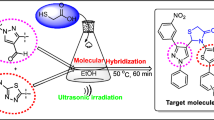
Abstract
A facile one-pot reaction has been set up for the synthesis of novel 5-oxo-2-pyrrolidinecarboxamides and 7-oxo-2-azepanecarboxamides 4a-j from three-component reaction of keto carboxylic acids 1a-d, various primary amines 2a-b and isocyanides 3a-b under ultrasound irradiation. The present protocol offers attractive characteristics such as easy handling methodology, good-to-excellent yields, environmental friendliness, clean reaction, higher atom economy, convenient operation, and shorter reaction time. The newly synthesized 5-oxo-2-pyrrolidinecarboxamides and 7-oxo-2-azepanecarboxamides 4a-j have been screened for their antimicrobial activity and the majority of these cyclic amides exhibited weak antimicrobial activity.





Similar content being viewed by others
References
Cella R, Stefani HA (2009) Ultrasound in heterocycles chemistry. Tetrahedron 65:2619–2641. https://doi.org/10.1016/j.tet.2008.12.027
Dandia A, Singh R, Joshi J, Kumari S (2013) An eco-compatible synthesis of medicinally important novel class of trispiroheterocyclic framework using 2, 2, 2-trifluoroethanol as a reusable medium. J Fluorine Chem 156:283–289. https://doi.org/10.1016/j.jfluchem.2013.07.008
Hanusch-Kompa C, Ugi I (1998) Multi-component reactions 13: Synthesis of γ-lactams as part of a multiring system via Ugi-4-centre-3-component reaction. Tetrahedron Lett 39:2725–2728. https://doi.org/10.1016/S0040-4039(98)00428-6
Harriman GC (1997) Synthesis of small and medium sized 2, 2-disubstituted lactams via the “intramolecular” three component Ugi reaction. Tetrahedron Lett 38:5591–5594. https://doi.org/10.1016/S0040-4039(97)01265-3
Hu Y, Zou Y, Wu H, Shi D (2012) A facile and efficient ultrasound-assisted synthesis of novel dispiroheterocycles through 1, 3-dipolar cycloaddition reactions. Ultrason Sonochem 19:264–269. https://doi.org/10.1016/j.ultsonch.2011.07.006
Hulme C, Dietrich J (2009) Emerging molecular diversity from the intra-molecular Ugi reaction: iterative efficiency in medicinal chemistry. Mol div 13:195. https://doi.org/10.1007/s11030-009-9111-6
Ivachtchenko AV, Ivanenkov YA, Kysil VM, Krasavin MY, Ilyin AP (2010) Multicomponent reactions of isocyanides in the synthesis of heterocycles. Russ Chem Rev 79:787. https://doi.org/10.1070/RC2010v079n09ABEH004086
Jassem AM, Al-Ajely HM, Almashal FA, Chen B (2019a) Application of the cleavable isocyanide in efficient approach to pyroglutamic acid analogues with potential biological activity. Russ J Gen Chem 89:2562–2570. https://doi.org/10.1134/S1070363219120363
Jassem AM, Almashal FA, Jaber H (2020a) Solvent-free microwave assisted synthesis of novel pyrazole-oxopyrrolidine and pyrazole-oxopiperidine derivatives and their antimicrobial activity. Russ J Gen Chem 90:895–900. https://doi.org/10.1134/S1070363220050230
Jassem AM, Almashal FA, Mohammed MQ, Jabir H (2020b) A catalytic and green method for one-pot synthesis of new Hantzsch 1, 4-dihydropyridines. SN Appl Sci 2:359. https://doi.org/10.1007/s42452-020-2165-x
Jassem AM, Dhumad AM (2020) Microwave-assisted green synthesis, antimicrobial activity, and drug-likeness of novel isoindolinone derivatives. Monatsh Chem 151:1433–1442. https://doi.org/10.1007/s00706-020-02661-y
Jassem AM, Dhumad AM, Almashal FA, Alshawi JM (2020c) Microwave-assisted synthesis, molecular docking and anti-HIV activities of some drug-like quinolone derivatives. Med Chem Res 29:1067–1076. https://doi.org/10.1007/s00044-020-02546-z
Jassem AM, Dhumad AM, Almashal FAK (2020d) Synthesis of new drug-like piperazine-2,5-diones by the Ugi/tandem process catalyzed by TMSOTf and their molecular docking. Russ J Gen Chem 90:2181–2188. https://doi.org/10.1134/S1070363220110262
Jassem AM, Raheemah AH, Radhi WA, Ali AM, Jaber HA (2019b) Highly diastereoselective metal-free catalytic synthesis of drug-like spiroimidazolidinone. Russ J Org Chem 55:1598–1603. https://doi.org/10.1134/S107042801910021X
