Abstract
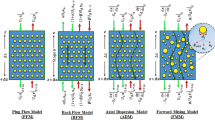
The present work is performed to assess the hydrodynamic parameters and mass transfer coefficients in a PRDC column for the chemical reaction systems. By considering the selective extraction of molybdenum from tungsten, the extraction parameters and synergistic enhancement factor were interpreted at the batch experiments. In the column experiments, the impacts of operating conditions such as the agitation rate, inlet solvent phase velocity as well as inlet aqueous phase velocity were studied on the drop behavior, overall mass transfer coefficients, and extraction efficiencies. The modified correlations by using relationship of dimensionless numbers were illustrated to estimate the hydrodynamic factors (holdup value and the Sauter mean drop diameter) under the physical and reactive systems. The mass transfer coefficients for the continuous phase were evaluated by the axial diffusion method in both extraction and stripping stages. It was observed that the column performance was primarily affected by the agitation speed and mass transfer direction, but this phenomenon slightly changes with the inlet phase velocities. The previous empirical models were compared with the obtained mass transfer data, and the available models were failed to precisely anticipate the experimental data due to chemical reaction conditions. Finally, two new models based on the dimensionless numbers were derived for calculating the overall Sherwood number. The value of separation factor in the extractor indicated the column capacity with the excellent separation for molybdenum from tungsten in the sulfate solution.















Similar content being viewed by others
Abbreviations
- AARE:
-
Average absolute relative error (-)
- a :
-
Interfacial area (m2/m3)
- d 32 :
-
Sauter mean drop diameter (m)
- D :
-
Distribution coefficient (-)
- D col :
-
Column diameter (m)
- D c :
-
Molecular diffusivity of continuous phase (m2/s)
- D R :
-
Disk diameter (m)
- D S :
-
Stator ring diameter (m)
- E :
-
Axial mixing coefficient (m2/s)
- E%:
-
Extraction efficiency (-)
- g :
-
Acceleration due to gravity (m/s2)
- H :
-
Effective height of the column (m)
- h c :
-
Compartment height (m)
- K oc :
-
Overall continuous mass transfer coefficient (m/s)
- K c :
-
Continuous mass transfer coefficient (m/s)
- K d :
-
Dispersed mass transfer coefficient (m/s)
- m :
-
Distribution ratio (-)
- N :
-
Agitation speed (1/s)
- N oc :
-
Number of ‘true’ transfer unit (-)
- Re:
-
Reynolds number (-)
- Sc:
-
Schmitt number (-)
- P:
-
Péclet number (-)
- P n org log :
-
Probability of number density (-) for normal or logarithmic function
- Pec :
-
Continuous-phase Péclet number (= d32Vs/Dc)
- [M]:
-
Concentration of metal ions (mg/L)
- [M]aq, s :
-
Concentration of metal ions in the stripping solution (mg/L)
- V org :
-
Volume of organic phase (ml)
- V aq :
-
Volume of aqueous phase (ml)
- V aq, s :
-
Volume of aqueous phase in the stripping solution (ml)
- d e :
-
Equivalent diameter (m)
- d i :
-
Droplet diameter (m)
- n i :
-
Number of droplets of mean diameter di (-)
- SF:
-
Separation factor (-)
- SEF:
-
Synergistic enhancement factor (-)
- Sh:
-
Sherwood number (-)
- S%:
-
Stripping efficiency (-)
- NDP:
-
Number values of data points
- V :
-
Superficial velocity (m/s)
- ∀ :
-
Specific volume (m3)
- \(\overline{V}_{\text{c}}\) :
-
Continuous phase true velocity (m/s)
- V s :
-
Slip velocity (m/s)
- x :
-
Mass fraction of elements in continuous phase (-)
- x * :
-
Equilibrium mass fraction (-)
- y :
-
Mass fraction of elements in dispersed phase (-)
- α :
-
Constant parameter of probability of density function (-)
- β :
-
Constant parameter of probability of density function (-)
- ρ c :
-
Density of continuous phase (Kg/m3)
- ρ d :
-
