger RC, Bean MF, Taylor PB, Caranfa MJ, Breen AL, Bartus HR (1993) The inophyllums, novel inhibitors of HIV-1 reverse transcriptase isolated from the Malaysian tree, Calophyllum inophyllum Linn. J Med Chem 36(26):4131–4138. https://doi.org/10.1021/jm00078a001
Piller NB (1975) A comparison of the effectiveness of some anti-inflammatory drugs on thermal oedema. British J Experim Pathol
Poole SK, Poole CF (1994) Thin-layer chromatographic method for the determination of the principal polar aromatic flavour compounds of the cinnamons of commerce. Analyst 119(1):113–120. https://doi.org/10.1039/AN9941900113
Portugal J (2003) Chartreusin, elsamicin A and related anti-cancer antibiotics. Curr Med Chem Anti-Cancer Agents 3(6):411–420. https://doi.org/10.2174/1568011033482215
Preat J, Jacquemin D, Perpète EA (2005) Theoretical investigations of the UV spectra of coumarin derivatives. Chem Phy Letters 415(1–3):20–24
Qian Y, Yang B, Shen Y, Du Q, Lin L, Lin J, Zhu H (2013) A BODIPY–coumarin-based selective fluorescent probe for rapidly detecting hydrogen sulfide in blood plasma and living cells. Sens Actuators B Chem 182:498–503
Riveiro ME, De Kimpe N, Moglioni A, Vazquez R, Monczor F, Shayo C, Davio C (2010) Coumarins: old compounds with novel promising therapeutic perspectives. Curr Med Chem 17(13):1325–1338. https://doi.org/10.2174/092986710790936284
Rosselli S, Maggio AM, Faraone N, Spadaro V, Morris-Natschke SL, Bastow KF, Lee KH, Bruno M (2009) The cytotoxic properties of natural coumarins isolated from roots of Ferulago campestris (Apiaceae) and of synthetic ester derivatives of aegelinol. Nat Prod Commun 4(12):1934578X0900401219. https://doi.org/10.1177/1934578X0900401219
Roussakis E, Pergantis SA, Katerinopoulos HE (2008) Coumarin-based ratiometric fluorescent indicators with high specificity for lead ions. Chem Commun 26(46):6221–6223. https://doi.org/10.1039/B811569A
Satpati AK, Kumbhakar M, Maity DK, Pal H (2005) Photophysical investigations of the solvent polarity effect on the properties of coumarin-6 dye. Chem Phys Lett 407(1–3):114–118. https://doi.org/10.1016/j.cplett.2005.03.070
Sethna SM, Shah NM (1945) The chemistry of coumarins. Chem Rev 36(1):1–62. https://doi.org/10.1021/cr60113a001
Shabbir M, Sultani SZ, Jabbar A, Choudhary MI (1997) Cinnamates and coumarins from the leaves of Murraya paniculata. Phytochemistry 44(4):683–685. https://doi.org/10.1016/S0031-9422(96)00617-6
Shastri L, Kalegowda S, Kulkarni M (2007) The synthesis of pyrrole bis-coumarins, new structures for fluorescent probes. Tetrahedron Lett 48(40):7215–7217. https://doi.org/10.1016/j.tetlet.2007.07.189
Shin E, Choi KM, Yoo HS, Lee CK, Hwang BY, Lee MK (2010) Inhibitory effects of coumarins from the stem barks of Fraxinus rhynchophylla on adipocyte differentiation in 3T3-L1 cells. Biol Pharm Bull 33(9):1610–1614. https://doi.org/10.1248/bpb.33.1610
Shukla VS, Dutta SC, Baruah RN, Sharma RP, Thyagarajan G, Herz W, Kumar N, Watanabe K, Blount JF (1982) New 5-methylcoumarins from Ethulia conyzoides. Phytochemistry 21(7):1725–1731. https://doi.org/10.1016/S0031-9422(82)85048-6
Signore G, Nifosi R, Albertazzi L, Storti B, Bizzarri R (2010) Polarity-sensitive coumarins tailored to live cell imaging. J Am Chem Soc 132(4):1276–1288. https://doi.org/10.1021/ja9050444
