Abstract
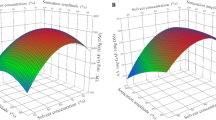
The total saponins from Aralia elata (Miq.) Seem fruits and rachises were isolated by microwave-assisted extraction (MAE), ultrasonic extraction, heat reflux extraction, and soxhlet extraction. These techniques were compared from four perspectives: extraction kinetics, saponin yield, disintegration to the raw materials, and antioxidant activity. MAE had the highest extraction efficiency, shortest time, and greatest disintegration to the raw materials. The extracts obtained with MAE had the highest antioxidant activities, as the DPPH free radical scavenging abilities of the fruit and rachis extracts were 70.86 ± 1.33 and 50.85 ± 1.68 mg VC g−1md, respectively. The MAE parameters were optimized by the response surface methodology. The optimum conditions (92% and 91% ethanol, solid-to-liquid ratios of 1:30 and 1:22, microwave powers of 530 W and 400 W, microwave times of 40 s and 54 s) provided extraction yields of 2.98 ± 0.037 mg g−1 and 0.94 ± 0.026 mg g−1 from the fruits and rachises, respectively.



Similar content being viewed by others
References
Belwal T, Dhyani P, Bhatt ID, Rawal RS, Pande V (2016) Optimization extraction conditions for improving phenolic content and antioxidant activity in Berberis asiatica fruits using response surface methodology (RSM). Food Chem 207:S0308814616304551
Benzie IFF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of “Antioxidant Power”: the frap assay. Anal Biochem 239:70–76. https://doi.org/10.1006/abio.1996.0292
Chen Y, Xue Y (2019) Optimization of microwave assisted extraction, chemical characterization and antitumor activities of polysaccharides from porphyra haitanensis. Carbohydr Polym 206:179–186. https://doi.org/10.1016/j.carbpol.2018.10.093
Chen L, Jin H, Ding L, Zhang H, Li J, Qu C, Zhang H (2008) Dynamic microwave-assisted extraction of flavonoids from Herba Epimedii. Sep Purif Technol 59:50–57. https://doi.org/10.1016/j.seppur.2007.05.025
Chouaibi M, Rezig L, Hamdi S, Ferrari G (2019) Chemical characteristics and compositions of red pepper seed oils extracted by different methods. Ind Crop Prod 128:363–370. https://doi.org/10.1016/j.indcrop.2018.11.030
Da Porto C, Natolino A (2018) Extraction kinetic modelling of total polyphenols and total anthocyanins from saffron floral bio-residues: comparison of extraction methods. Food Chem 258:137–143. https://doi.org/10.1016/j.foodchem.2018.03.059
Dong Z, Gu F, Xu F, Wang Q (2014) Comparison of four kinds of extraction techniques and kinetics of microwave-assisted extraction of vanillin from Vanilla planifolia Andrews. Food Chem 149:54–61. https://doi.org/10.1016/j.foodchem.2013.10.052
Fattouch S et al (2007) Antimicrobial activity of tunisian quince (Cydonia oblonga Miller) pulp and peel polyphenolic extracts. J Agric Food Chem 55:963–969. https://doi.org/10.1021/jf062614e
Gao Z, Khalid M, Jan F, Saeed-ur R, Jiang X, Yu X (2019) Effects of light-regulation and intensity on the growth, physiological and biochemical properties of Aralia elata (miq.) seedlings. S Afr J Bot 121:456–462. https://doi.org/10.1016/j.sajb.2018.12.008
Hiai S, Oura H, Nakajima T (1976) Color reaction of some sapogenins and saponins with vanillin and sulfuric acid. Planta Med 29:116–122. https://doi.org/10.1055/s-0028-1097639
Huang D, Ou B, Prior RL (2005) The chemistry behind antioxidant capacity assays. J Agric Food Chem 53:1841–1856. https://doi.org/10.1021/jf030723c
Jesus MS, Genisheva Z, Romaní A, Pereira RN, Teixeira JA, Domingues L (2019) Bioactive compounds recovery optimization from vine pruning residues using conventional heating and microwave-assisted extraction methods. Ind Crop Prod 132:99–110. https://doi.org/10.1016/j.indcrop.2019.01.070