Density of dispersed phase (Kg/m3)
- µ d :
-
Viscosity of dispersed phase (pa.s)
- µ c :
-
Viscosity of continuous phase (pa.s)
- φ :
-
Dispersed phase holdup (-)
- σ :
-
Interfacial tension (N/m)
- κ :
-
Viscosity ratio of µd/µc
References
Asadollahzadeh M, Ghaemi A, Torab-Mostaedi M, Shahhosseini S (2016a) Experimental mass transfer coefficients in a pilot plant multistage column extractor. Chin J Chem Eng 24:989–999. https://doi.org/10.1016/j.cjche.2016.02.004
Asadollahzadeh M, Shahhosseini S, Torab-Mostaedi M, Ghaemi A (2016b) Drop behavior in a pilot plant Oldshue-Rushton extraction column for three various liquid–liquid systems. Sep Purif Technol 159:7–17. https://doi.org/10.1016/j.seppur.2015.12.050
Asadollahzadeh M, Torab-Mostaedi M, Shahhosseini S, Ghaemi A (2016c) Holdup, characteristic velocity and slip velocity between two phases in a multi-impeller column for high/medium/low interfacial tension systems. Chem Eng Process 100:65–78. https://doi.org/10.1016/j.cep.2015.11.013
Asadollahzadeh M, Hemmati A, Torab-Mostaedi M, Shirvani M, Ghaemi A, Mohsenzadeh Z (2017) Use of axial dispersion model for determination of Sherwood number and mass transfer coefficients in a perforated rotating disc contactor. Chin J Chem Eng 25:53–61. https://doi.org/10.1016/j.cjche.2016.06.007
Asadollahzadeh M, Torab-Mostaedi M, Torkaman R (2018) Drop behavior in a pilot plant asymmetric rotating disc extraction column for three various liquid–liquid systems. Chem Eng Res Des 138:366–373. https://doi.org/10.1016/j.cherd.2018.03.013
Cavalcanti MTH, Carneiro-da-Cunha M, Brandi I, Porto T, Converti A, Lima Filho J, Porto A, Pessoa A (2008) Continuous extraction of α-toxin from a fermented broth of Clostridium perfringens Type A in perforated rotating disc contactor using aqueous two-phase PEG–phosphate system. Chem Eng Process 47:1771–1776. https://doi.org/10.1016/j.cep.2007.09.018
Coca J, Díez FV, Morís MA (1990) Solvent extraction of molybdenum and tungsten by Alamine 336 and DEHPA. Hydrometallurgy 25:125–135. https://doi.org/10.1016/0304-386X(90)90034-Y
De AK, Khopkar SM, Chalmers RA (1970) Solvent extraction of metals. Van Nostrand Reinhold Co, New York
Dutta BK (2007) Principles of mass transfer and seperation processes. PHI Learning Pvt. Ltd., New York
Elwell WT, Wood DF (2013) Analytical chemistry of molybdenum and tungsten: including the analysis of the metals and their alloys, vol 47. Elsevier, New York
Ferreira AE, Agarwal S, Machado RM, Gameiro MLF, Santos SM, Reis MTA, Ismael MRC, Correia MJN, Carvalho JM (2010) Extraction of copper from acidic leach solution with Acorga M5640 using a pulsed sieve plate column. Hydrometallurgy 104:66–75. https://doi.org/10.1016/j.hydromet.2010.04.013
Ghadiri M, Ashrafizadeh SN, Taghizadeh M (2014) Study of molybdenum extraction by trioctylamine and tributylphosphate and stripping by ammonium solutions. Hydrometallurgy 144:151–155. https://doi.org/10.1016/j.hydromet.2014.02.009
Godfrey JC, Slater MJ (1994) Liquid-liquid extraction equipment. Wiley, New York
Guan W, Zhang G, Gao C (2012) Solvent extraction separation of molybdenum and tungsten from ammonium solution by H2O2-complexation. Hydrometallurgy 127:84–90. https://doi.org/10.1016/j.hydromet.2012.07.008
Hemmati A, Shirvani M, Torab-Mostaedi M, Ghaemi A (2015a) Hold-up and flooding characteristics in a perforated rotating disc contactor (PRDC). RSC Adv 5:63025–63033. https://doi.org/10.1039/C5RA08938G
Hemmati A, Torab-Mostaedi M, Shirvani M, Ghaemi A (2015b) A study of drop size distribution and mean drop size in a perforated rotating disc contactor (PRDC). Chem Eng Res Des 96:54–62. https://doi.org/10.1016/j.cherd.2015.02.005
Hemmati A, Shirvani M, Torab-Mostaedi M, Ghaemi A (2016) Mass transfer coefficients in a perforated rotating disc contactor (PRDC). Chem Eng Process 100:19–25. https://doi.org/10.1016/j.cep.2015.11.011