Song CE, Jung DU, Choung SY, Roh EJ (2004) dramatic enhancement of catalytic activity an ionic liquid: highly practical Fridel-Crafts alkenylation of arenes with alkynes catalyzed by metal triflates. Angew Chem Int Ed 43(45):6183–8185. https://doi.org/10.1002/anie.200460292
Wald RW, Feuer G (1971) Molecular orbital calculations on coumarins and the induction of drug-metabolizing enzymes. J Med Chem 14(11):1081–1084. https://doi.org/10.1021/jm00293a016
Wang X, Krebs LJ, Al-Nuri M, Pudavar HE, Ghosal S, Liebow C, Nagy AA, Schally AV, Prasad PN (1999) A chemically labeled cytotoxic agent: two-photon fluorophore for optical tracking of cellular pathway in chemotherapy. Proc Natl Acad Sci 96(20):11081–11084. https://doi.org/10.1073/pnas.96.20.11081
Wang ZS, Cui Y, Hara K, Dan-oh Y, Kasada C, Shinpo A (2007) A high-light-harvesting-efficiency coumarin dye for stable dye-sensitized solar cells. Adv Mater 19(8):1138–1141. https://doi.org/10.1002/adma.200601020
Whang WK, Park HS, Ham I, Oh M, Namkoong H, Kim HK, Hwang DW, Hur SY, Kim TE, Park YG, Kim JR (2005) Natural compounds, fraxin and chemicals structurally related to fraxin protect cells from oxidative stress. Exp Mol Med 37(5):436–446. https://doi.org/10.1038/emm.2005.54
Witaicenis A, Seito LN, Di Stasi LC (2010) Intestinal anti-inflammatory activity of esculetin and 4-methylesculetin in the trinitrobenzenesulphonic acid model of rat colitis. Chem Biol Interact 186(2):211–218. https://doi.org/10.1016/j.cbi.2010.03.045
Wu W, Cao Z, Zhao Y (2012) Theoretical studies on absorption, emission, and resonance Raman spectra of coumarin 343 isomers. J Chem Phys 136(11):114305. https://doi.org/10.1063/1.3693264
Xie L, Chen Y, Wu W, Guo H, Zhao J, Yu X (2012) Fluorescent coumarin derivatives with large stokes shift, dual emission and solid state luminescent properties: an experimental and theoretical study. Dyes Pigm 92(3):1361–1369. https://doi.org/10.1016/j.dyepig.2011.09.023
Ye D, Wang L, Li H, Zhao J, Cao D (2013) Synthesis of coumarin-containing conjugated polymer for naked-eye detection of DNA and cellular imaging. Sens Actuators B: Chem 181:234–243. https://doi.org/10.1016/j.snb.2013.02.040
Yusupov MI, Sidyakin GP (1975) Fraxidin and isofraxidin from Artemisia scotina. Chem Nat Comp 11(1):94. https://doi.org/10.1007/BF00567042
Żamojć K, Wiczk W, Zaborowski B, Jacewicz D, Chmurzyński L (2014) Analysis of fluorescence quenching of coumarin derivatives by 4-hydroxy-TEMPO in aqueous solution. J Fluoresc 24(3):713–718. https://doi.org/10.1007/s10895-013-1342-3
Zhao Y, Yu T, Wu Y, Zhang H, Fan D, Gan Z, Yang L, Han X, Zhang Y (2012) Synthesis, photo-and electro-luminescence of 3-benzoxazol-2-yl-coumarin derivatives. J Fluoresc 22(2):631–638. https://doi.org/10.1007/s10895-011-0999-8
Acknowledgements
The authors will like to thank Higher Education Commission for providing the necessary facilities and fundings through NRPU project number 9922.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fatima, S., Mansha, A., Asim, S. et al. Absorption spectra of coumarin and its derivatives. Chem. Pap. 76, 627–638 (2022). https://doi.org/10.1007/s11696-021-01902-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-021-01902-6