Kedare SB, Singh RP (2011) Genesis and development of DPPH method of antioxidant assay. J Food Sci Technol 48:412–422. https://doi.org/10.1007/s13197-011-0251-1
Kwiatkowski M, Kravchuk O, Skouroumounis GK, Taylor DK (2020) Response surface parallel optimization of extraction of total phenolics from separate white and red grape skin mixtures with microwave-assisted and conventional thermal methods. J Clean Prod 251:119563. https://doi.org/10.1016/j.jclepro.2019.119563
Liu G, Sheng Y, Zhang M, Sun D (2016) A polysaccharide from the leaves of Aralia elata induces apoptosis in U-2 OS cells via mitochondrial-dependent pathway. Int J Biol Macromol 93:418–425. https://doi.org/10.1016/j.ijbiomac.2016.08.067
Lou Z, Wang H, Zhu S, Zhang M, Gao Y, Ma C, Wang Z (2010) Improved extraction and identification by ultra performance liquid chromatography tandem mass spectrometry of phenolic compounds in burdock leaves. J Chromatogr A 1217:2441–2446. https://doi.org/10.1016/j.chroma.2009.12.022
Luo Y, Dong X, Yu Y, Sun G, Sun X (2015) Total aralosides of Aralia elata (Miq) Seem (TASAES) ameliorate nonalcoholic steatohepatitis by modulating IRE1α-mediated JNK and NF-κB pathways in ApoE–/– mice. J Ethnopharmacol 163:241–250. https://doi.org/10.1016/j.jep.2015.01.017
Mirzadeh M, Arianejad MR, Khedmat L (2020) Antioxidant, antiradical, and antimicrobial activities of polysaccharides obtained by microwave-assisted extraction method: a review. Carbohydr Polym 229:115421. https://doi.org/10.1016/j.carbpol.2019.115421
Otsuka H et al (2014) Aralin, a type II ribosome-inactivating protein from Aralia elata, exhibits selective anticancer activity through the processed form of a 110-kDa high-density lipoprotein-binding protein: a promising anticancer drug. Biochem Biophys Res Commun 453:117–123. https://doi.org/10.1016/j.bbrc.2014.09.067
Piechowiak T, Grzelak-Błaszczyk K, Bonikowski R, Balawejder M (2020) Optimization of extraction process of antioxidant compounds from yellow onion skin and their use in functional bread production. LWT 117:108614 https://doi.org/10.1016/j.lwt.2019.108614
Ramli NH, Yusup S, Quitain AT, Johari K, Kueh BWB (2019) Optimization of saponin extracts using microwave-assisted extraction as a sustainable biopesticide to reduce Pomacea canaliculata population in paddy cultivation. Sustain Chem Pharm 11:23–35. https://doi.org/10.1016/j.scp.2018.12.002
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biol Med 26:1231–1237. https://doi.org/10.1016/S0891-5849(98)00315-3
Ren Y, Chen Y, Hu B, Wu H, Lai F, Li X (2015) Microwave-assisted extraction and a new determination method for total steroid saponins from Dioscorea zingiberensis CH Wright. Steroids 104:145–152. https://doi.org/10.1016/j.steroids.2015.09.008
Rodriguez-Jasso RM, Mussatto SI, Pastrana L, Aguilar CN, Teixeira JA (2011) Microwave-assisted extraction of sulfated polysaccharides (fucoidan) from brown seaweed. Carbohydr Polym 86:1137–1144. https://doi.org/10.1016/j.carbpol.2011.06.006
Sarvin B, Stekolshchikova E, Rodin I, Stavrianidi A, Shpigun O (2018) Optimization and comparison of different techniques for complete extraction of saponins from T. terrestris. J Appl Res Med Aroma 8:75–82. https://doi.org/10.1016/j.jarmap.2017.12.002
Shikov AN, Pozharitskaya ON, Makarov VG (2016) Aralia elata var. mandshurica (Rupr. & Maxim.) J. Wen: an overview of pharmacological studies. Phytomedicine 23:1409–1421. https://doi.org/10.1016/j.phymed.2016.07.011
Sparg SG, Light ME, van Staden J (2004) Biological activities and distribution of plant saponins. J Ethnopharmacol 94:219–243. https://doi.org/10.1016/j.jep.2004.05.016