Kadam B, Joshi J, Patil R (2009) Hydrodynamic and mass transfer characteristics of asymmetric rotating disc extractors. Chem Eng Res Des 87:756–769. https://doi.org/10.1016/j.cherd.2008.10.001
Kawabuchi K, Kuroda R (1969) A combined ion-exchange spectrophotometric method for the determination of molybdenum and tungsten in sea water. Anal Chim Acta 46:23–30. https://doi.org/10.1016/0003-2670(69)80037-1
Kawabuchi K, Kuroda R (1970) Anion-exchange separation and spectrophotometric determination of molybdenum and tungsten in silicate rocks. Talanta 17:67–73. https://doi.org/10.1016/0039-9140(70)80051-0
Kirou V, Tavlarides LL, Bonnet J, Tsouris C (1988) Flooding, holdup, and drop size measurements in a multistage column extractor. AIChE J 34:283–292. https://doi.org/10.1002/aic.690340212
Kumar A, Hartland S (1992) Prediction of axial mixing coefficients in rotating disc and asymmetric rotating disc extraction columns. Can J Chem Eng 70:77–87. https://doi.org/10.1002/cjce.5450700112
Kumar A, Hartland S (1999) Correlations for prediction of mass transfer coefficients in single drop systems and liquid–liquid extraction columns. Chem Eng Res Des 77:372–384. https://doi.org/10.1205/026387699526359
Liu C, Li M, Liang C, Wang W (2013) Measurement and analysis of bimodal drop size distribution in a rotor–stator homogenizer. Chem Eng Sci 102:622–631. https://doi.org/10.1016/j.ces.2013.08.030
Maaß S, Wollny S, Voigt A, Kraume M (2011) Experimental comparison of measurement techniques for drop size distributions in liquid/liquid dispersions. Exp Fluids 50:259–269. https://doi.org/10.1007/s00348-010-0918-9
Mahmoud MH, Nakamura S, Akiba K (1996) Extraction separation of molybdenum (VI) and tungsten (VI) by α-hydroxy oxim. Solv Extr Ion Exch 14:203–217. https://doi.org/10.1080/07366299608918335
Nekovář P, Schrötterová D (2000) Extraction of V (V), Mo (VI) and W (VI) polynuclear species by primene JMT. Chem Eng J 79:229–233. https://doi.org/10.1016/S1385-8947(00)00207-2
Nguyen TH, Lee MS (2015) Separation of molybdenum (VI) and tungsten (VI) from sulfate solutions by solvent extraction with LIX 63 and PC 88A. Hydrometallurgy 155:51–55. https://doi.org/10.1016/j.hydromet.2015.04.014
Nguyen TH, Lee MS (2016) A review on the separation of molybdenum, tungsten, and vanadium from leach liquors of diverse resources by solvent extraction. Geosyst Eng. 19:247–259. https://doi.org/10.1080/12269328.2016.1186577
Oliveira N, Silva DM, Gondim M, Mansur MB (2008) A study of the drop size distributions and hold-up in short Kühni columns. Braz J Chem Eng 25:729–741. https://doi.org/10.1590/S0104-66322008000400010
Porto ALF, Sarubbo LA, Moreira KA, Melo HJFd, Lima-Filho JL, Campos-Takaki GM, Tambourgi EB (2004) Recovery of ascorbic oxidoreductase from crude extract with an aqueous two-phase system in a perforated rotating disc contactor. Braz Arch Biol Technol 47:821–826. https://doi.org/10.1590/S1516-89132004000500019
Qingyuan Z, Huihao F (1986) Separation of molybdenum from tungsten by di-2-ethylhexyl phosphoric acid extractant. Hydrometallurgy 16:263–270. https://doi.org/10.1016/0304-386X(86)90002-2
Rincón-Rubio L, Kumar A, Hartland S (1993) Characterization of flooding in a Wirz extraction column. Can J Chem Eng 71:844–851. https://doi.org/10.1002/cjce.5450710604
Saily A, Khurana U, Yadav S, Tandon S (1996) Thiophosphinic acids as selective extractants for molybdenum recovery from a low grade ore and spent catalysts. Hydrometallurgy 41:99–105. https://doi.org/10.1016/0304-386X(95)00052-I
Samani MG, Haghighi Asl A, Safdari J, Torab-Mostaedi M (2012) Drop size distribution and mean drop size in a pulsed packed extraction column. Chem Eng Res Des 90:2148–2154. https://doi.org/10.1016/j.cherd.2012.06.002