Kaveti B, Ramírez-López SC, Montaño RG (2018) Ultrasound-assisted green one-pot synthesis of linked bis-heterocycle peptidomimetics via IMCR/post-transformation/tandem strategy. Tetrahedron lett 59:4355–4358. https://doi.org/10.1016/j.tetlet.2018.10.062
Kitas EA, Galley G, Jakob-Roetne R, Flohr A, Wostl W, Mauser H, Alker AM, Czech C, Ozmen L, David-Pierson P (2008) Substituted 2-oxo-azepane derivatives are potent, orally active γ-secretase inhibitors. Bioorg Med Chem Lett 18:304–308. https://doi.org/10.1016/j.bmcl.2007.10.074
Masse CE, Morgan AJ, Adams J, Panek JS (2000) Syntheses and biological evaluation of (+)-lactacystin and analogs. Eur J Org Chem 2000:2513–2528. https://doi.org/10.1002/1099-0690(200007)2000
Nenajdenko VG, Reznichenko AL, Balenkova ES (2007) Diastereoselective Ugi reaction without chiral amines: the synthesis of chiral pyrroloketopiperazines. Tetrahedron 63:3031–3041. https://doi.org/10.1016/j.tet.2007.01.056
Sgouras D, Maragkoudakis P, Petraki K, Martinez-Gonzalez B, Eriotou E, Michopoulos S, Kalantzopoulos G, Tsakalidou E, Mentis Α (2004) In vitro and in vivo inhibition of Helicobacter pylori by Lactobacillus casei strain Shirota. Appl Environ Microbiol 70:518–526. https://doi.org/10.1128/AEM.70.1.518-526.2004
Shabalala N, Maddila S, Jonnalagadda SB (2016) Catalyst-free, one-pot, four-component green synthesis of functionalized 1-(2-fluorophenyl)-1, 4-dihydropyridines under ultrasound irradiation. New J Chem 40:5107–5112. https://doi.org/10.1039/C5NJ03574K
Short KM, Mjalli AM (1997) A solid-phase combinatorial method for the synthesis of novel 5-and 6-membered ring lactams. Tetrahedron Lett 38:359–362. https://doi.org/10.1016/S0040-4039(96)02303-9
Suslick KS (1990) Sonochemistry. Science 247:1439–1445. https://doi.org/10.1126/science.247.4949.1439
Suslick KS (2013) Applications of ultrasound to materials chemistry. MRS Bull 20:29–34. https://doi.org/10.1557/S088376940004464X
Suslick KS, Hammerton DA, Cline RE (1986) Sonochemical hot spot. J Am Chem Soc 108:5641–5642. https://doi.org/10.1021/ja00278a055
Tsaloev A, Ilyin A, Tkachenko S, Ivachtchenko A, Kravchenko D, Krasavin M (2011) Cyclic products of the Ugi reaction of aldehydo and keto carboxylic acids: chemoselective modification. Tetrahedron lett 52:1800–1803. https://doi.org/10.1016/j.tetlet.2011.02.028
Ugi I, Demharter A, Hörl W, Schmid T (1996) Ugi reactions with trifunctional α-amino acids, aldehydes, isocyanides and alcohols. Tetrahedron 52:11657–11664. https://doi.org/10.1016/0040-4020(96)00647-3
Ugi I, Dömling A, Hörl W (1994) Multicomponent reactions in organic chemistry. Endeavour 18:115–122. https://doi.org/10.1016/S0160-9327(05)80086-9
Van Berkel SS, Bögels BG, Wijdeven MA, Westermann B, Rutjes FP (2012) Recent advances in asymmetric isocyanide-based multicomponent reactions. Eur J Org Chem 2012:3543–3559. https://doi.org/10.1002/ejoc.201200030
Zou Y, Wu H, Hu Y, Liu H, Zhao X, Ji H, Shi D (2011) A novel and environment-friendly method for preparing dihydropyrano[2,3-c]pyrazoles in water under ultrasound irradiation. Ultrason Sonochem 18:708–712. https://doi.org/10.1016/j.ultsonch.2010.11.012
Acknowledgements
Authors gratefully acknowledge financial support from Ministry of Higher Education and Scientific Research (Iraq). The authors would express sincere thanks to University of Sheffield, Uk for performing 1H, 13C NMR, HRMS and FTIR spectra. Authors are also grateful to staff (Cell and Molecular Biology Lab, Department of Zoology and Cytology, Government College University, Pakistan) for performing the antimicrobial activity.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jassem, A.M., Chen, B. Ultrasound-assisted synthesis and in vitro antimicrobial activity of novel 5-oxo-2-pyrrolidinecarboxamides and 7-oxo-2-azepanecarboxamides. Chem. Pap. 75, 3575–3586 (2021). https://doi.org/10.1007/s11696-021-01603-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-021-01603-0