Suh SJ et al (2007) Triterpenoid saponin, oleanolic acid 3-O-β-d-glucopyranosyl(1 → 3)-α-l-rhamnopyranosyl(1 → 2)-α-l-arabinopyranoside (OA) from Aralia elata inhibits LPS-induced nitric oxide production by down-regulated NF-κB in raw 264.7 cells. Arch Biochem Biophys 467:227–233. https://doi.org/10.1016/j.abb.2007.08.025
Wang W, Yao G-D, Shang X-Y, Gao J-C, Zhang Y, Song S-J (2018) Eclalbasaponin I from Aralia elata (Miq.) Seem. reduces oxidative stress-induced neural cell death by autophagy activation. Biomed Pharmacother 97:152–161. https://doi.org/10.1016/j.biopha.2017.10.106
Wei M-C, Yang Y-C (2015) Kinetic studies for ultrasound-assisted supercritical carbon dioxide extraction of triterpenic acids from healthy tea ingredient Hedyotis diffusa and Hedyotis corymbosa. Sep Purif Technol 142:316–325. https://doi.org/10.1016/j.seppur.2015.01.008
Xie P, Huang L, Zhang C, Deng Y, Wang X, Cheng J (2019) Enhanced extraction of hydroxytyrosol, maslinic acid and oleanolic acid from olive pomace: process parameters, kinetics and thermodynamics, and greenness assessment. Food Chem 276:662–674. https://doi.org/10.1016/j.foodchem.2018.10.079
Yan M-M, Liu W, Fu Y-J, Zu Y-G, Chen C-Y, Luo M (2010) Optimisation of the microwave-assisted extraction process for four main astragalosides in Radix Astragali. Food Chem 119:1663–1670. https://doi.org/10.1016/j.foodchem.2009.09.021
Zeković Z et al (2017) Utilization of sage by-products as raw material for antioxidants recovery—Ultrasound versus microwave-assisted extraction. Ind Crop Prod 99:49–59. https://doi.org/10.1016/j.indcrop.2017.01.028
Zhang M, Liu G, Tang S, Song S, Yamashita K, Manabe M, Kodama H (2006) Effect of five triterpenoid compounds from the buds of Aralia elata on stimulus-induced superoxide generation, tyrosyl phosphorylation and translocation of cytosolic compounds to the cell membrane in human neutrophils. Planta Med 72:1216–1222. https://doi.org/10.1055/s-2006-951679
Zhang J, Lu S, Wang H, Zheng Q (2013a) Protective role of Aralia elata polysaccharide on mercury(II)-induced cardiovascular oxidative injury in rats. Int J Biol Macromol 59:301–304. https://doi.org/10.1016/j.ijbiomac.2013.04.047
Zhang J, Wang H, Xue Y, Zheng Q (2013b) Cardioprotective and antioxidant activities of a polysaccharide from the root bark of Aralia elata (Miq.) Seem. Carbohydr Polym 93:442–448. https://doi.org/10.1016/j.carbpol.2012.12.048
Zhang J, Wang H, Zheng Q (2013c) Cardioprotective effect of Aralia elata polysaccharide on myocardial ischemic reperfusion (IR) injury in rats. Int J Biol Macromol 59:328–332. https://doi.org/10.1016/j.ijbiomac.2013.04.060
Zhao C, Yang X, Tian H, Yang L (2020) An improved method to obtain essential oil, flavonols and proanthocyanidins from fresh Cinnamomum japonicum Sieb. leaves using solvent-free microwave-assisted distillation followed by homogenate extraction Arabian. J Chem 13:2041–2052. https://doi.org/10.1016/j.arabjc.2018.03.002
Acknowledgements
This work was supported by the National Key R&D Program of the 13th Five-Year Project of China: Research, Development, and Demonstration of the Technology for Ecological Sustainability of Typical Mountain Vegetable Resources in Forests of Northeast China (2016YFC0500307). The first-class discipline team of Northeast Agricultural University, “Storage, Transportation and Processing of Forest Fruits and Vegetables”, is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tian, Yq., Zhao, Ht., Zhang, Xl. et al. Comparison of different extraction techniques and optimization of the microwave-assisted extraction of saponins from Aralia elata (Miq.) Seem fruits and rachises. Chem. Pap. 74, 3077–3087 (2020). https://doi.org/10.1007/s11696-020-01140-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-020-01140-2