Sarubbo L, Oliveira L, Porto A, Campos-Takaki G, Tambourgi E (2005) Studies of efficiency in a perforated rotating disc contactor using a polymer-polymer aqueous two-phase systems. Braz J Chem Eng 22:489–493. https://doi.org/10.1590/S0104-66322005000300016
Seader JD, Henley EJ, Roper DK (1998) Separation process principles, vol 25. Wiley, New York
Shakib B, Torab-Mostaedi M, Outokesh M, Asadollahzadeh M (2019a) Mass transfer coefficients of extracting Mo (VI) and W (VI) in a stirred tank by solvent extraction using mixture of Cyanex272 and D2EHPA. Sep Sci Technol. https://doi.org/10.1080/01496395.2019.1672741.28
Shakib B, Torab-Mostaedi M, Outokesh M, Asadollahzadeh M (2019b) Direct extraction of Mo (VI) from sulfate solution by synergistic extractants in the rotation column. Chin J Chem Eng 28:445–455. https://doi.org/10.1016/j.cjche.2019.11.011
Soltanali S, Ziaie-Shirkolaee Y, Amoabediny G, Rashedi H, Sheikhi A, Chamanrokh P (2009) Hydrodynamics and mass transfer performance of rotating sieved disc contactors used for reversed micellar extraction of protein. Chem Eng Sci 64:2301–2306. https://doi.org/10.1016/j.ces.2009.02.005
Talla R, Gaikwad S, Pawar S (2010) Solvent extraction and separation of Mo (VI) and W (VI) from hydrochloric acid solutions using cyanex-923 as extractant. Ind J Chem Technol 17:436–440
Torab-Mostaedi M, Asadollahzadeh M (2015) Mass transfer performance in an asymmetric rotating disc contactor. Chem Eng Res Des 94:90–97. https://doi.org/10.1016/j.cherd.2014.12.007
Torab-Mostaedi M, Safdari J (2009) Mass transfer coefficients in a pulsed packed extraction column. Chem Eng Process 48:1321–1326. https://doi.org/10.1016/j.cep.2009.06.002
Torab-Mostaedi M, Safdari S, Moosavian M, Maragheh MG (2008) Mass transfer coefficients in a Hanson mixer-settler extraction column. Braz J Chem Eng 25:473–481. https://doi.org/10.1590/S0104-66322008000300005
Torab-Mostaedi M, Ghaemi A, Asadollahzadeh M, Pejmanzad P (2011) Mass transfer performance in pulsed disc and doughnut extraction columns. Braz J Chem Eng 28:447–456. https://doi.org/10.1590/S0104-66322011000300010
Torkaman R, Asadollahzadeh M, Torab-Mostaedi M, Maragheh MG (2017) Reactive extraction of cobalt sulfate solution with D2EHPA/TBP extractants in the pilot plant Oldshue-Rushton column. Chem Eng Res Des 120:58–68. https://doi.org/10.1016/j.cherd.2017.02.004
Wang Y, Fei W, Sun J, Wan Y (2002) Hydrodynamics and mass transfer performance of a modified rotating disc contactor (MRDC). Chem Eng Res Des 80:392–400. https://doi.org/10.1205/026387602317446434
Wilke C, Chang P (1955) Correlation of diffusion coefficients in dilute solutions. AIChE J 1:264–270. https://doi.org/10.1002/aic.690010222
Xia Y, Xiao L, Xiao C, Zeng L (2015) Direct solvent extraction of molybdenum (VI) from sulfuric acid leach solutions using PC-88A. Hydrometallurgy 158:114–118. https://doi.org/10.1016/j.hydromet.2015.10.016
Zhao Z, Li J, Cao C, Huo G, Zhang G, Li H (2010) Recovery and purification of molybdenum from Ni–Mo ore by direct air oxidation in alkaline solution. Hydrometallurgy 103:68–73. https://doi.org/10.1016/j.hydromet.2010.02.018
Zhao Z, Cao C, Chen X, Huo G (2011) Separation of macro amounts of tungsten and molybdenum by selective precipitation. Hydrometallurgy 108:229–232. https://doi.org/10.1016/j.hydromet.2011.04.006
Funding
This work was not supported by any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shakib, B., Torkaman, R., Torab-Mostaedi, M. et al. Revealing mass transfer and hydrodynamic effects in a PRDC column by using the integration of extraction and separation for molybdenum and tungsten ions from aqueous solution. Chem. Pap. 74, 4295–4313 (2020). https://doi.org/10.1007/s11696-020-01241-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-020-01241-y